Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
finally someone bought "1" share.......wow!!... stupid co and CEOs, morons in the management can buy stocks right, they never touched it?....and zero volume for months and years!?...suckers
Under the Radar, grossly neglected and not known stock!, this sleeping Penny is long due for a Run, with the new CEO announcement, the mgmt made a good move!..
CVR Medical Corp. Announces New Chief Executive Officer
Newsfile Corp - Thu Sep 2, 8:01AM CDT
Vancouver, British Columbia--(Newsfile Corp. - September 2, 2021) - CVR Medical Corp. (TSXV: CVM) (OTC Pink: CRRVF) (" CVR Medical " or the " Company " or " we ") a Canadian listed and US based healthcare company in the medical device market is pleased to announce the appointment of Darren Day as Chief Executive Officer (CEO), effective immediately.
Leadership issues I think. Not sure.
So what up is this a scam?
Check out news tab.
Any one see the news regarding the CVM Medical/Global shares and property split. Pretty interesting.
Good idea, first write to the company CEO or management/investors dept, if you do not get any informative reply, sure OTC may have answer.
it is plausible, companies can publish or make a release as they desired, but OTC supposed to post filings news..!. any SEC filings too will show up!..
Can a company file filings with the OTC markets and not have them published publicly? I am guessing that is the only way that they would have gotten pink status is if they have filed but for some reason they have an arrangement to not have them posted on their page. I may email OTC markets and ask them about that.
From CEOs message, it is possible they are holding on to the press release or any filing, as restructuring effort is ongoing!?...it might come out after they complete or come to a decision on restructuring, this is just my view point or Opinion only!.
Good PR, still confused how they are pink current now without filing anything. LOL Will be one to watch.
Joe,
there is a recent message from CEO, and it is quite interesting because it is the latest update, reveals the future potential in this co.
https://www.barchart.com/story/stocks/quotes/CRRVF/overview/1104673/cvr-medical-corp-message-from-the-chief-executive-officer
CEO and other stakeholders are working on a restructuring, the details are not provided, but it sounds pretty favorable for enhanced share performance!!..
They are pink current effective 2/19/2021, don't know why. It doesn't appear they have filed anything.
https://www.otcmarkets.com/market-activity/corporate-actions
Change the drop down from symbol changes to tier changes.
Oh well, they just filled it!? can't believe!!.....
it seems the stock is active and brokerage is allowing it to be traded!!...
No Bid, No Ask, and fidelity never fills the orders?....
Oh well, that Grey market sign on OTC...!
what is going on?...
is this co is still active and alive?
Nice find.
Stock up today.
Details....
Summary
Show definitions
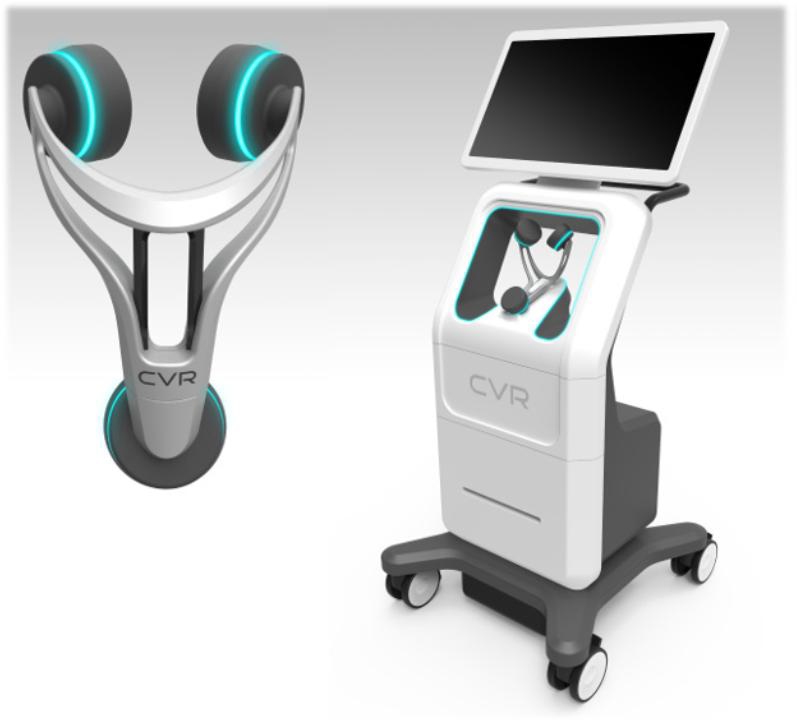
The purpose of this study is to determine the accuracy of a new non-invasive device, the Carotid Stenotic Scan (CSS), to check for stenosis of the internal carotid artery (ICA) as compared to a carotid duplex ultrasound study.
Description
Currently, carotid duplex ultrasound is the primary diagnostic tool for the evaluation for ICA stenosis and must be performed by a trained and certified vascular technologist using advanced duplex imaging equipment and with subsequent interpretation by a trained physician. this test is not considered suitable for screening for disease. It would be of value to develop an accurate, reliable, low-cost, and easily accessible tool to screen for extra-cranial ICA disease in an office based setting. However, such a tool would require novel technology that allows for quick, accurate, reproducible, and safe evaluation.
This study will evaluate a new technology called the Carotid Stenotic Scan (CSS) developed by CVR Medical. The CSS instrument uses sensitive transducers to detect low frequency pressure fluctuations associated with flow disturbances downstream from areas of arterial narrowing.
Subjects will undergo a clinically ordered clinical carotid duplex ultrasound as part of standard of care. Subjects will have a noninvasive CSS assessment either before or after the carotid duplex ultrasound.
Details
Treatment Carotid Stenotic Scan
Clinical Study Identifier NCT04057183
Sponsor Phillip J Bendick
Last Modified on 7 December 2020
Wondering about the price rise? Heres why.....
Human Trials Started
Suspended pending news - Voluntary
Your opinion is your opinion. I am not in pennies because I think I am going to get rich. I just am hopeful because the technology is promising. Hope you feel good now you got your negative crap off your chest. I am currently not interested in what you have to say unless you can have a civil discussion. GLTA.
What for Bigbet . . .perpetual disappointment . . .total loss of investment . . .you are completely deluded if you think the shower of sh*t that makes up this board of directors has the ability to make this turd smell nice!!
Looks like it's going sub penny soon.
Still not impressed.
I’m ready......
CVR Medical(CVM) - Resumption of Trading on the TSX Today!
IIROC Trading Resumption - CVM
June 10 2020 - 04:41PM
PR Newswire (Canada)
? Print
VANCOUVER, BC, June 10, 2020 /CNW/ - Trading resumes in:
Company: CVR Medical Corp
TSX-Venture Symbol: CVM
Resumption (ET): 9:30 AM 6/11/2020
IIROC can make a decision to impose a temporary suspension (halt) of trading in a security of a publicly-listed company. Trading halts are implemented to ensure a fair and orderly market. IIROC is the national self-regulatory organization which oversees all investment dealers and trading activity on debt and equity marketplaces in Canada.
SOURCE Investment Industry Regulatory Organization of Canada (IIROC) - Halts/Resumptions
Copyright 2020 Canada NewsWire
CVR Medical (TSXV:CVM)
Historical Stock Chart
From May 2020 to Jun 2020 Click Here for more CVR Medical Charts.
CVR Medical (TSXV:CVM)
Historical Stock Chart
From Jun 2019 to Jun 2020 Click Here for more CVR Medical Charts.
CVR Medical (TSXV:CVM)
Intraday Stock Chart
Thursday 11 June 2020 Click Here for more CVR Medical Charts.
Still here. Patience is a virtue and this technology is disruptive.
CVR Medical (CVM:TSX)to be reinstated to TSX from Thursday.
Lets roll!
CVR Medical (CVM) Reinstated on the TSX
CVM Reinstatement
Your wish has been granted :)
"VANCOUVER, BC, June 9, 2020 /CNW/ -
TSX VENTURE COMPANIES
CVR MEDICAL CORP. ("CVM")
BULLETIN TYPE: Reinstated for Trading
BULLETIN DATE: June 9, 2020
TSX Venture Tier 2 Company
Further to the Exchange bulletin dated May 7, 2019; the Exchange has been advised that the Cease Trade Order issued by the British Columbia Securities Commission dated, May 6, 2019, has been revoked.
Further to the news release dated June 8, 2020, effective at the opening, Thursday, June 11, 2020, trading will be Reinstated in the securities of the Company."
CVR Medical re-admitted for trading on TSX
After reading through the PD's I find them to be going through a bumpy road. I don't really see anything positive with the CEO resignation and share issues. Not ready to buy any shares at the moment until they begin trading again on TSX and there is more ground made with the technology.
I’m still here waiting for the news. Solid product.
Device is still solid. Ex CEO Bakuma was inept because of ???! Share holders meeting this month. Any originals this board still here? Trading volume still great regardless.
Def positive news...I believe some dilution is taking place. Are the results enough to beat millions of dollars that go into preventative screenings? I think so... seems as if it’s an uphill battle
CVR Medical Investor Overview on Wesite. Advised FDA quest for approval starts again Jan.1st 2020. De Novo still in gear. Canon still in play. Positive news.
Ant doesn’t know what he is talking about. No other posts just trying to stir up anxiety.
What was the statement? Link? TIA
The statement put out was disgraceful. Bakema should be fired and there should be a lawsuit against him for fraud.
Canadian side longs have had it rough! What I can glean from a few posts from them...reasonable and substantive...FDA wants more statistics re broader range regarding race, preconditions,etc which may already have begun. So my range is a guesstamation of time table.
Hi gc2,
How do you reckon the time frame for FDA comeback?
Predict Money Managers will dance this down to .009 and below before FDA news within the month. YUMMY!
Nope! Added some, sold none.
Approval on the way and when it does....BANG!
This looks so tasty! Wow
Imagine none of our serious longs have sold this low. Money managers have danced this down. No other logical explanations! Trying to scrounge some more funds. A failing stock would not have this volume..300$ would nail 0ver 13000 shares with .0225 pps!!!!!
Imagine none of our serious longs have sold this low. Money managers have danced this down. No other logical explanations! Trying to scrounge some more funds. A failing stock would not have this volume..300$ would nail 0ver 13000 shares with .0225 pps!!!!!
|
Followers
|
17
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
486
|
|
Created
|
03/16/17
|
Type
|
Free
|
| Moderators | |||









Michael Rhodes
Vice President of Quality Systems
•20 Years of Quality Know-how
•Former Vice President of Quality for HSBC & Motorola
•Experience in multiple markets including healthcare, defense, public safety, retail, energy, and manufacturin

Dr. W. Douglas Weaver
CVR Global Board of Directors / Scientific Advisory Board
•Past President of the American College of Cardiology
•Former Vice President and System Medical Director of Heart and Vascular Services at Henry Ford Health System
•Over 330 publications, most of which are related to drug and device discovery and methods toward improving the care provided to patients with cardiovascular disease

| Shares on Issue: | 73,832,151 |
| Options: | 4,050,000 |
| Warrants ($0.40): | 6,382,949 (Expires September 15, 2018) |
| Warrants ($0.70): | 2,197,591 (Expires August 22, 2018) |
| Warrants ($0.70): | 3,261,120 (Expires October 19, 2018)) |
| Warrants ($0.70): | 830,000 (Expires October 22, 2018) |
| Warrants ($0.70): | 444,600 (Expires February 26, 2019) |
| Warrants ($0.70): | 408,286 (Expires March 6, 2019) |
| Warrants ($0.70): | 107,500 (Expires March 16, 2019) |
| Warrants ($0.70): | 712,823 (Expires March 21, 2019) |
| Warrants ($0.70): | 700,000 (Expires May 02, 2019) |
| Warrants ($0.70): | 855,193 (Expires June 18, 2019) |
| Warrants ($1.50): | 2,518,600 (Expires April 6, 2020) |
| Trading Symbols: | TSX.V: CVM FRANKFURT: B3BN OTCQB: CRRVF |
| CUSIP #: | 126632108 |
| ISIN #: | CA1266321084 |
| Transfer Agents: | AST Trust Company (Canada)1600 – 1066 West Hastings St. American Stock Transfer & Trust Company, LLC6201 15th Avenue |
| IR Contact: | Brisco Capital Partners Corp.Scott Koyich, President |
CVR Intellectual Property Summary & Legal Comment
CVR has instituted a comprehensive IP policy to cover several aspects of the CSS device as it prepares to go to market. The IP includes 1 issued US patent, US 9,101,274. An European Counterpart is pending at the moment that parallels the allowed US case. A continuation-in-part, Application No. 14/803,389 is also pending, with a priority date extending back to June 24, 2010.
In June of 2015, CVR file two applications covering the Yoke, utilized for holding the sensors and positioning on a patient, and methods of quantifying and detecting sounds in the carotid artery. Both applications were converted into PCT applications in June of 2016 and now await conversion into the US and other jurisdictions at the end of 2017. These applications have been assigned to a joint venture, but an exclusive license remains with CVR.
In June of 2016, CVR filed five additional provisional patent applications covering several aspects of the CSS device, including additional methods for detecting blockage in a fluid flow vessel, disposable sensing elements (think of the razor/razor blade model), methods for generating clean data for analysis, methods for detecting sounds and determining stenosis, methods for determining appropriate location for detecting fluid flow in the body, and methods for detecting and ablating stenosis.
In December of 2016, CVR filed an additional provisional application directed towards sensing pods that do not need a yoke, in order to position the sensing pods for determining stenosis.
Finally, in January of 2017, CVR filed two design patents, one directed towards the device cart itself, and the second to a version of the Yoke for positioning sensors on a patient.
A summary of these are included in the attached Excel file. These filings are highly confidential and not to be disseminated without authorization by CVR.
CVR continues to develop and expand its IP portfolio as development continues. It is expected that additional applications will be filed to cover final versions of any product that will go to market. Such protection will include patent, design patent, trademark, trade dress, and copyright, where applicable.
CVR also expects that the several pending provisional applications will be converted into PCT applications and that the pending PCT applications (now assigned to a joint venture), will also be converted in due course.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
