Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
something smells fish here. where is the big power hour breakout? hhm...
Wow!! 134million Volume with a 7million Float. Yet it's not going straight up??
Im seeing a late day run and close at $8
Done until after hours...
May go sub $7 here...shorts running the show.
Market cap only $50M right now. Should be at atleast $100-200M+ pre-approval
Only matter of time until it breaks $9.00
I know damn sellers let’s push it hard
Etrade extended hours trading and it's commission free on these bigger non OTC stocks. There is no premarket trading on OTC only the bigger boards. Looks like it was a mistake to sell at 6.50 premarket, but these NASDAQ ones seem to commonly run premarket and then crash and I wasn't chancing that!! Still a great gain.
its in new price discovery process. hard to find an accurate share count
OS around 7.5 million with float as high as 6.1 million
either way, the volume is ROCKING!!!!!
will be a new liquid stock for at least all of June IMO
whaaaa happen?
I did a 40k market buy the other day that filled at 3.57 we'll see what happens
Can't seem to hold $8.00 break.
Yup waiting and loading
This can EASILY... EASILY run over $10 it just needs a push
This thing is so coiled up, once she busts thru $9 its gonna be insane
80 million volume! over 25x the float
and its not even lunch
are you high it halts up one there will be no hope here
More Eyes on BCTX as volume picks up!
We’re trading 20 times the OS of 6.7M ?
And we can’t get past $10 ?
THE SQUEEZE IS GOING TO BE INSANE.
The real fun is when she gets over $10. This could be the most epic run ever.
The warrants BCTXW are worth over $3 per share right now and can be exercised immediately.
Mike the market cap only $40M right now should be worth over $400M
Warrants lookin even yummier at only $2.04 now
Check out BCTXW the warrants can be exercised at $5.42
With this kind of news could easily see $20+
Market cap only 40M??? WOWWWWWWWWWWWW
we going HIGHER!!!!!!!!!
31 million volume already. 10x the float
https://finviz.com/quote.ashx?t=bctx
would like to see this continue into $10+
glad i was still holding this. great morning surprise
Lol, how is this news “fluff”
Hi King. Which broker are you using to buy and sell premarket? Do they charge commission fees per transaction? TIA
Hi King. Which broker are you using to buy and sell premarket? Do they charge commission fees per transaction? TIA
6.50 premarket. I sold some there with extended market hours trading
There are similarities in the chart: SKLZ hit $46.30 in Feb so it's not that outlandish.
https://schrts.co/jsGNHavB
|
Followers
|
20
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
331
|
|
Created
|
03/14/15
|
Type
|
Free
|
| Moderators | |||
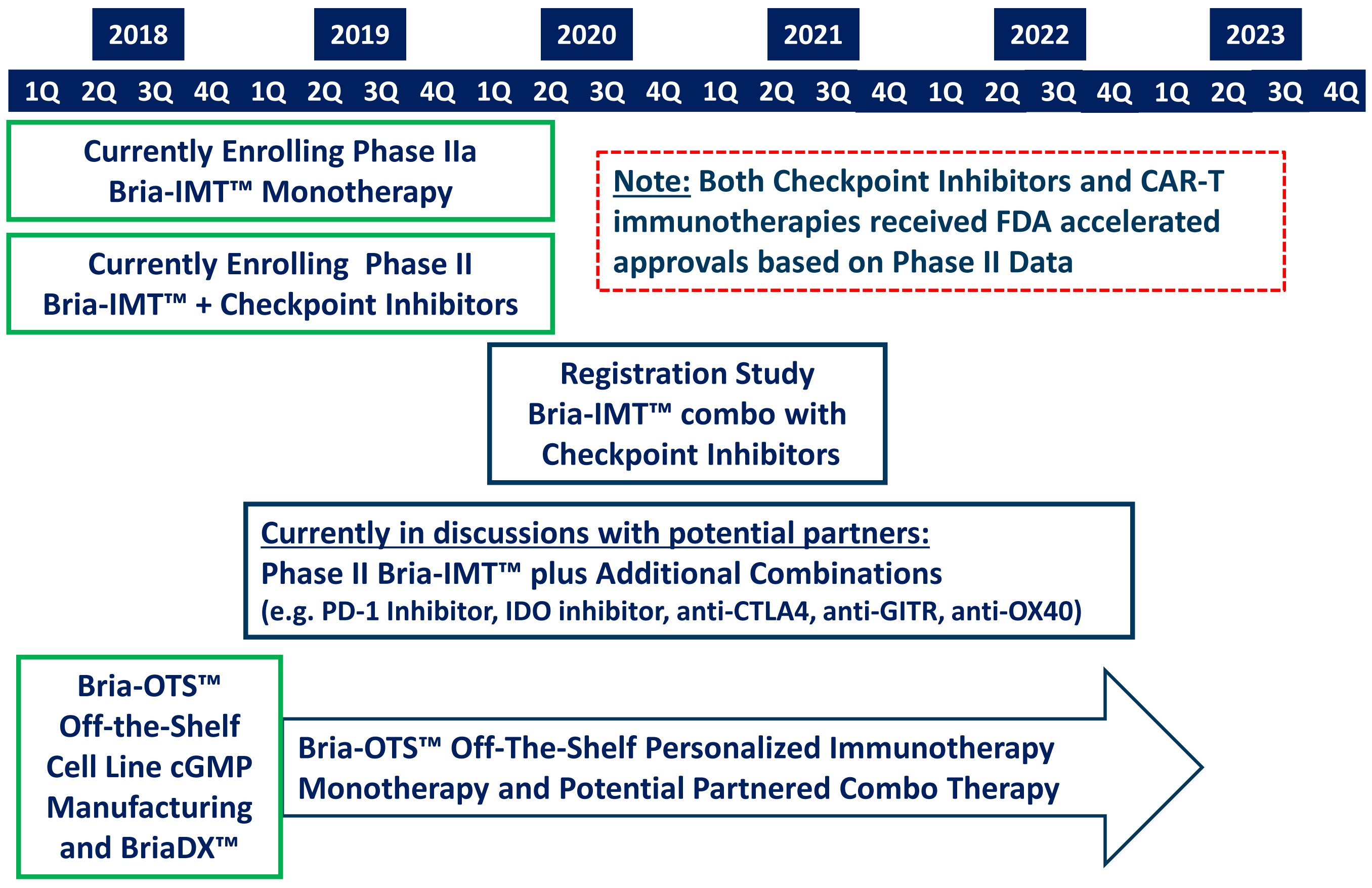
Dedicated to enhance the lives of cancer patients who are facing limited therapeutic options, BriaCell Therapeutics Corp. (OTCQB: BCTXF; TSX: BCT.V)’s mission has been to develop novel immunotherapies, as the most cutting edge technology to fight cancer. Immunotherapies have become the forefront of the cancer treatments because they use the body’s immune system to destroy the cancer cells, offering higher levels of safety and efficacy than chemotherapy, with less likeliness of recurrence.
Designed by our team of scientists and clinicians, BriaCell’s proprietary whole-cell based vaccine technology platform continues to show its impressive potential to establish a new model for treating cancer patients. Our lead product candidate, Bria-IMT™ (SV-BR-1-GM), is a genetically engineered whole-cell vaccine derived from a human breast cancer cell line. Bria-IMT™ is used in combination with multiple immune-modulators to powerfully trigger the immune system to recognize and eliminate cancerous cells.
In a preliminary Phase I clinical study in metastatic (i.e., Stage IV) breast cancer patients who had failed multiple treatments, Bria-IMT™ treatment significantly reduced the tumor size, without serious side effects. Importantly, the tumor regression was observed in other sites including the lung and even the brain -a difficult site. Impressively, the median lifespan of the patients was substantially longer than that of the comparable trials.
At our laboratory facility in Berkeley, CA, we are planning to expand our pipeline of oncology immunotherapy candidates using our proprietary technology platform.
Using the clinical data from the patients from the clinical studies, we are working to identify the molecular fingerprint of the patients for which the vaccine would be highly effective, and are planning to develop diagnostic testing products to identify this group. By directing the drug to the top-responders, we expect this approach to increase the likelihood of clinical trial success of Bria-IMT™ -to bring hope to thousands of cancer patients with few-to-no treatment options.
The company is currently recruiting its Phase I/IIa trial to further test Bria-IMT™ safety and activity in metastatic breast cancer patients. BriaCell maintains global rights for Bria-IMT™.
BriaCell is currently recruiting patients to evaluate the safety and activity of Bria-IMT™ in an additional Phase I/IIa study in 25-40 metastatic breast cancer patients who have failed at least one course of treatment.
For the details of the trial, please visit For the details of the trial, please visit https://www.clinicaltrials.gov/ct2/show/NCT03066947
FDA has approved the roll-over combination study of Bria-IMT™ with pembrolizumab (Keytruda; manufactured by Merck & Co., Inc.) or ipilimumab (Yervoy; manufactured by Bristol-Myers Squibb Company) for patients previously treated with BriaVax™ from the ongoing Phase I/IIa Clinical Trial in Advanced Breast Cancer. The study is available for patents on the SV-BR-1-GM Phase I/IIa study who develop progressive disease. These patients will be eligible to roll-over into combination therapy with Keytruda or Yervoy, depending on the type of breast cancer they have.
More information on the roll-over combination study of Bria-IMT™ with either ipilimumab or pembrolizumab will be available on ClinicalTrials.gov (https://clinicaltrials.gov/ct2/show/NCT03328026)
Manufactured by Merck & Co., Inc., KEYTRUDA® (pembrolizumab) is a prescription medicine that may treat certain cancers by working with the immune system. It has been approved for the treatment of a number of cancer indications excluding breast cancer.
For more information on pembrolizumab, please see:
http://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf
Manufactured by Bristol-Myers Squibb Company, YERVOY® (ipilimumab) is a prescription medicine used in adults and children 12 years and older to treat melanoma (a kind of skin cancer) that has spread (metastatic) or cannot be removed by surgery (unresectable). It is a monoclonal antibody that works to activate the immune system and enabling them to recognize and destroy cancer cells.
For more informations on Ipilimumab, please see:
https://packageinserts.bms.com/pi/pi_yervoy.pdf
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
