Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
EYES.
Second Sight Medical Products, Inc. to Present at the B. Riley & Co. China Healthcare Investment Partnering Symposium
Print
Alert
SECOND SIGHT MEDICAL PRODUCTS IN (NASDAQ:EYES)
Intraday Stock Chart
Today : Thursday 8 March 2018
Click Here for more SECOND SIGHT MEDICAL PRODUCTS IN Charts.
Second Sight Medical Products, Inc. (NASDAQ: EYES) ("Second Sight" or "the Company"), a developer, manufacturer and marketer of implantable visual prosthetics that are intended to create an artificial form of useful vision to blind patients, today announced that Will McGuire, President and Chief Executive Officer, will present at the B. Riley & Co. China Healthcare Investment Partnering Symposium as follows:
Date: Friday, March 16, 2018
Presentation Time: 11:30 a.m. China Standard Time
Location: InterContinental Hangzhou - Hangzhou, China
Webcast: http://wsw.com/webcast/brileyco20/eyes/
ADVM.
Adverum Biotechnologies Reports Fourth Quarter 2017 Financial Results and Provides Corporate Update
Print
Alert
Adverum Biotechnologies, Inc. (NASDAQ:ADVM)
Intraday Stock Chart
Today : Tuesday 6 March 2018
Click Here for more Adverum Biotechnologies, Inc. Charts.
Adverum Biotechnologies, Inc. (Nasdaq:ADVM), a clinical-stage gene therapy company targeting unmet medical needs in serious rare and ocular diseases, today reported financial results for the fourth quarter ended December 31, 2017 and provided a corporate update.
“We enter 2018 with significant momentum following a year of critical execution to transform Adverum into a clinical-stage company,” said Amber Salzman, Ph.D., president and chief executive officer of Adverum Biotechnologies. “In ADVANCE, our Phase 1/2 clinical trial of ADVM-043 in alpha-1 antitrypsin deficiency, we have completed enrollment in the first dose cohort and have initiated patient enrollment in the second intermediate-dose cohort. We are on track to report preliminary data from this study in the second half of 2018. In addition, we plan to submit two Investigational New Drug Applications with the FDA in the second half of 2018, for ADVM-022 in wet AMD and ADVM-053 in hereditary angioedema. We begin this exciting year of clinical development and regulatory progress in a strong position, funded to execute our three lead programs through the end of 2019 with preliminary clinical data for at least two of these programs.”
Recent Progress
In February 2018, Adverum completed the dosing and evaluation of patients (n=2) in the first cohort of the ADVANCE Phase 1/2 trial for ADVM-043 in alpha-1 antitrypsin (A1AT) deficiency. Based on a review of the preliminary safety information, the independent data monitoring committee (DMC) recommended proceeding to the second cohort of patients, which is open for enrollment. Adverum has initiated patient enrollment in the second intermediate-dose cohort, which will receive an intermediate dose of ~5E12 vg/kg (equivalent to ~4E14 total vg based on an 80-kg patient). The primary endpoint is safety and tolerability, and secondary endpoints include changes in plasma concentrations of both total and M-specific A1AT levels. Additional information about this clinical trial can be found at ClinicalTrials.gov under trial identifier number NCT02168686.
In February 2018, Adverum raised $69.0 million in gross proceeds from an underwritten public offering of its common stock.
In January 2018, Adverum and Editas Medicine, Inc. announced the extension of the companies’ collaboration agreement to explore the delivery of genome editing medicines to treat up to five inherited retinal diseases.
2018 Outlook - Planned Pipeline Milestones
ADVM-043 for A1AT Deficiency
Report preliminary data from the ADVANCE Phase 1/2 clinical trial in the second half of 2018.
ADVM-022 for wAMD
Report 12-month efficacy data in non-human primates in the first half of 2018.
Complete Investigational New Drug (IND)-enabling preclinical studies.
Submit an IND Application to the U.S. Food and Drug Administration (FDA) in the second half of 2018.
ADVM-053 for Hereditary Angioedema (HAE)
Complete IND-enabling preclinical studies.
Submit an IND Application to the FDA in the second half of 2018.
Upcoming Events
Adverum plans to attend the following upcoming conferences:
° Cowen 38th Annual Health Care Conference in Boston on March 12, 2018 at 1:30 pm ET
° Cowen 17th Annual Life Sciences Winter Meeting in Colorado, March 20-23, 2018
° ARM Cell & Gene Therapy Investor Day in New York, April 17, 2018
° ARVO 2018 Annual Meeting in Honolulu, April 29-May 3, 2018
• Poster titled “Therapeutic potential and safety of sequential intravitreal dosing to the contralateral eye of novel AAV vectors in non-human primates” on May 3, 2018, 8:15 – 10:00 am HST
• Poster titled “Long-term functional delivery of the human L-opsin cDNA via intravitreal administration of an AAV vector in Mongolian gerbils” on May 3, 2018, 8:15 - 10:00 am HST
° 2nd Annual Gene Therapy for Rare Disorders 2018 Meeting in Boston, April 30-May 2, 2018
° ASGCT 21st Annual Meeting in Chicago, May 16-19, 2018
Financial Results for the Three Months Ended December 31, 2017
Cash, cash equivalents and marketable securities were $190.5 million as of December 31, 2017, compared to $186.6 million as of September 30, 2017 and $222.2 million as of December 31, 2016. The year-end cash position, added with approximately 64 million in net proceeds raised in February 2018, is expected to fund the three lead gene therapy programs through the end of 2019, including preliminary clinical data for at least two of these programs, and through the initial stage of scaling up manufacturing capabilities.
Revenues, consisting of revenue from collaborative research, were $0.5 million for the three months ended December 31, 2017, compared to $0.5 million for the same period in 2016.
Research and development expenses were $12.0 million for the three months ended December 31, 2017, compared to $7.9 million for the same period in 2016. This increase was due to an overall increase in research and development activities for the Company’s gene therapy programs, primarily for material production costs for the ADVANCE clinical trial for ADVM-043.
General and administrative expenses were $4.0 million for the three months ended December 31, 2017, compared to $4.8 million for the same period in 2016. This decrease was primarily due to lower legal fees.
Net loss attributable to common stockholders was $14.8 million, or $0.32 per basic and diluted share, for the three months ended December 31, 2017, compared to $22.4 million, or $0.54 per basic and diluted share, for the same period in 2016.
Shares of common stock outstanding were 62.2 million as of February 28, 2018.
EYES.
Second Sight Announces Latest Expansion of Medicare Coverage for Argus II Retinal Prosthesis System
Print
Alert
SECOND SIGHT MEDICAL PRODUCTS IN (NASDAQ:EYES)
Intraday Stock Chart
Today : Tuesday 6 March 2018
Click Here for more SECOND SIGHT MEDICAL PRODUCTS IN Charts.
-- Argus II Covered in Majority of Medicare Administrative Contractor Jurisdictions Across 31 States --
Second Sight Medical Products, Inc. (NASDAQ:EYES) ("Second Sight" or "the Company"), a developer, manufacturer and marketer of implantable visual prosthetics intended to create an artificial form of useful vision for blind individuals, today announced that Palmetto GBA (Jurisdiction JJ) is the latest Medicare Administrative Contractor (MAC) to provide coverage for the Argus® II Retinal Prosthesis System (Argus II) and the related surgical procedure. As a result of this decision, coverage for the Argus II will now include Alabama, Georgia and Tennessee. This brings the total coverage of the Argus II to 31 states, two territories and the District of Columbia.
“We are pleased that blind individuals with Retinitis Pigmentosa covered by Medicare now have greater access to the Argus II with five of seven MACs nationwide covering Argus II. We continue to work with the few remaining MACs and payers towards the goal of achieving full coverage in the U.S. The significant momentum in this effort reflects recognition of our technology as the standard of care for these blind individuals,” said Will McGuire, President and CEO of Second Sight.
Effective February 26, 2018, Palmetto GBA (Jurisdiction JJ), published a local coverage article on the Argus II placement procedure (0100T). This decision authorizes coverage of Argus II under the Local Coverage Article A53044, when medically necessary, to Medicare beneficiaries in Alabama, Georgia and Tennessee.
BCRX. BioLineRx Ltd. (BLRX)
Highlights and achievements in 2017 and to date:
Continued progress and execution according to plan on multiple clinical trials for the Company's lead oncology program, BL-8040:
•Initiation of pivotal Phase 3 GENESIS study with BL-8040 as novel stem cell mobilization treatment for autologous bone-marrow transplantation, following successful meeting with the FDA earlier in the year;
•Partial monotherapy results from Phase 2a COMBAT study, investigating the combination of BL-8040 and Merck's PD-1 inhibitor, Keytruda® (pembrolizumab), in pancreatic cancer, showed significantly increased infiltration of T cells into the tumor, as well as robust mobilization of immune cells;
•Initiation of three Phase 1b/2 studies under collaboration with Genentech, exploring the combination of BL-8040 with Tecentriq® (atezolizumab), Genentech's anti-PD-L1 cancer immunotherapy agent;
•Overall long-term survival results in Phase 2a trial in relapsed/refractory AML demonstrated that the combination of BL-8040 with high-dose Ara-C (HiDAC) significantly improved overall survival, compared with historical data of HiDAC monotherapy;
•Partial results of Phase 2 study for BL-8040 as novel stem cell mobilization treatment for allogeneic bone-marrow transplantation support BL-8040 as a one-day dosing regimen for rapid mobilization of stem cells.
The Company also announced progress in expanding and accelerating its growth potential and strengthening its balance sheet:
•Acquired Agalimmune Ltd., a UK-based biopharmaceutical company developing cancer immunotherapy treatments, thereby broadening BioLineRx's position in the immuno-oncology field with a second novel lead compound, AGI-134. Pre-clinical data presented at ASCO-SITC showed complete tumor regression in the majority of mice treated with AGI-134;
•Completed underwritten public offering of American Depository Shares for gross proceeds of $28.9 million led by BVF Partners, L.P; the Company also received an additional $9.6 million direct investment from BVF Partners.
Expected significant upcoming milestones for 2018:
•Top-line results in immuno-oncology Phase 2a COMBAT study in pancreatic cancer for BL-8040 in combination with Merck's KEYTRUDA, expected in H2 2018;
•Results from the lead-in stage of the Phase 3 GENESIS study in stem-cell mobilization, expected in H2 2018;
•Initiation of Phase 1b/2 immuno-oncology study for BL-8040 in combination with Genentech's atezolizumab for non-small cell lung cancer. Partial results in Phase 1b/2 trials under collaboration with Genentech expected in H2 2018;
•Initiation of Phase 1/2a immuno-oncology study for AGI-134 in several solid tumor indications expected in mid-2018;
•Top-line results of Phase 2 study for BL-8040 in stem-cell mobilization for allogeneic transplantation expected by mid-2018.
https://ih.advfn.com/p.php?pid=nmona&article=76873656
Form 20-F
https://www.sec.gov/Archives/edgar/data/1498403/000117891318000765/zk1821295.htm
http://www.biolinerx.com/
EYES. webcast at the LD Micro Virtual Investor Conference, as follows:
Date: Wednesday, March 7, 2018
Presentation Time: 3:30 p.m. Pacific Standard Time
Webcast: http://www.investorcalendar.com/console/conference/?id=26039
ZIOP. Earnings tomorrow. Hopeing for something good on call. They are way over due for something big. Hopefully IL 12 data or a partner deal.
Oncolix, OTCQB: ONCX is a pharmaceutical company that is in phase one, of FDA clinical human trials for Cancer treatment.
Early funding for this research came from the Susan G. Komen Foundation and the U.S. Government and Oncolix maintains their connection with MD Anderson Treatment Centers.
Oncolix as a private company rose over $15,000,000 through several rounds of private offerings that are only available to accredited investors, family trusts, endowments and institutional investors.
Oncolix officers have agreed to have roughly $1,300,000 in compensation deferred and have also agreed to lock up their shares along with their principal shareholder until late 2018.
Greenville Health System is the principal shareholder.
GHS is not-for-profit healthcare network serving the medical needs of their communities for over a century and currently has over 15,000 employees.
Current President of GHS is Spence M. Taylor MD, who is the custodian of ONCX shares on behalf of Greenville.
Accredited investors include the Texas Emerging Technology Fund and the Ernest Mario Family Trust among others.
Ernest Mario served as Chief Executive Officer of Glaxo Holdings, plc, based out of London, England, known today as GlaxoSmithKline NYSE:GSK.
Ernest Mario was also the Chairman and CEO of Alza Corporation, based in Palo Alto, CA, from 1993 until the company was purchased by Johnson & Johnson, Inc, NYSE: JNJ in 2001.
The biographies of Oncolix officers, Board of Directors and company advisors have been released to the public and are available on the company website.
The Oncolix Team.
Management


Board Members


Prolanta Inventor

Company Advisors


Oncolix has a worldwide patent portfolio that is also available to view on the Oncolix website.
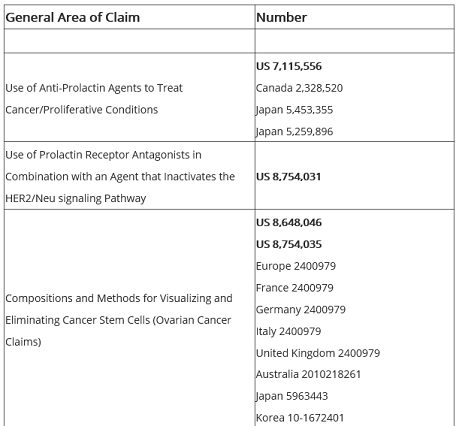
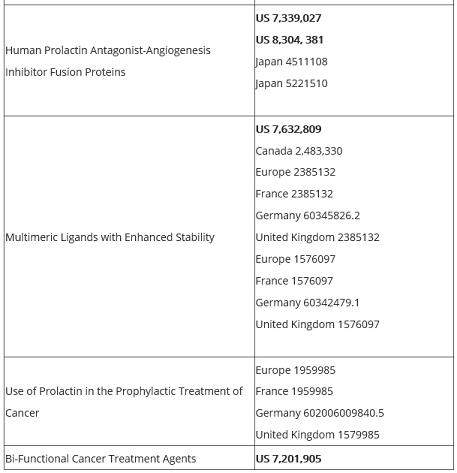
Leveraging these worldwide patents, Oncolix could be in a position to realize substantial gains in revenue and market capitalization.
IMO and FWIW.
DCAR was mentioned on CNBC expose on RIOT today. Be careful imo
BLPH. Large owner. https://ih.advfn.com/p.php?pid=nmona&article=76719923&symbol=BLPH
ZIOP new presentation. http://www.ziopharm.com/pipeline/presentations-publications
IPCI news...they finally received FDA approval to begin oral and nasal studies on their OxyContin product. Of interesting note said studies are supposed to take 6 months then they're gonna resubmit their oxy NDA to the FDA this August
EYES. Second Sight Medical Products, Inc. (NASDAQ:EYES) ("Second Sight" or "the Company"), a developer, manufacturer and marketer of implantable visual prosthetics that are intended to create an artificial form of useful vision to blind patients, today provided an update on several key performance metrics, demonstrating the Company’s continued progress in executing its near- and long-term goals.
Business Highlights as of January 31, 2018:
Implanted 30 Argus® II Retinal Prosthesis Systems worldwide during the fourth quarter of 2017 for a total of 75 implants in 2017. This compares to seven implants in the fourth quarter of 2016 and a total of 42 implants in 2016, representing implant volume growth of 329% and 79%, respectively;
The first human patient was implanted with the Orion™ Cortical Visual Prosthesis System (Orion) by Nader Pouratian, MD, PhD, Vice Chairman of the Department of Neurosurgery at the Ronald Reagan UCLA Medical Center (UCLA) in late January as part of Second Sight’s feasibility clinical study;
Enrollment of the first patient(s) is expected during Q1 2018 in a clinical study of better-sighted patients with Retinitis Pigmentosa (RP) in Germany. Two sites are actively screening subjects with a third site to begin screening shortly;
Added a new Center of Excellence in Houston, TX in early January, further validating the success of the Company’s business model that emphasizes close partnering with implanting sites; and,
Reimbursement for the Argus II in Germany was renewed for the 2018 calendar year, marking the eighth year that Argus II implants have been reimbursed through the NUB program.
“We are excited about our milestones and achievements in 2017, which speak to the strength of our business and the momentum we expect for continued success in 2018,” said Will McGuire, President and CEO of Second Sight.
“The first-in-human implant of the Orion is a significant milestone for Second Sight, and a critical step forward in meeting our Company’s mission to develop devices that could potentially treat nearly all forms of blindness. We expect that an additional four patients will be included in this feasibility study at UCLA and the Baylor College of Medicine in Houston,” stated Dr. Robert Greenberg, Chairman of the Board.
“We continue to advance our Argus II R&D efforts and have initiated screening for a clinical trial in better-sighted RP patients. Second Sight is committed to executing on our strategy to expand the availability of the Argus II to a larger patient population and explore the potential of our technology to treat those suffering from other forms of blindness. We look forward to updating investors on our continued progress,” concluded McGuire.
About the Argus II Retinal Prosthesis System
Second Sight's Argus II System provides electrical stimulation that bypasses the defunct retinal cells and stimulates remaining viable cells inducing visual perception in individuals with severe to profound Retinitis Pigmentosa. The Argus II works by converting images captured by a miniature video camera mounted on the patient's glasses into a series of small electrical pulses, which are transmitted wirelessly to an array of electrodes implanted on the surface of the retina. These pulses stimulate the retina's remaining cells, intending to result in the perception of patterns of light in the brain. The patient must learn to interpret these visual patterns, having the potential to regain some visual function. The Argus II was the first artificial retina to receive widespread commercial approval, and is offered at approved centers in Canada, France, Germany, Italy, Russia, Saudi Arabia, South Korea, Spain, Taiwan, Turkey, the United Kingdom, and the United States. Further information on the long-term benefits and risks can be found in the peer reviewed paper at: http://www.sciencedirect.com/science/article/pii/S0161642016305796
About the Orion Visual Cortical Prosthesis System
Like the Argus II, the Orion converts images captured by a miniature video camera mounted on the patient's glasses into a series of small electrical pulses. The Orion is designed to transmit these electrical pulses wirelessly to an array of electrodes implanted on the surface of the visual cortex, intended to result in the perception of patterns of light. By bypassing the retina and optic nerve and directly stimulating the visual cortex, a cortical prosthesis system has the potential to restore useful vision to patients completely blinded due to many reasons, including glaucoma, diabetic retinopathy, or forms of cancer and trauma – which has the potential to treat many fold more patients than for the current Argus II indications. No clinical data is yet available for the Orion.
About Second Sight
Second Sight's mission is to develop, manufacture and market innovative implantable visual prosthetics to enable blind individuals to achieve greater independence. Second Sight has developed, and now manufactures and markets, the Argus® II Retinal Prosthesis System. Enrollment has been completed in a feasibility trial to test the safety and utility of the Argus II in individuals with Dry Age-Related Macular Degeneration. New hardware and software to improve the quality of the vision produced is underway. A clinical trial to study the Argus II in better-sighted subjects earlier in the disease was recently approved in Germany. Second Sight is also developing the Orion™ Visual Cortical Prosthesis to restore some vision to individuals who are blind due to causes other than preventable or treatable conditions. Second Sight’s U.S. Headquarters are in Sylmar, California, and European Headquarters are in Lausanne, Switzerland. For more information, please visit www.secondsight.com
This company has come a long way.
Their manufacturing plant is complete and Health Canada approved.
Maybe it's finally IGXT's time.
http://www.biotuesdays.com/briefs/2018/1/29/aegis-starts-intelgenx-at-buy-pt-3
An Alzheimer's phase II trial is looking good for IGXT.
Here's a link.
https://alzheimersnewstoday.com/2018/01/29/asthma-drug-montelukast-tested-as-alzheimers-treatment-in-proof-of-concept-trial/
NeuroMetrix Reports Q4 and Full Year 2017 Financial Results
GlobeNewswireJanuary 25, 2018
WALTHAM, Mass., Jan. 25, 2018 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (NURO), today reported financial and business highlights for the quarter ended December 31, 2017.
The Company develops and markets novel therapies, based on neurostimulation and digital medicine, for chronic health conditions. The Company’s primary product is Quell®, which is an over-the-counter wearable neurostimulation device for treating chronic pain. The Company also has a diagnostic business based on its DPNCheck® product, which is a point-of-care test that provides accurate and cost-effective screening, diagnosis and monitoring of diabetic peripheral neuropathy (DPN).
Highlights:
Q4 2017 revenue of $4.9 million grew 33% from $3.7 million in Q4 2016. Quell contributed $3.7 million and DPNCheck added $0.8 million revenue.
Full year 2017 revenue of $17.1 million grew 42% from $12.0 million in 2016. Quell contributed $12.4 million and DPNCheck contributed $3.1 million revenue.
Fourth quarter Quell shipments totaled 26,496 devices and 36,556 electrode reorder packages. This compared with 14,301 devices and 19,993 electrode reorder packages in Q4 2016.
Quell intellectual property assets were expanded in the fourth quarter with the issuance of a U.S. utility patent for novel technology that enhances Quell usability for those with an active lifestyle.
In early Q1 2018 the Company reported a strategic collaboration with GlaxoSmithKline (GSK) under which GSK Consumer Healthcare acquired Quell market rights outside the US while NeuroMetrix retained ownership for the US market. The companies will co-fund Quell technology development. GSK Consumer Healthcare paid $5 million for the Quell assets related to markets outside the US and will pay up to $21.5 million upon the achievement of certain development and commercialization milestones.
"Our revenue growth in Q4 and full year 2017 reflects strong Quell unit volume growth throughout the periods. It continues the upward trend of the past two and a half years since Quell launch in Q2 2015," said Shai N. Gozani, M.D., Ph.D., President and Chief Executive Officer of NeuroMetrix. "We are pleased to begin our partnership with GSK Consumer Healthcare. This will expand access to Quell technology among chronic pain sufferers around the world while we continue to focus on building Quell into a leading U.S. consumer healthcare brand. It will also strengthen our capital structure.”
Financial Highlights:
The Company reported its financial highlights results for Q4 2017. Total revenues were $4.9 million versus $3.7 million for Q4 2016, an increase of 33%. Gross margin was 42.0% of total revenues versus 45.5% in Q4 2016, reflecting an increased weighting of Quell in revenues. Operating expenses were $5.0 million compared to $4.6 million in Q4 2016. Net loss was approximately $2.9 million in Q4 2017 compared to $2.8 million in Q4 2016. Net cash usage in Q4 2017 was $3.3 million, an improvement from $3.6 million in Q4 2016. The Company ended Q4 2017 with cash and cash equivalents of $4.0 million. The GSK payment in early Q1 2018 described above, increased the Company’s cash resources to approximately $9.0 million on a pro forma basis.
For the years ended December 31, total revenues were $17.1 million in 2017 compared to $12.0 million in 2016. Net loss was $12.9 million in 2017, an improvement of $2.0 million from $14.9 million in 2016.
Company to Host Live Conference Call and Webcast
NeuroMetrix management will host a conference call today, January 25, 2018 at 8:00 a.m., Eastern Time. To access the call in the United States, dial 844-787-0799 and use the confirmation code 9869499. Internationally, the conference call may be accessed by dialing (661) 378-9630 and using the same confirmation code. The call will also be webcast and will be accessible from the Company's website at http://www.NeuroMetrix.com under the "Investor Relations" tab. A replay of the conference call will be available starting two hours after the call by dialing 855-859-2056, domestically and 404-537-3406, internationally. The confirmation code to access the replay is 9869499. The replay will be available for one week after the conference call.
BPMX- BPX-04 P2 Trials starts this qrt and BPX-01 worth billions and first of its kind approved by FDA in Nov/Dec of 2017 now actively evaluating Partnership to fund P3..
Volume has been growing massively over the past 2 weeks and mark my word a major pop is coming.
NMUS@.21 anyone like this ticker? GLTUA!!!
NURO. NeuroMetrix, Inc. Announces Date for Fourth Quarter and Full Year 2017 Financial Results Conference Call
11:40 am, January 18, 2018
Portfolios: Ptx
Tickers: NURO
Read the full article
WALTHAM, Mass., Jan. 18, 2018 (GLOBE NEWSWIRE) -- NeuroMetrix, Inc. (NASDAQ:NURO) announced today that it plans to issue its fourth quarter and full year 2017 financial results before the opening of the market on January 25, 2018. The Company will host a conference call at 8:00 a.m., Eastern Time on January 25, 2018 to discuss its financial results as well as business developments affecting the Company.
The conference call may be accessed in the United States by dialing 844-787-0799 and using the confirmation code 9869499. Internationally, the conference call may be accessed by dialing 661-378-9630 and using the same confirmation code. The earnings press release and accompanying condensed financial statements will be accessible from the Company's website at www.NeuroMetrix.com under the "Investor Relations" tab.
A replay of the conference call will be available starting two hours after the call by dialing 855-859-2056, domestically and 404-537-3406, internationally. The confirmation code to access the replay is 9869499. The replay will be available for one week after the conference call.
$SIGO is going to be a runner!
$SIGO can cultivate, manufacture, and soon to distribute.
California MJ Baby!
War
$SIGO should run up high in the morning! .79 a share what a steal!!!!! Get in as soon as you can a $3 stock near term!!!
So you are betting on good results?
ZIOP. Good stuff coming out of the JPM conference. https://www.investorvillage.com/smbd.asp?mb=16353&pt=m
LPCN ADCOM thread on Yahoo. https://finance.media.yahoo.com/quote/LPCN/community?p=LPCN
Not good for patients. Who ever gets the first cure will instantly become one of the largest MC companies. Pfizer ends research for new Alzheimer‘s, Parkinson‘s…
January 9, 2018 Staff Writer
ABIO $1.77 one of my fave pics going into phase 2 data in March
LPCN 3.60 AH. GL to us longs tomorrow!
ALERT-------ALERT --- $PPPMF ----- $PPPMF 500% to 1000% Gains possibly
NURO. NeuroMetrix Reports Preliminary 2017 Financial Highlights
WALTHAM, Mass., Jan. 8, 2018 (Globe Newswire) -- NeuroMetrix, Inc. (NASDAQ:NURO) today reported preliminary financial highlights for the fourth quarter and for the full year 2017.
GAAP revenue for the fourth quarter of 2017 is estimated at approximately $4.9 million in comparison with $3.7 million in Q4 2016. During the fourth quarter of 2017 the Company shipped approximately 26,500 Quell® devices and 36,600 electrode packages in comparison with 14,300 devices and 20,000 electrode packages in Q4 2016.
For the full year 2017, revenue is estimated at approximately $17.1 million in comparison with $12.0 million in 2016. Approximately 80,900 Quell devices were shipped in 2017 versus 45,700 devices in 2016. Also, approximately 121,400 electrode packages were shipped in 2017 versus 52,700 electrode packages in 2016. The Company reported approximately $4.0 million in cash and cash equivalents at the end of 2017.
“Early results indicate an over 40% revenue increase from 2016 reflecting strong Quell unit volume growth in the fourth quarter and for the full year 2017. This continues the upward trend we’ve reported since the product was launched in Q2 2015,” said Shai N. Gozani, M.D., Ph.D. President and CEO of NeuroMetrix, Inc.
Note Regarding Forward-Looking Statements
This press release contains estimates underlying the Q4 and full year 2017 financial results and other forward-looking statements. Actual results may differ from those indicated as a result of finalization of fourth quarter and full year financial statements as well as other risks and uncertainties, including those described from time to time in reports filed by NeuroMetrix with the Securities and Exchange Commission. NeuroMetrix undertakes no obligation to update information contained in this release. For further information regarding risks and uncertainties associated with the Company's business, please refer to the Company's filings with the Securities and Exchange Commission.
There is nothing on the FDA catalyst calendar about that. What catalysts are you talking about?
Tell me more about it
AVXL
On the verge of several catalysts!
LPCN - I don't think it's as risky as most people think. This isn't their first try. Panel may vote no (by a narrow margin) maybe. But FDA pulled the last CRL BS out of it's hat on the last CRL. They would have to make up more new crap to reject this time.
Stock needs a little momo up and it will take off. It's lack of trading making people slowly sell off.
Can't believe no articles have been written on this lately when a year ago it was $12 and the try before that it was over $20 The market is HUGE also.
Someone light a fire under LPCN I won't take much
MICT.
Micronet Enertec Technologies Signs Definitive Agreement to Sell its Defense and Aerospace Subsidiary, Enertec Systems
MONTVALE, N.J., Jan. 2, 2018 /PRNewswire/ -- Micronet Enertec Technologies, Inc. (NASDAQCM: MICT), (the "Company"), announced today that it has entered into a definitive agreement with Coolisys Technologies, Inc., a subsidiary of DPW Holdings, Inc. (NYSE American: DPW), to sell its wholly owned subsidiary, Enertec Systems 2001 Ltd. Pursuant to the terms of the agreement, Coolisys will pay the Company $5,250,000 in cash upon closing (of which 10% will be held in escrow for up to 14 months after the Closing to satisfy certain potential indemnification claims) and assume $4,000,000 in Enertec bank debt, paying effectively a total of $9,250,000 for all of Enertec's assets and liabilities. The transaction is scheduled to close the later of 60 days as of signing or 15 days after delivery of Enertec System's audited financial statements.
"We are extremely excited to enter into this agreement, which strongly supports and fully aligns with our roadmap to increase the value of our Company by focusing all our resources on the MRM space. As the second phase of the Electronic Logging Device (ELD) mandate is now in effect, we believe 2018 will produce an even greater market demand, which we believe may increase the revenues and profitability of our MRM business in the coming years," said David Lucatz, Chief Executive Officer of Micronet Enertec Technologies.
"This transaction fortifies our balance sheet by increasing our cash position and significantly reducing debt, while improving our income statement by eliminating all General and Administrative costs associated with Enertec. In addition, we believe our improved cash position will enable us to expand our MRM offerings," continued David Lucatz.
"The sale of Enertec to Coolisys empowers the Company to grow its MRM business more aggressively both organically and through acquisition. As our MRM revenues continue to increase each quarter, we aim to use this momentum and further invest all our focus and efforts to become a leader in the MRM space," Lucatz added.
About Micronet Enertec Technologies, Inc.
Micronet Enertec Technologies, Inc. (NASDAQCM: MICT) provides high tech solutions for severe environments and the battlefield, including missile defense technologies for Aerospace & Defense and rugged mobile devices for the growing commercial Mobile Resource Management (MRM) market. MICT designs, develops, manufactures and supplies customized military computer-based systems, simulators, automatic test equipment and electronic instruments, addressing a multi-billion-dollar defense industry. Solutions and systems are integrated into critical systems such as command and control, missile fire control, maintenance of military aircraft and missiles for the Israeli Air Force, Israeli Navy and by foreign defense entities. MICT's MRM division develops, manufactures and provides mobile computing platforms for the multibillion dollar mobile logistics management market in the U.S., Europe and Israel. American-manufactured systems are designed for outdoor and challenging work environments in trucking, distribution, logistics, public safety and construction.
Forward-looking Statements
This press release contains express or implied forward-looking statements within the Private Securities Litigation Reform Act of 1995 and other U.S. Federal securities laws. These forward-looking statements include, but are not limited to, those statements regarding the expected closing of the sale of Enertec to Coolisys, the Company's belief that 2018 will produce greater MRM market demand that may increase the revenues and profitability of the Company's MRM division in the coming years, the Company's intention to focus its resources on the MRM division and markets and increase its MRM offerings and, as a result of the sale of Enertec to Coolysis, grow its MRM business more aggressively both organically and through acquisition in an effort. to become a leader in the MRM space. Such forward-looking statements and their implications involve known and unknown risks, uncertainties and other factors that may cause actual results or performance to differ materially from those projected. The forward-looking statements contained in this press release are subject to other risks and uncertainties, including those discussed in the "Risk Factors" section and elsewhere in the Company's annual report on Form 10-K for the year ended December 31, 2016 and in subsequent filings with the Securities and Exchange Commission. Except as otherwise required by law, the Company is under no obligation to (and expressly disclaims any such obligation to) update or alter its forward-looking statements whether as a result of new information, future events or otherwise.
Cision View original content:http://www.prnewswire.com/news-releases/micronet-enertec-technologies-signs-definitive-agreement-to-sell-its-defense-and-aerospace-subsidiary-enertec-systems-300576376.html
SOURCE Micronet Enertec Technologies, Inc.
Copyright 2018 PR Newswire
BVXV. BiondVax Universal Flu Vaccine Patent Granted in India
NESS ZIONA, Israel, Jan. 2, 2018 /PRNewswire/ -- BiondVax Pharmaceuticals Ltd. (BVXV) (BVXV), developer of M-001, the Universal Flu Vaccine candidate currently in preparation for a Phase 3 clinical trial, announced today that one of its key patents titled "A Synthetic or Recombinant Influenza Multi-Epitope Polypeptide" has been granted in India. The patent belongs to BiondVax's portfolio family titled "Multimeric Multi-Epitope Influenza Vaccines" and has now been granted in over 30 countries.
With a population of over 1.3 billion people, and high population density in some of the major cities, India is an important potential market for BiondVax and a universal flu vaccine may significantly improve public health.
Dr. Tamar Ben-Yedidia, CSO of BiondVax, commented, "We are pleased and thankful that the Government of India's Patent Office accepted the uniqueness of our universal flu vaccine solution and approved this main patent of BiondVax. Authorities worldwide recognize that currently marketed influenza vaccines fall short. I am proud that M-001, which is designed to cover current, future, seasonal and pandemic flu strains, is preparing to enter Phase 3 trials later in 2018."
There is a general recommendation for seasonal influenza vaccination in India for healthcare workers, pregnant women, and people with some chronic illnesses.[1] However, Northern and Southern Hemisphere flu strains dominate at different times and in different regions of the Indian subcontinent. A universal flu vaccine, designed to cover all flu strains, would provide consistent coverage, and be appropriate for vaccination throughout the year.
The patent describes influenza vaccines comprised of multiple copies of several epitopes, such as M-001 which contains nine common and conserved influenza virus epitopes.
About BiondVax Pharmaceuticals Ltd
BiondVax is an advanced clinical stage biopharmaceutical company developing a universal flu vaccine. The vaccine candidate, called M-001, is designed to provide multi-season protection against current and future, seasonal and pandemic influenza virus strains. BiondVax's proprietary technology utilizes a unique combination of conserved and common influenza virus peptides, activating both arms of the immune system for a cross-protecting and long-lasting effect. In a total of 6 completed Phase 1/2 and Phase 2 human clinical trials, covering 698 participants, the vaccine has been shown to be safe, well-tolerated, and immunogenic. BiondVax is traded on NASDAQ: BVXV and TASE: BVXV. Please visit www.biondvax.com.
PRED $1.58 Mark This Post and see if my $30.00 Prediction for this stock comes true in 2018.
http://predictivetechnologygroup.com/
XGTI.
xG Technology's IMT Vislink Business Begins Shipping World's-First HCAM HEVC 4K Wireless Camera Systems in Fulfillment of Cli...
Print
Alert
XG Technology, Inc. (NASDAQ:XGTI)
Intraday Stock Chart
Today : Friday 29 December 2017
Click Here for more XG Technology, Inc. Charts.
SARASOTA, Fla., Dec. 29, 2017 /PRNewswire/ -- xG Technology, Inc. ("xG" or the "Company") (Nasdaq: XGTI, XGTIW), a leading provider of wireless video solutions to broadcast, law enforcement and defense markets, and private mobile broadband networks for critical communications, announced today that its IMT Vislink business has begun initial deliveries of HCAM, the world's-first 4K UHD wireless camera systems, to its customers.
IMT Vislink has today delivered its first HCAM systems of 2017 to Broadcast RF, and will enter 2018 with a growing run rate of orders for the systems. This reflects a continuing ramping of demand for the systems that began with their introduction in 2017 and many high-profile order commitments from worldwide clients. HCAM will be deployed at prestigious international sporting competitions in 2018, including the Winter Olympics and Commonwealth Games.
HCAM system deliveries would have been even higher, if not for the constrained supply of a critical subcomponent. This will negatively impact revenues attributable to HCAM systems in Q4 2017, and, consequently, IMT Vislink will enter 2018 with a significant backlog of orders that were unable to be manufactured and shipped this calendar year.
George Schmitt, Executive Chairman and CEO of xG Technology, said, "We are pleased to report that we have commenced delivery of our HCAM systems, and we fully expect the growing popularity of this all-new technology will play a significant role in our revenue plans in 2018 and beyond. HCAM systems are driving the worldwide migration from existing H.264 technology to HEVC and low-latency 4K UHD for wireless camera systems. At the same time, due to component supply issues, revenue from HCAM orders will be in excess of $3 million below those forecast for fiscal year 2017. We are working closely with our subcontractor partners to alleviate these supply challenges, and expect to accelerate HCAM system deliveries in Q1 2018. We will advise the market in due course on our progress in these efforts."
(LPCN)ADCOM on January 10 = 200% possible / Low Valuation of $74 million and only 21.2 million shares outstanding makes this stock attractive .
HIGH RISK Bio Stock but with significant upside potential if everything goes well . ADCOM meeting on January 10 for Lipocine´s Tlando the potential FIRST Oral product for the treatment of hypogonadism , if outcome is positive the stock will likely jump close or above $10 easily especially because of its low float more infos below and good luck
Lipocine (LPCN)
Market Cap: $74 Million
Cash: $25.7 Million
Price: $3.50
Shares Out: 21.2 Million
Latest Presentation
http://files.shareholder.com/downloads/AMDA-2BVSIV/5783990236x0x958103/2EB5BE41-D62A-4270-9634-2DAE6A0E1438/LPCN_11.08.17_long_FINAL__002_.pdf
The previously scheduled Bone, Reproductive and Urologic Drugs Advisory Committee (“BRUDAC”) meeting on January 10, 2018 to discuss the NDA for TLANDO remains unchanged.
The FDA has assigned a new Prescription Drug User Fee Act (“PDUFA”) goal date of May 8, 2018.
TLANDO™: Potential first oral TRT option
-Differentiated product targeting ~$2.0 billion established US TRT market
-Poised to meet and exceed unmet need
Ok, was in Aruba, and I think I saw Bob?
XGTI.
xG Technology's IMT Vislink Business Completes Delivery of First Order Against $12.5 Million U.S. Army Contract
Print
Alert
XG Technology, Inc. (NASDAQ:XGTI)
Intraday Stock Chart
Today : Wednesday 27 December 2017
SARASOTA, Fla., Dec. 27, 2017 /PRNewswire/ -- xG Technology, Inc. ("xG" or the "Company") (Nasdaq: XGTI, XGTIW), a leading provider of wireless video solutions to broadcast, law enforcement and defense markets, and private mobile broadband networks for critical communications, announced today that its IMT Vislink business has completed delivery of the first order against the previously announced $12.5 million U.S Army contract award. The contract covers the supply of hand-held intelligence, surveillance and reconnaissance receiver devices. Delivery of the first order was completed significantly in advance of the required delivery date of Feb. 15, 2018.
George Schmitt, Executive Chairman and CEO of xG Technology, said, "We are pleased to have successfully completed, shipped and delivered the first part of the Army order well in advance of the date by which we were obligated to deliver. We all appreciate the efforts of our team in both the United Kingdom and the U.S. to get the job done. We look forward to continuing to deliver these state-of-the-art devices to our people in uniform to help them in their job of protecting all of us."
AXSM= 2 BIG P3 READOUT THIS MONTH=$$$$$
AXSM (MC $120 M) (Cash $40 M) Shares Out 25.4 M // 7x Phase 3 programs in various indications targeting billion dollar markets ..2x Phase 3 readout expected within THIS MONTH =HUGE Upside Potential on this undiscovered low float gem .GL
Axsome Therapeutics (AXSM)
Market Cap: $120 M
Cash $40 M
Price: $4.75
Shares Out : 25.4 M ( 16+ Million shares held alone by Management & Institutions)
Presentation from mid November (see slide 36 for upcoming milestones)
http://media.corporate-ir.net/media_files/IROL/25/254022/Axsome_Presentation_13%20Nov%202017.pdf
Anticipated Near-Term Clinical Milestones
Clinical Trial Readouts:
-- Phase 3 COAST-1 trial of AXS-02 in knee OA associated with BMLs, interim analysis (4Q 2017)
-- Phase 3 CREATE-1 trial of AXS-02 in CRPS, interim efficacy analysis (4Q 2017)

Where in the world is Bob? He must be on vacation?
|
Followers
|
399
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
23979
|
|
Created
|
03/06/13
|
Type
|
Free
|
| Moderator Bob Stocks | |||
| Assistants trade2much jbem777 | |||
|
Posts Today
|
0
|
|
Posts (Total)
|
23979
|
|
Posters
|
|
|
Moderator
|
|
|
Assistants
|
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
