Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Note from Bizarro world. White Sox fan says up to 22 we go. No reason given.
Share price today was fantastic, as opposed to what American politics has descended into
Lithium is not the future. Solid state batteries are the future. Future generations deserve better from us. We should all want to pass on a healthy planet. So instead of looking for reasons to say we can’t do this. We should all be looking for solutions. I am a true Reagan conservative who happens to live in Colorado breathing the smoke from all the recent fires (along with my grand daughter and grand son) knowing that Climate change is a real issue. I am not supportive of this current Republican Party. I don’t watch cable news. They are not the party that I once knew. I also despise the beliefs of the far left. Look for solutions together in the center. Dissension is much healthier there.
Can we leave off the political commentary?
Since I’ve been affected along with a majority of posters on this board in our investment here and across all markets... interesting commentary by someone who has more dough than all of us combined...mr bezos on Saturday...” Ouch. Inflation is far too important a problem for the White House to keep making statements like this. It’s either straight ahead misdirection or a deep misunderstanding of basic market dynamics. ....imao...r
HappySafe 4thOfJuly 3DayWeekend Celebrations To All StockHOLDERS! Eom.
Well i hope the eventual paperwork submitted to the FDA isn't prepared by Mr. Spock....r
TO REITERATE & ACCENTUATE THE POSITIVES: Avid Bioservices, Inc
AD/PD Laboratories Open Exactly Eight Months After Announced Plans to Build Viral Vector Facility
Build-Out of Facility’s CGMP Manufacturing Suites Continues, Expected to Come Online in Mid-Calendar 2023
TUSTIN, Calif., June 14, 2022 (GLOBE NEWSWIRE) ... Avid Bioservices, Inc. (NASDAQ:CDMO), a dedicated biologics contract development and manufacturing organization (CDMO) working to improve patient lives by providing high quality development and manufacturing services to biotechnology and pharmaceutical companies, today announced the opening of the analytical and process development (AD/PD) suites within the company’s new, world-class viral vector development and CGMP manufacturing facility. The launch of the AD/PD labs comes eight months to the day that Avid announced its intention to expand its CDMO service offering into the rapidly growing cell and gene therapy market. Build-out of the viral vector facility’s CGMP manufacturing suites is ongoing, with those capabilities expected to come online in mid-calendar 2023.
The company’s new AD/PD labs are equipped with complete upstream, downstream and analytical development capabilities and are able to support up to 500 liter suspension culture in single-use bioreactors and various adherent cell modes. The company’s viral vector AD/PD team, which is led by Elie G. Hanania, Ph.D., has deep expertise in developing and implementing state-of-the-art enabling technologies for viral vector production and purification processes focused on adeno-associated viruses (AAVs), lentiviruses, oncolytic viruses and other viruses for gene therapy and vaccine applications.
“Avid’s entry into the cell and gene therapy sector is a critical component of the company’s growth strategy over both the short- and long-term. We believe that we are uniquely positioned to leverage our deep expertise in the manufacturing of traditional biologics to address the growing demand for high quality manufacturing of cell and gene therapies,” said Nick Green, president and chief executive officer of Avid Bioservices. “Today’s launch of our AD/PD labs is a crucial first step in our expansion into the viral vector space as we remain laser focused on smart and sustainable revenue growth for our business. We now look forward to engaging in a meaningful way with potential viral vector clients and offering this latest tangible evidence of the quality of Avid’s facilities and the capabilities of our team.”
In October 2022, Avid announced plans to construct a purpose-built 53,000 square foot viral vector facility in Costa Mesa, CA, approximately five miles from Avid’s existing operations in Tustin, CA. The strategic decision was driven by continued strong growth in the cell and gene therapy market combined with the CDMO industry’s overall lack of proven, high-quality CGMP manufacturing expertise and capacity for viral vectors. With more than 17 years of experience in commercial manufacturing of biologics underpinned by a strong quality ethos and a customer-centric approach to doing business, Avid offers a strong value proposition to prospective customers in the cell and gene therapy market.
“The opening of our newly constructed viral vector process and analytical development laboratories within eight months of our announced investment is truly a testament to the capability and dedication of our Avid team, as well as our partners at CRB. We are excited to begin working on customer projects in this new facility and look forward to completing the ongoing construction of our new vial vector CGMP manufacturing suites,” said Drew Brennan, general manager of viral vector technologies of Avid Bioservices.
r622102675, it has to be anti-body. probably not a typo but a spell check miss-click.
Speculative, I know, but anti-human wouldn't fit at all to qualify Lenz, would it?
AIMO
From the HGEN release yesterday...God i hope its only a typo... "Within ACTIV-5, lenzilumab is the first and only anti-human GM-CSF treatment to be tested in ACTIV as a concomitant therapy with remdesivir compared with placebo plus remdesivir."...r
It is true that for 2 super quarters (best of all times) we can't really profit from the PPS up movement because the same day the markets are under pressure in general.
Of course that is true for all stock, not only CDMO. What is important is:
- Super revenue
- big backlog
- quicker availability of expansion projects (and related revenue)
- hiring extra people because the work is there
- 350-400 Mil target in view
- The tax benefits (I have been saying that since about our quarters started to be profitable and now we have full FY that are profitable).
- Increasing margin.
Where better can your money be? And certainly if you are in a non-US Dollar country because the USD is getting stronger by the day. It touch 1.03 vs the Euro.
AIMO
I was misunderstood. The quote was taken out of context. I said if this was a bull market and the pps was much higher but the company's future net income prospects looked bleak who would pay a premium over the current pps. Nobody would.
Likewise, the company would not accept a premium over a very depressed price when the company's future income prospects look great. The future income prospects should determine the buyout price much more so than the current pps.
r622, thanks. Excellent summary of diminishing tax loss carry-forward.
Single digits coming! NOTHING was blowout about this earnings report. All in line with analysts estimates and a slight miss on projected revenues.
Already sub $14 and headed LOWER!
The drive to move production of our products on shore makes our prospects even stronger.
We understand the situation here in this macro-environment but you surely must
not be referring to the company, CDMO, when you say
but the company's future net income prospects looked bleak because of increasingly more fierce competion and waning future demand and other problems who would pay a premium for such a company?
Single digits coming! NOTHING was blowout about this earnings report. All in line with analysts estimates and a slight miss on projected revenues.
Already sub $14 and headed LOWER!
The buyout price would be based on projected future net income for years to come. If this was a bull market and the pps was much higher but the company's future net income prospects looked bleak because of increasingly more fierce competion and waning future demand and other problems who would pay a premium for such a company?
I’d have to think north of $30.
The morning line on futures portends a gruesome opening...we may not be able
to catch a break and buck this trend, but perhaps our "diamond in the rough"
(CDMO) can be purchased on the cheap for squirreling away....!!
We are hitting our projections and the pace of the expansion is ahead of
schedule..................................not a bad HOLD in this market...GLTA..
What would $400 mil in revenues support for the pps? Nick said in a year we will have almost $400 mil capacity.
Wow. RBC maintains Outperform but lowers price target to $22 from $32. Hitting on all cylinders is not even enough for this bear market.
Thanks for pointing out the deep stuff, r! We knew there were various loss carryforwards and this lays them out nicely.
Let's hope our NOL carryforwards do the same thing for Avid's market perception and stock price as Tesla's sale of emission credits did for Elon. Musk became the world's richest man by getting a few "profitable" quarters under his belt - all due to the emission credit sales, not the sale of cars.
All in the messaging. If we can somehow sell the story that toll processing drugs reduces CO2 emissions, we'd be golden!
All TIC or course. I think.
Regards,
WH
Taxes (now that we are making a decent profit)... per the 10k... "At April 30, 2022, we had federal NOL carry forwards of approximately $383.7 million. The federal NOL carry forwards generated prior to January 1, 2018 expire in fiscal years 2024 through 2038, unless previously utilized. The federal NOL generated after January 1, 2018 of $19.6 million can be carried forward indefinitely. Utilization of NOLs generated subsequent to 2020 are limited to 80% of future taxable income. We also have California state NOL carry forwards of approximately $312.2 million at April 30, 2022, which begin to expire in fiscal year 2023. We also have other state NOL carry forwards of approximately $0.3 million at April 30, 2022, which begin to expire in fiscal year 2037.
Additionally, the future utilization of our NOL carry forwards to offset future taxable income may be subject to an annual limitation, pursuant to Internal Revenue Code Section 382, as a result of ownership changes. A Section 382 analysis has been completed through April 30, 2022, and it was determined that no significant change in ownership had occurred. However, ownership changes occurring subsequent to April 30, 2022 may impact the utilization of NOL carry forwards and other tax attributes in future periods.
At April 30, 2022, we had $5.8 million and $1.5 million of federal and California research and development credit carry forwards. The California research credits do not expire and the federal credits begin to expire in fiscal year 2026." ....r
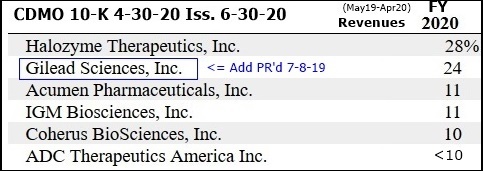
They are definitely spreading the customer base out...HALO was 41% of revs and IGM Bio was 11%...balance was all less than 10%...r
And the update statement regarding HGEN...During the fiscal year ended April 30, 2022, changes in estimates for variable consideration resulted in a decrease in revenues of $14.7 million. These changes in estimates for variable consideration can primarily be attributed to a dispute with a customer over the payment of certain cancellation fees due to us under the terms of the related customer contract which resulted in a decrease in revenues of $11.7 million for the fiscal year ended April 30, 2022. We believe we have a contractual right to the disputed amount, but as this contractual right is being disputed and therefore may be uncollectible, we have not recorded revenue associated with the disputed amount. ...r
Btw...re:HGEN...from the 10k... We currently are not a party to any legal proceedings, the adverse outcome of which, in management’s opinion, individually or in the aggregate, would have a material adverse effect on our consolidated financial condition or results of operations. ...r
He's a real leader....far cry from the previous regime.
Fell real good about him at the helm
S-8 filed for another 3.4 mil shares for incentive plan. 100 new employees added this last year, more to come.
This Registration Statement is being filed for the purpose of registering an additional 3,400,000 shares of Common Stock that were reserved for issuance under the 2018 Omnibus Incentive Plan. The Registrant previously filed with the Securities and Exchange Commission (the “SEC”) a registration statement on Form S-8 on December 10, 2018 (SEC File No. 333-228735) (the “Prior Registration Statement”), registering shares of Common Stock issuable under the 2018 Omnibus Incentive Plan. This Registration Statement relates to securities of the same class as those to which the Prior Registration Statement relates and is submitted in accordance with General Instruction E of Form S-8 regarding Registration of Additional Securities. Pursuant to General Instruction E of Form S-8, the contents of the Prior Registration Statement are incorporated herein by reference and made part of this Registration Statement, except for Items 3 and 8, which are being updated by this Registration Statement.
Obviously, Nick is very skilled and very good in his role. I really love how he dishes out the compliments to his team. A head coach lots of folks (superstars included) would like to play for.
Excellent cc. Hard to top that one.
Regards,
WH
30% gross margins expected with little inflationary influences.
Increasing capacity driving revenues. All positive.
Talk about blowout earnings!
Look out ahead. All systems GO!
Oh boy, oh boy, oh boy is this great. Copyright Animal House.
Will $CDMO BreakOut In AfterHours? Glta StockHOLDERS! eom.
Daaaaamn. Go widdle cdmo go
Uhhhh….bad call on your part. 1.65 earnings per diluted share. 15.92 last.
Avid Bioservices Reports Financial Results for Fourth Quarter and Full Fiscal Year Ended April 30, 2022 and Recent Developments
June 29, 2022 at 4:05 PM EDT
Download PDF
-- Recorded Fourth Quarter and Full Fiscal Year Revenue of $31 Million and $120 Million, Respectively --
-- Signed $44 Million in Net New Business Orders and Ended the Quarter with a Record High Backlog of $153 Million --
-- New Cell and Gene Therapy Analytical and Process Development Suites Now Operational and CGMP Manufacturing Suites Continue On Schedule; Myford South Facility Construction On Schedule --
-- Project Fiscal 2023 Revenue of $140 to $145 Million, Representing 17% - 21% Growth Over Fiscal 2022 --
TUSTIN, Calif., June 29, 2022 (GLOBE NEWSWIRE) -- Avid Bioservices, Inc. (NASDAQ:CDMO), a dedicated biologics contract development and manufacturing organization (CDMO) working to improve patient lives by providing high quality development and manufacturing services to biotechnology and pharmaceutical companies, today announced financial results for the fourth quarter and full fiscal year, ended April 30, 2022.
Highlights from the Quarter and Fiscal Year Ended April 30, 2022, and Other Events:
“We made considerable progress during fiscal 2022. During the year, we achieved revenue of $120 million, representing a doubling of revenues recorded in fiscal 2020. Notably, Q4 fiscal 2022 was the eighth consecutive quarter of operational profitability for the company. The company signed net new project orders for $155 million in fiscal 2022, leading to a backlog of $153 million; Avid’s largest backlog to-date. Supporting this growth, as well as that which we anticipate in the coming years, our facilities and service expansions continue to proceed according to plan,” stated Nick Green, president and chief executive officer of Avid Bioservices.
“Our momentum in fiscal 2022 was driven in part by the exceptional performance of our enhanced commercial team. Over the last six months, we expanded the sales teams for both our mammalian and our cell and gene therapy businesses. We also expanded our business operations team to best support our growing project pipeline. This new organization has had great success, highlighted by the fact that it signed the same number of new projects in the second half of fiscal 2022 as signed in all of fiscal 2021.
“During the period, our facilities and service expansions continued to advance on a timeline that will allow us to meet the demand of existing customers that are expanding their manufacturing work with us as well as our newest and prospective customers. With respect to our 53,000 square foot cell and gene therapy facility, we recently completed the first phase of a two-phase construction plan, opening our new analytical and process development laboratories eight months to the day after we announced our intention to expand into the cell and gene therapy business. Construction of the CGMP suites for our cell and gene therapy facility, the second phase of this expansion, remains on track and those manufacturing suites are expected to come online in mid-calendar 2023. The expansion of our Myford facility, which houses our mammalian operations, is also being constructed in a two-phase process. As reported last quarter, the company completed the first phase with the opening of a new downstream suite. The second phase of this project is focused on the Myford South facility and is on track to come online at the beginning of calendar year 2023, which at our current growth rate will be ideally timed to provide much needed capacity for fiscal 2024.
“Finally, we are delighted to announce a further expansion of our process development capacity for our mammalian cell business. Once complete at the end of this calendar year, these new suites will double our current process development capacity which came online in October of 2019. This new line will significantly increase capacity at the front-end of our mammalian cell business, which is critical to the efficient on-boarding of new clients. It is expected that this expansion will cost approximately $6 million, and upon completion, will have the potential to generate approximately $20 million in additional revenue capacity.
“Given the growth momentum achieved during fiscal 2022, our significant year-end backlog, and the increase in demand anticipated during fiscal 2023, we are pleased to announce revenue guidance for fiscal 2023 of between $140 and $145 million.”
Financial Highlights and Guidance
The company is providing revenue guidance for fiscal 2023 of $140 million to $145 million, a 17% - 21% increase over fiscal 2022.
Revenues for the fourth quarter of fiscal 2022 were $31.2 million, representing a 13% increase compared to $27.6 million recorded in the prior year period. The increase in revenues for the quarter can primarily be attributed to an increase in the scope of in-process and completed manufacturing runs and an increase in process development revenues primarily associated with services provided to new customers as compared to the prior year period. For the 2022 full fiscal year, revenues were $119.6 million, a 25% increase compared to $95.9 million in the prior year period. The increase in revenues for the 2022 full fiscal year, as compared to the prior year period can primarily be attributed to an increase in the number and scope of in-process and completed manufacturing runs, unutilized reserved capacity fees, and process development revenues.
As of April 30, 2022, revenue backlog was $153 million, representing a net increase of 30% compared to $118 million at the end of fiscal 2021. The company expects to recognize the majority of this backlog during fiscal 2023.
Gross margin for the fourth quarter of fiscal 2022 was 22%, compared to a gross margin of 29% for the fourth quarter of fiscal 2021. Factors impacting the gross margin for the quarter were primarily from increases in costs associated with the growth of our business and our facility expansions including compensation and benefit expenses as well as increases in facility and related expenses, partially offset by higher revenues during the period. Gross margin for the 2022 full fiscal year was 31%, consistent with 31% for the prior year period.
Selling, general and administrative expenses (“SG&A”) for the fourth quarter of fiscal 2022 were $5.9 million, an increase of 17% as compared to $5.1 million recorded for the fourth quarter of fiscal 2021. The increase in SG&A for the fourth quarter was primarily due to higher compensation and benefit expenses as well as increased facility and related expenses. For the 2022 full fiscal year, SG&A expenses were $21.2 million as compared to $17.1 million for the prior year. The increase in SG&A during the 2022 full fiscal year was primarily due to compensation and benefit expenses, facility and related expenses, advertising costs and legal and accounting fees.
During the fourth quarter of fiscal 2022 we recorded a non-cash income tax benefit of $115 million, or $1.63 per diluted share, due to release of our valuation allowance recorded against the company's deferred tax assets (DTAs). The company previously maintained a valuation allowance on its DTAs until there is sufficient evidence to support the reversal of all or some portion of these allowances. On a periodic basis, the company reassesses the valuation allowance of its DTAs, weighing all positive and negative evidence, to assess if it is more-likely-than-not that some or all of the company's DTAs will be realized. As of the fourth quarter of fiscal 2022, the company has demonstrated profitability and cumulative pretax income as well as forecasting revenue growth. After assessing both the positive and negative evidence, the company determined that it was more-likely-than-not that its DTAs would be realized and released the valuation allowance related to federal and state DTAs as of April 30, 2022.
For the fourth quarter of fiscal 2022, the company recorded net income attributable to common stockholders of $115.6 million or $1.87 per basic and $1.65 per diluted share, as compared to a net loss attributable to common stockholders of $2.7 million or $0.04 per basic and diluted share, for the fourth quarter of fiscal 2021. For the 2022 full fiscal year, the company recorded net income attributable to common stockholders of $127.7 million or $2.08 per basic and $1.84 per diluted share, compared to net income attributable to common stockholders of $3.3 million or $0.06 per basic and diluted share, for the 2021 full fiscal year. Excluding the non-cash income tax benefit of $115.0 million recorded during the fourth quarter of fiscal 2022, the company’s net income attributable to common stockholders was approximately $600 thousand or $0.01 per basic and diluted share for the quarter, and $12.7 million or $0.21 per basic and diluted share for the full fiscal year 2022.
The company reported $126.2 million in cash and cash equivalents as of April 30, 2022 compared to $169.9 million as of the prior fiscal year ended April 30, 2021.
More detailed financial information and analysis may be found in Avid Bioservices’ Annual Report on Form 10-K, which will be filed with the Securities and Exchange Commission today.
Recent Corporate Developments
The company’s commercial team signed multiple new project orders during the fourth quarter, totaling approximately net $44 million. For the 2022 full fiscal year, the company signed new project orders totaling approximately net $155 million. These projects span all areas of the business, from process development to commercial manufacturing.
The company continues to make progress with both the Myford South expansion, as well as the construction of its new dedicated cell and gene therapy facility. The company currently expects to complete the second phase of its Myford South expansion, which includes both upstream and downstream CGMP manufacturing suites, during the first quarter of calendar 2023. With respect to the cell and gene therapy business, the company brought its process and analytical development capacity online in mid-June 2022. The company remains on track to bring the CGMP manufacturing suites online in mid-calendar 2023. Please visit the Avid website Facilities page for more information about the company’s expansions and videos documenting progress (https://avidbio.com/expansion-updates/).
Okay so I'm not familiar with these things on other stocks...but looking at your link Chey it seems we have odd/small trades over and over. WTH is that all about? That can't be normal is it?
Buy volume 241K sell vol 59K.
https://ih.advfn.com/stock-market/NASDAQ/avid-bioservices-CDMO/trades
Shaping up to be another epic TCLN/PPHM/CDMO earnings report! I'm betting single digit share price by the end of next week!
Viral vector build suppose to take 18 months but done in 8 months?
I didn't have to
Nick Green says he loves quickly building businesses.
An indicator of the breakneck speed of the chief executive of Avid Bioservices Inc. (Nasdaq: CDMO) came earlier this month when it announced completion of Phase 1 of its $75 million “world-class” manufacturing facility in Costa Mesa, only eight months after announcing its plans.
The 53,000-square-foot viral vector facility was designed and built by Kansas City, Mo.-based CRB, a construction firm that specializes in the life sciences industry. The second phase of the facility’s development is expected to become operational in mid-calendar year 2023.
“Considering the challenges of the current supply chain environment, it has been nothing short of remarkable for Avid and CRB to be opening the doors of Avid’s new viral vector process development labs here in June,” said Mike Alston Jr., director of project engineering for Avid Bioservices.
The site, at 3030 Airway Ave., is near John Wayne Airport, and about a 15-minute drive from Avid’s multi-building headquarters base in Tustin.
The Costa Mesa expansion came after Avid announced plans to invest nearly another $70 million to upgrade facilities at its Tustin base in early 2021.
Booming Sales
In the past 30 years, Green has headed 31 global pharmaceutical facilities in nine countries on four continents.
At every company he’s worked at, he’s grown compounded annual growth rate by at least 20%.
“I love growing businesses,” he told the Business Journal last October. “It’s what I’ve done most of my life.”
Green’s been aided by paradigm shift in the industry. While drug manufacturing may be 5% to 10% cheaper in places like China, the pandemic has caused drug company executives to realize the fragility of their supply chains. They now want to keep more of their manufacturing within North America.
In 2021, Green’s first full year as CEO, Avid almost doubled sales in 2021 to $95.9 million. Analysts are expecting Avid sales to climb 22% this year to $117.3 million and another 25% in 2023 to $146.5 million.
Green said Avid has the potential to generate $350 million in annual sales.
Shares have more than doubled since Green became CEO. The Tustin-based company now sports a $940 million market cap.
New Industry
Avid is known as a contract manufacturer of biologics for pharmaceutical companies in more than 90 countries.
The Costa Mesa facility was built so Avid can make drugs for cell and gene therapy, a market that’s expected to grow more than 18% annually through 2028.
“Avid’s entry into the cell and gene therapy sector is a critical component of the company’s growth strategy over both the short- and long-term,” Green said in a statement. “We believe that we are uniquely positioned to leverage our deep expertise in the manufacturing of traditional biologics to address the growing demand for high quality manufacturing of cell and gene therapies.
“We now look forward to engaging in a meaningful way with potential viral vector clients and offering this latest tangible evidence of the quality of Avid’s facilities and the capabilities of our team.”
The company is continuing to expand its development capacity for its mammalian cell business by spending $6 million on larger state-of-the-art laboratories that could support an additional $20 million in annual process development revenue, doubling the company’s current process development capacity.
Mammalian cell culture is the process of growing cells obtained from mammals in vitro, outside their original tissue.
Completion is anticipated by the end of calendar 2022.
“With several expansion projects underway across various facilities and business segments, we feel that we are well positioned to take advantage of the increasing demand for high-quality CDMO services and continue to rapidly grow our revenue,” Green said.
gotta log into that paper..... Can you cut-n-paste the article for us.
Me thinks it's going to be a good call
https://www.ocbj.com/drugs-2/avid-bioservices-opens-75m-facility-in-8-months/
Not all of it, but just enough. And why not, historynis on my side!
|
Followers
|
832
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
345997
|
|
Created
|
11/07/03
|
Type
|
Free
|
| Moderators Preciouslife1 4OurRetirement | |||

Avid Bioservices, Inc. (Nasdaq “CDMO”), Tustin CA. http://www.avidbio.com
President/CEO: Nicholas Green (eff. 7-30-20 https://tinyurl.com/yczapcc7 )
Four Avid Fact Sheets a/o 2-2020: https://avidbio.com/resources/fact-sheets/
Avid Bioservices is a CDMO committed to improving the lives of patients by manufacturing products derived from mammalian cell culture for the biotechnology & biopharmaceutical industries. Services include cGMP clinical & commercial product manufacturing, purification, bulk packaging, stability testing & regulatory strategy, submission, and support. The company also provides a variety of process development activities, including cell line development & optimization, cell culture & feed optimization, analytical methods development, and product characterization.
12-2019/Video: Tour of Myford Facility (2mins.): https://vimeo.com/380135562
7-2020: AVID’s Push Towards cv19 (I don’t capitalize things I hate) contracts: https://tinyurl.com/y8wzgneh
UPCOMING EVENTS: https://avidbio.com/events/
Sep1(Tue): Q1/FY21 (qe 7-31-20) Financials & Conf. Call - Transcript: https://tinyurl.com/y329llow
Sep17/Fireside Chat w/Nick Green: 18th Annual Morgan Stanley Global Healthcare Conf.: https://tinyurl.com/y4cbjjrc
Sep21-24/Virtual: Biotech Week Conf., Boston
Sep21-24/Virtual: BioProcess Intl. US Wes Conf., Santa Clara CA
...Time TBD: Presentation by Haiou Yang, PhD "Facility-Fit Driven Dev. for a mAb Production Process"
Oct20 10amPT/VIRTUAL: Annual Shareholders Mtg. https://tinyurl.com/y28blkjn 14A Proxy: https://tinyurl.com/y46ga9el
...ASM Replay (9:45): https://www.virtualshareholdermeeting.com/CDMO2020 (Control# not needed)
Apr20-22 2021/Booth#1159: INTERPHEX, NYC
Dec2(Wed) after mkt: Q2/FY21 (qe 10-31-20) Financials & Conf. Call - Transcript: https://tinyurl.com/y66xk888
Dec14-17: Antibody Engineering & Therapeutics Conf., San Diego
Quotes: Yahoo: Yahoo.com/CDMO Nasdaq: http://www.nasdaq.com/symbol/cdmo RT: http://www.nasdaq.com/symbol/cdmo/real-time
• IR: Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com, Tim Brons (Media) 415-675-7402 tbrons@vidasp.com
SEC:
Latest 10Q 10-31-20 iss. 12-2-20 https://tinyurl.com/y3ournzr (Cash 10-31-20=$35.7mm)
Latest 10K 4-30-20 iss. 6-30-20 https://tinyurl.com/yak25nco (Cash 4-30-20=$36.3mm)
Latest DEF14A/Proxy iss. 8-27-20 (re: 10-20-20 ASM): https://tinyurl.com/y46ga9el
ALL SEC filings for CDMO: https://tinyurl.com/yc4zjyzv
CDMO's Corp. Bylaws (a/o 11-14-14): http://tinyurl.com/y8hsppea
Poison Pill adopted 3-16-06 for 10yrs: http://tinyurl.com/yvypvh 44-pg SEC filing: http://tinyurl.com/5m57ut BUNGLER explains in plain language: http://tinyurl.com/mft4nd6
...3-17-16 Poison Pill extended until 3-16-2021 https://tinyurl.com/y74glo2n (Form8A Amendment #1)
...Poison Pill TERMINATED eff. 9-23-2019 https://tinyurl.com/y2mzx3xo
SHARES:
Shares O/S as of 11-23-20=56,726,334 - history since 4-2006: https://tinyurl.com/y66xk888 (at bottom)
...A/O 10-31-20: 3,451,000 stock options outstanding at a wgt.avg. exercise price of $6.41 (pg.16 10Q).
...MarketWatch.com for CDMO (shows Float): https://www.marketwatch.com/investing/stock/cdmo
1-12-18: S-3 Shelf Registration filed for up to $125mm https://tinyurl.com/y9qtewpw
...12-14-20: Avid raises ~$34M gross, selling 3,833,335@9.00/sh. (underwriter: RBC Capital) https://tinyurl.com/y92yr9g4
...2-20-18: Avid Raises ~$21.8M net, selling 10,294,445@$2.25 (underwriter: Wells Fargo) 8-K: https://tinyurl.com/ya3nenth 424B5: https://tinyurl.com/ycpshgxl
Total INST.+LARGE Holdings now 41,617,965 73.5% a/o 9-30-20 https://tinyurl.com/y652nxpr
13 LARGEST SHAREHOLDERS a/o 9-30-20:
1. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A/Proxy: https://tinyurl.com/y9c972fa – orig. 13G filed 11-2015)
2. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
3. Blackrock Inc. (Larry Fink): 3,834,816 6.8% (-111,172 a/o 9-30-20 Nasdaq Inst.)
4. IsZo Capital Mgt. (Brian Sheehy): 3,459,888 6.1% (-88,144 a/o 9-30-20 Nasdaq Inst.)
5. Millennium Mgt. 2,678,984 4.7% (+633,918 a/o 9-30-20 Nasdaq Inst.)
6. Vanguard Group 2,659,738 4.7% (-19,553 q/e 9-30-20 Nasdaq Inst.)
7. Snyder Capital 2,302,621 4.1% (-2,258 a/o 9-30-20 Nasdaq Inst.)
8. Altravue Capital 1,973,349 3.5% (+39,534 a/o 9-30-20 Nasdaq Inst.)
9. Wellington Mgt. 1,503,162 2.7% (+236,947 a/o 9-30-20 Nasdaq Inst.)
10. Sargent Invest. Grp. 1,052,099 1.9% (-11,000 a/o 9-30-20 Nasdaq Inst.)
11. State Street 992,280 1.8% (-65,922 a/o 9-30-20 Nasdaq Inst.)
12. Portolan Capital 963,374 1.7% (+528,662 a/o 9-30-20 Nasdaq Inst.)
13. Silvercrest Asset Mgt. 878,805 1.6% (-16,746 a/o 9-30-20 Nasdaq Inst.)
Inst. Holdings (Nasdaq) - updated 45-days after each qtr-end: http://www.nasdaq.com/symbol/cdmo/institutional-holdings
INSIDER-Trans: https://tinyurl.com/ycpw4j9z (formerly PPHM thru 2017: http://tinyurl.com/ypkow8 )
Short Interest, updated twice a month: https://www.nasdaq.com/symbol/cdmo/short-interest
A-T-M (At-The-Market) Sales 3/2009 - 7/14/17 ($272,500,000gr./34,869,747sh=$7.81/sh): http://tinyurl.com/yagmu2on
PPHM shares were 1:7 Reverse Split eff. 7-10-17 (315mm/$.606=>45mm/$4.24) http://tinyurl.com/yymtzcm9
PPHM shares were 1:5 Reverse Split eff. 10-19-09 (~237mm/$.64=>~47.4mm/$3.20) http://tinyurl.com/ykuw588
Potential Value: 9-19-2017: Catalent acquires CDMO Cook Pharmica for $950M ($179Msales x 5.3; EV/EBITDA=17.3) http://tinyurl.com/yd46m8py
AVID TEAM: https://avidbio.com/leadership/ - Open Jobs: https://avidbio.applicantpro.com/jobs
7-30-20: Nicholas Green (ex-Therapure Biopharma) becomes President/CEO of Avid - 6-25-20 PR: https://tinyurl.com/yczapcc7
4-13-20: Avid Hires 2 Senior Directors of Bus. Dev: Jason C. Brady & Sylvia Hinds https://tinyurl.com/sq2679g
1-8-20: Avid Appoints Timothy Compton as Chief Commercial Officer (repl. Tracy Kinjerski) https://tinyurl.com/ydww58sn
7-26-19: Catherine Mackey, PhD (ex Pfizer VP) joins Avid's BOD, replacing Joel McComb https://tinyurl.com/y3xhqmvm
5-8-19: Richard (Rick) B. Hancock named Interim President/CEO; Roger Lias resigns https://tinyurl.com/y4pqdwyc
7-16-18: Daniel Hart (ex-ENO Holdings) joins Avid as CFO https://tinyurl.com/ychz45p2
7-16-18: Michael Faughnan (ex-Lonza/WuXi Biologics) joins Avid as Sr.Dir./BusDev/WestC https://tinyurl.com/yaozdggz (Left 5-2019)
5-29-18: Sandra C. Carbonneau (ex-Lonza) joins Avid as Dir./BusDev./EastCoast https://tinyurl.com/ybvrwn2l (left 12-2019)
5-8-18: Magnus Schroeder (ex-AGC Biologics) joins Avid as VP/Process Sciences https://tinyurl.com/y7tpswaw
2-23-18: CFO Paul Lytle voluntarily resigns eff. 5-24-18 (2-23-18 +90days), "CFO search underway" https://tinyurl.com/ybnjzbv6
9-11-17: Roger J. Lias (ex-Allergan) to become Avid’s CEO and join PPHM’s BOD eff. 9-25-17 https://tinyurl.com/yddufw4s (left 5-8-19)
11-29-17: Tracy L. Kinjerski joins Avid as VP/Bus.Operations https://tinyurl.com/yc4zenkc (Left 1-8-2020)
Profiles of all 7 BOD members: http://ir.avidbio.com/corporate-governance/board-of-directors
...Joseph Carleone/Chair, Nicholas Green(CEO), Richard Hancock, Gregory Sargen, Mark Bamforth, Patrick Walsh, Catherine Mackey

10-24-17: Peregrine Adds Patrick D. Walsh to BOD, "An Industry Veteran with 30+yrs Experience Leading Successful CDMO Org’s” http://tinyurl.com/y3jo2rv7
10-19-17: Peregrine Adds Mark R. Bamforth to BOD, "10yrs/Genzyme; 30yrs of biologics leadership experience, incl. founding 2 CDMOs" http://tinyurl.com/y6ydcaof
ANALYST COVERAGE: http://ir.avidbio.com/analyst-coverage
Also see: https://www.marketbeat.com/stocks/NASDAQ/CDMO/price-target
Janney Montgomery Scott - Paul Knight PT=$10
H.C. Wainwright & Co. - Joseph Pantginis PT=$9
Craig-Hallum Capital Group - Matt G. Hewitt PT=$10
First Analysis Securities - Joseph Munda PT=$7.50
Stephens Inc. - Jacob Johnson PT=11 (10=>11 9-2-20)
FINANCIALS & BUSINESS DEV.:
2-3-21: Avid to Mfg. CV19 Therapeutic Lenzilumab for Humanigen https://tinyurl.com/1hzzksxa
8-26-20: Mapp Biopharmaceutical signs w/Avid for ClinDev of Antiviral Antibody (MBP091) https://tinyurl.com/yxwvr949 BARDA #HHSO100201900018C($16.5M+Opt/$30M), see: https://tinyurl.com/yye8t5nx
8-20-20: Oragenics signs Dev/Mfg Agreement with Avid for COVID-19 Vaccine “TerraCoV2” https://tinyurl.com/yxqg3w4v
8-6-20: Iovance Biotherapeutics signs w/Avid(+Aragen) to Dev+Mfg. IOV-3001 (IL-2 Analog) https://tinyurl.com/y2lgzh6x
7-28-20: Avid Teams with Argonaut to add “Parenteral Fill-Finish Services” https://tinyurl.com/y2lgzh6x
5-6-20: Avid Teams with Aragen to speed up drug dev./delivery timelines https://tinyurl.com/ycuu2m5r
4-23-20: Avid receives $4.4M PPP/CV19 Loan (can apply for forgiveness) 8K: https://tinyurl.com/yaltupxa
...5-12-20: Avid Pays Back $4.4M PPP/CV19 Loan (“New SBA Guidance/abundance of caution”) 8K: https://tinyurl.com/y993tkk6
3-10-20: CDMO's Revs & Burns By Qtr Table, FY07/Q1 thru FY20/Q3 (q/e 1-31-20): https://tinyurl.com/s9cmzmf
......Avid FY20 (fye 4-30-20) revenues guidance: $55-59M; committed backlog=$58mm at 1-31-2020.
7-8-19: Avid Signs New Top10 Global Pharma and Expands Existing Cust. Relationship https://tinyurl.com/yyq8zgb9
6-27-19: "The 5 new clients signed in late FY2018 contributed significantly to revenue diversification in FY2019." SEE CUST SPLITS for FY19: http://tinyurl.com/y5j4dlsv
4-24-18: ”In recent weeks, Avid has signed new agreements with 3 addl. undisclosed intl. drug dev. companies (that’s 4 thus far in CY2018)” https://tinyurl.com/y89whc8d
3-1-18: Acumen Pharm. selects Avid for Process Dev. & Mfg. of ACU193 (Alzheimer’s) https://tinyurl.com/y8jvwleq http://www.acumenpharm.com
2-21-18: Enzyvant selects Avid to commercially mfg. RVT-801 (Farber disease) https://tinyurl.com/yd5xhcx8 http://www.enzyvant.com
2-12-18: Peregrine’s Legacy PS-Targeting IP Sold to ONCOLOGIE INC. (Boston, CEO: Laura E. Benjamin) for $8M/upfront, $95M/milestones https://tinyurl.com/yam8gb3h
...NOTE: 2-13-18: Oncologie Licenses Mologen’s immunotherapeutic ‘lefitolimod’ (TLR9 agonist) https://tinyurl.com/y9z54f4x “ONCOLOGIE is backed by top-tier intl. investors and has the objective to dev. novel personalized medicines in the field of immuno-oncology."
2-28-17: Avid & Cook (acq. by Catalent 12-2017 for $950M; $179Msales x 5.3; EV/EBITDA=17.3) remain Halozyme's 2 CMO's (“working to scale-up/validate/qualify Avid II/Myford for Roche collab.”): http://tinyurl.com/h75teta
FACILITIES: https://avidbio.com/facilities/
10-8-19: Avid Announces Completion of Expansion of Process Dev. Capabilities & Labs https://tinyurl.com/y3hfwst9
...will “significantly accelerate Avid’s ability to drive efficient & rapid on-boarding of new cust. pgms progressing to mfg.”
4-24-18: Avid adding Process Dev. Labs, upgrading capabilities (in phases; total will be >6000sq; 1st one ready Q3CY18/Jul-Sep'18) https://tinyurl.com/y89whc8d
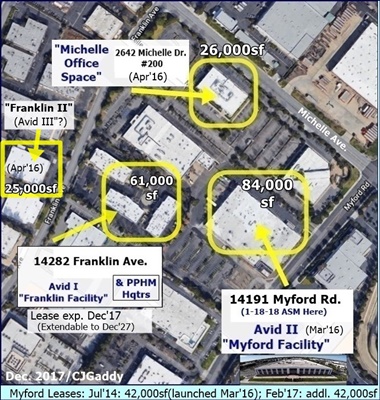
5-2017: Avid II (Myford) adds 2 MilliporeSigma Mobius 2,000L single-use bioreactors; total mfg. capacity now ">11,000L". http://tinyurl.com/ky7bmu4
5-10-17: Halozyme comments on Avid II(Myford) expansion in their 3-31-17/10Q pub. 5-9-17: http://tinyurl.com/mrl34uk
..."validation of the new facility is scheduled to end in Q2/2017… Once this new facility is approved, it will become the primary source for Roche of bulk rHuPH20.”
6-2-16: Corp.Update – Avid Expansion & Drug Development - http://tinyurl.com/zvmhqmr
3-7-16/Avid II: Formal Commissioning of Avid's New 40,000sq "Myford" Facility, “single-use/fully disposable” (potentially $40M addl revs) http://tinyurl.com/y5jmfpo3
12-10-15/Avid II: Avid Expansion into MYFORD Facility now GMP-run ready (potential +$40mm sales) - contemplating further expansion http://tinyurl.com/y539yut4
12-10-14: Avid to Double Mfg. Capacity(“Myford Expansion”) http://tinyurl.com/y2sqoy2u & http://tinyurl.com/kmdgq8t

PRESENTATIONS & ARTICLES: https://avidbio.com/events/
Sep17/Fireside Chat w/Nick Green: 18th Annual Morgan Stanley Global Healthcare Conf.: https://tinyurl.com/y4cbjjrc
5-18-20: Avid Listed In Top CMO Awards Article https://tinyurl.com/yb5cnh6m
...”Avid received 5 CMO Leadership Awards categories, incl. 2 Champion designations: Expertise & Service.”
10-25-18/BioPharma: Roger Lias Interview, “Avid expansion to Triple Process Dev. Capacity” https://tinyurl.com/ybtuk29m
Sept2018/BioTechWEEK: Tracy Kinjerski (VP/BusOP) interview: Avid’s differentiators (6mins.) https://www.youtube.com/watch?v=x3tzo4pZivk
1-29-18: NobleCon14 - 14th Annual Inv. Conf., Ft.Laud. => Roger Lias' webcast/slideshow https://tinyurl.com/yanwk9yo
11-15-17/Outsourcing-Pharma: “How This R&D Company (Peregrine) is Transitioning to a Pure-Play CDMO: ‘Opportunities Are Almost Endless’" - Recap of interviews with Steve King & Avid Pres. Roger Lias http://tinyurl.com/y7dv9faz
CONFERENCE CALLS & ASM's:
12-2-20: Qtly. Conf. Call (Green/Hart/Compton) PR & Transcript https://tinyurl.com/y66xk888
10-20-20 Annual Shareholders Meeting - Replay (9:45): https://www.virtualshareholdermeeting.com/CDMO2020 (Control# not needed)
9-1-20: Qtly. Conf. Call (Green/Hart/Compton) PR & Transcript https://tinyurl.com/y329llow
...CEO N.Green: ”My first few weeks have done nothing but confirm my view that I believe Avid to be a strong and state-of-the-art company with significant opportunity for growth."
6-30-20: Qtly. Conf. Call (Hancock/Hart/Compton) PR & Transcript https://tinyurl.com/y9zbgmos
...RickH: "Based on our customers' growing demand and our expanding business development activity, we believe that we will significantly increase capacity utilization in 2021 and beyond. Accordingly, we have entered into a new phase of planning for the expansion that will support our growing business in the years to come.”
...”Top10 Pharma customer added 7-2019 named in 10-K: GILEAD SCIENCES.”
3-10-20: Qtly. Conf. Call (Hancock/Hart/Compton) PR & Transcript https://tinyurl.com/s9cmzmf
12-9-19: Qtly. Conf. Call (Hancock/Hart/Kinjerski) PR & Transcript https://tinyurl.com/tfewuzc
9-5-19: Qtly. Conf. Call (Hancock/Hart/Kinjerski) PR & Transcript https://tinyurl.com/y6t8zfur
6-27-19: Qtly. Conf. Call (Hancock/Hart/Kinjerski) PR & Transcript https://tinyurl.com/y6zdjyu2
...Rick Hancock, "Most importantly, during Q4, the Company achieved positive income, generating cash from dev. & mfg. operations for the 1st time since the beginning of the CDMO transition (Jan'18)."
3-11-19: Qtly. Conf. Call (Lias/Hart) PR & Transcript http://tinyurl.com/yy6dvjvc
12-3-18: Qtly. Conf. Call (Lias/Hart//Kinjerski) PR & Transcript https://tinyurl.com/y9n374kp
10-4-18: ASM/2018 (@Myford Facility) - Roger Lias’ Slideshow & Attendee Report https://tinyurl.com/yctfzhlb
9-10-18: Qtly. Conf. Call (Lias/Hart) PR & Transcript https://tinyurl.com/y8oc6hx8
...Roger Lias, "During the qtr, we advanced the projects of our existing active clients and continue to engage with numerous potential new customers."
7-16-18: Qtly. Conf. Call (Lias/Kinjerski) PR & Transcript https://tinyurl.com/yaozdggz
...Roger Lias, “In a short period, we’ve established a targeted business dev. operation that is actively providing visibility for Avid Bioservices within our fast growing but competitive marketplace.”
3-12-18: Qtly. Conf. Call (Lias/Kinjerski/Lytle) PR & Transcript https://tinyurl.com/yakdl4wj
...Roger Lias, ”I'm pleased to be able to report that in a very short period of time we've generated significant interest from both emerging & growth biopharmaceutical players and from pharmaceutical multinationals. I'm confident that the plan we're executing will drive a considerable increase in backlog and the opportunity to further enhance capacity utilization in the future.”
......From the 3-12-18 PR: “At present, we are in late-stage negotiations with several potential new customers and expect to announce the executed agreements before the end of the FY[4-30-18].”
1-18-18: ASM/2017 (@Myford Facility) - Roger Lias’ Slideshow & Attendee Reports https://tinyurl.com/yca6enbr 12-7-17 PROXY/14A: https://tinyurl.com/y7qprpg9
12-11-17: Qtly. Conf. Call (Lias/Lytle) Transcript https://tinyurl.com/ybycb2s6
...Roger Lias, "the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
9-11-17: Qtly. Conf. Call (King/Lias/Lytle) Transcript http://tinyurl.com/y9y8qdac
...Steve King: “For this reason, we have concluded that in order to best position Peregrine’s R&D assets for successful development, they should be advanced by a partner with the appropriate expertise and ample resources to invest in the necessary clinical trials. To that end, we have been working diligently towards the transformation of the overall business to becoming a pure-play CDMO, while assessing the best strategic options for the R&D assets that would allow stockholders to directly see the future value from their continued developments. By partnering & eliminating future R&D expenditures, we believe we are best positioning Avid for future growth. Through reinvestment & expansion, we believe we will attract new customers and extend current contracts that will help position Avid as a leading U.S. CDMO. We are moving forward expeditiously with strategic discussions as we recognize the need to move quickly both from the R&D & CDMO standpoints. We hope to bring this process to completion over the coming months and will update you on our progress.”
7-14-17: Qtly. Conf. Call (King/Shan/Lytle) Transcript http://tinyurl.com/yb4wulvu
...Steve King: “We are seriously considering the possibility of separating our 2 distinct businesses, Avid and R&D/PS-Targeting.”
3-13-17: Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
...Steve King: “We believe the recent improvement of stock price is a growing recognition of the value of Avid, and having the full value of the Avid business reflected in our stock price is a top priority.”
12-12-16: Qtly. Conf. Call (King/Shan/Hutchins/Lytle) Transcript http://tinyurl.com/hhn4gga
...Steve King: “Our goal is to bring the overall company to profitability within the next 18mos. We believe just the value of Avid Bioservices is far greater than our current market cap and is only growing in value."
10-13-16 ASM/2016: ATTENDEE Reports & Link to CEO Steve King's 35min/45slide webcast: http://tinyurl.com/jx7ouay
Feb. 2018: Peregrine Pharmaceuticals completed its Mid’17-Early’18 Transition to a Pure-Play CDMO
(Contract Development & Manufacturing Organization), Avid Bioservices, Inc. - CEO: Roger Lias.
**FULL PPHM=>CDMO Transition History (Ronin/SWIM): https://tinyurl.com/ybqvzwhg
1-8-17: Peregrine chgs. name to “Avid Bioservices, Inc.”; new ticker: “CDMO” (+CDMOP) https://tinyurl.com/y8vhjow4
2-12-18: Peregrine’s Legacy PS-Targeting IP Sold to ONCOLOGIE INC. (Boston, CEO: Laura E. Benjamin) for $8M/upfront, $95M/milestones https://tinyurl.com/yam8gb3h
...12-13-18/8-K: Overview of Oncologie sale: https://tinyurl.com/yab9c6cr
NOTE: “PS-targeting Exosome tech. not included; back to UTSW", see: https://tinyurl.com/yakdl4wj
Click here for an ARCHIVE of the History of Peregrine’s Anti-PS/Bavituximab Platform - MOA, Trials, and Activity over the years, from early 2000’s thru 2017, prior to being Sold to ONCOLOGIE:
https://tinyurl.com/y8pq4rhc
Of Interest (post Oncologie Sale):
Oncologie's website: https://oncologie.com/true-home-v2/ NEWS: https://oncologie.com/newsroom/
4-20-18/AACR’18: MSKCC(LudwigCC) Tweets about 2 WolchokLAB/”PPHM” Anti-PS Posters https://tinyurl.com/ycgjhvqa
4-26-18: New Bavi+Keytruda/LIVER Ph2 IST Trial, Sponsor=UTSW, Collab=MERCK https://tinyurl.com/y7fd9vdb
6-7-18: Oncologie Obtains $16.5M Seed Funding Led by Pivotal bioVenture Partners China Fund https://tinyurl.com/ybrrbgg7
6-7-18/BioCentury: Laura Benjamin states, “Oncologie plans to begin a trial mid-summer '18 evaluating Bavi to treat HCC and a P-O-C trial in Gastric cancer in 1Q19/2Q19.” https://tinyurl.com/ycb8r7sm
12-3-18: During q/e 10-31-18, Avid sold remaining legacy R&D asset, r84(anti-VEGF), to Oncologie for $1.0M upfront. Avid is eligible to receive up to an addl. $21M in dev/reg./comm. milestones, and low to mid-single digit royalties on net sales upon commercialization of products utilizing r84 https://tinyurl.com/y9n374kp
11-4-19/PharmaBoardroom: Interview with Oncologie CEO Laura Benjamin discussing the 2 ongoing Bavi Trials w/Keytruda (USA UK Taiwan S.Korea) https://tinyurl.com/ydf6zhsv
...Dr. Benjamin, "We have high expectations of bavituximab and have obtained the global rights for this compound."
...Ongoing Trial #1 (N=80): Open Label, Bavi+Keytruda Adv. Gastric/GEJ Cancer https://clinicaltrials.gov/ct2/show/NCT04099641
......1-17-2020 ASCO/GI Poster: https://oncologie.com/wp-content/uploads/2020/01/2020-01-17-ASGO-GI-poster-final-draft.pdf
...Ongoing Trial #2 (N=34 UTSW): Open Label, Bavi+Keytruda Adv. Hepatocellular Carcinoma https://clinicaltrials.gov/ct2/show/NCT03519997
6-11-20/Fiercebiotech: Oncologie Reels In $80M To Push Clinical Pgms/Build Pipeline https://tinyurl.com/yany8f34 & https://tinyurl.com/y8p9artk
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
