Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
The put profits are enormous with this week's triple witching and should help drive the market higher over the next few weeks. Avid seems to be like a canary in the coal mine!
Trades so far tend to back this up....from zerohedge..."Will Today's "Massive" $3.4 Trillion Op-Ex Also Spark A Huge Short Squeeze"...covering to last until tuesday...r
Unusual
Date/Time Price Shares Exch/Mkt
06/17/2022 9:30:01 EDT F 12.66 100 NSD
06/17/2022 9:30:01 EDT I 12.64 2 IEX
06/17/2022 9:30:01 EDT I 12.79 3 IEX
06/17/2022 9:30:00 EDT 12.79 100 IEX
06/17/2022 9:30:00 EDT X 12.64 64913 NSD
06/17/2022 9:29:59 EDT T 12.61 100 NSD
eb0783, it is better this way. PR when there is something to say and else be quite.
To many PRs to say to little makes people not read them anymore.
Now we have 3 good PRs with useful content and good look out.
AIMO
We are on schedule with our build-out....we are in a good position covering
our loan with low interest rates.....the CDMO industry is in dire need of more available
capacity.......we are going to be a significant player in this market with close to 400Mil. capacity...............i would argue $12 is a pretty darn good entry point!!
BUT, Bear Markets tend to sink all ships....Nonetheless, I can't think of a better
industry with such huge expansion projections than ours...GLTA....
Not that it does any good in this market. My last buy was at 12.02.
Wow, 3 PRs in 4 days should help quell the chatter from those complaining about not hearing from the company.
Apparently the company is highlighting the fact that they are meeting their stated goals of when the expansion projects would be completed and affirm the sync up with the upcoming rev projections...imao...r
Avid Bioservices, Inc. and CRB highlight efforts that enabled opening of first phase of new viral vector facility in California in only eight months
June 16 2022 - 08:00AM
PR Newswire (US)
Alert
Print
Share On Facebook
53,000-square-foot facility addresses unique demands of commercial-scale viral vector production
COSTA MESA, Calif., June 16, 2022 /PRNewswire/ -- Avid Bioservices, Inc. (NASDAQ:CDMO), a dedicated contract development and manufacturing organization (CDMO) focused on development and CGMP manufacturing services for biotechnology and pharmaceutical companies, today highlighted the companies' collaborative efforts that enabled the phase-one opening of its new purpose-built, 53,000-square-foot viral vector and CGMP facility in Costa Mesa, California, in only eight months.
The facility was designed and constructed by CRB – a leading global provider of sustainable engineering, architecture, construction, and consulting solutions to the life sciences industry – and built using the company's innovative ONEsolution™ project delivery approach.
By leveraging a single project team to align with Avid on cost and schedule without sacrificing quality, CRB's design and execution fulfills Avid's mission to address a critical gap in the treatment of oncology and rare disease by prioritizing the specific needs of the unique process development and manufacturing of viral vectors used in those treatments.
"Expanding our offerings into viral vectors was a natural adjacency for Avid, as previously explained by our CEO, Nick Green. However, the work really came down to finding a design-build partner with the process, design, and construction expertise to help us deliver a best-in-class facility within a constrained schedule and budget. CRB has done a great job filling this need for Avid and partnering with us to ensure this new facility meets the needs of current and future clients," said Mike Alston Jr., director of project engineering for Avid Bioservices. "We have been able to leverage dedicated design and construction teams for phase 1 and 2 to enable continuity across the facility, and also placed a major focus on early procurement and scheduling. Considering the challenges of the current supply chain environment, it has been nothing short of remarkable for Avid and CRB to be opening the doors of Avid's new viral vector process development labs here in June."
The state-of-the-art facility at full build-out will include capacity for adherent and suspension cultures. The second phase is expected to become operational in mid-calendar year 2023.
"When presented with the opportunity to lead and support Avid's organic growth and expansion of our business in the cell and gene therapy CDMO space, I was tremendously excited to be a part of the team. Next when given the project parameters, schedule, budget, the first thing that came to mind, was ensuring we had trusted partner that could anticipate our needs and could work collaboratively and creatively to achieve our goals," said Rami Barghout, director of viral vector facilities for Avid Bioservices. "To that end, CRB has been an instrumental partner technically and in navigating us through construction to achieve our aims."
"We're so excited to have worked alongside Avid to help them realize their vision for a facility that answers an important but unmet need through the commercial-scale production of viral vectors," said Matthew Khair, CRB's vice president for the Western Region. "The close collaboration, enhanced planning and cultural alignment between teams created an unrivaled project experience and resulted in a facility that can close that production gap and speed therapies to patients."
About Avid Bioservices, Inc.
Avid Bioservices (NASDAQ:CDMO), an S&P SmallCap 600 company, is a dedicated contract development and manufacturing organization (CDMO) focused on development and CGMP manufacturing of biologics. The company provides a comprehensive range of process development, CGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With 28 years of experience producing monoclonal antibodies and recombinant proteins, Avid's services include CGMP clinical and commercial drug substance manufacturing, bulk packaging, release and stability testing and regulatory submissions support. For early-stage programs the company provides a variety of process development activities, including upstream and downstream development and optimization, analytical methods development, testing and characterization. The scope of our services ranges from standalone process development projects to full development and manufacturing programs through commercialization. www.avidbio.com
About CRB:
CRB is a leading provider of sustainable engineering, architecture, construction, and consulting solutions to the food and beverage and life sciences industries. Led by its innovative ONEsolution™ service, CRB provides successful integrated project delivery for clients demanding high-quality solutions, on time and within budget. The company's more than 1,600 employees provide world-class solutions that drive success and positive change for their clients, people, and communities. CRB is a privately held company with a rich history of serving clients throughout the world, consistently striving for the highest standard of technical knowledge, creativity, and execution.
CONTACT:
Avid Bioservices Inc.:
Stephanie Diaz (Investors)
Vida Strategic Partners
415-675-7401
sdiaz@vidasp.com
Tim Brons (Media)
Vida Strategic Partners
415-675-7402
tbrons@vidasp.com
Clarity Quest Marketing:
877-887-7611
Bonnie Quintanilla, bonnie@clarityqst.com
CRB:
816-200-5234
Chris Clark, chris.clark@crbgroup.com
https://fintel.io/so/us/cdmo
BLACKROCK, 10 Trillion under management, and SYNBIO
Largest shareholders include BlackRock Inc., Fmr Llc, IJR - iShares Core S&P Small-Cap ETF, State Street Corp, Vanguard Group Inc, XBI - SPDR(R) S&P(R) Biotech ETF, AltraVue Capital, LLC, Ranger Investment Management, L.P., VTSMX - Vanguard Total Stock Market Index Fund Investor Shares, and Invesco Ltd..
AVID at BIO2022. Photos.
Thank you to everyone who came out to make our #BIO2022 evening event in #SanDiego! We had a great turnout of both employees and guests. Check out the event with some photos below!#limitlesstogether #biotech #biopharma #genetherapy #cellandgenetherapy #cdmo pic.twitter.com/HeNmXouP64
— Avid Bioservices (@AvidBioservices) June 15, 2022
It would help if there would be a public statement of the fy2023 rev projections b4 the yr end cc...today would have been a good opportunity to do so even if it were a "range"...imao...r
My favorite parts.
"Today’s launch of our AD/PD labs is a crucial first step in our expansion into the viral vector space"
"we remain laser focused on smart and sustainable revenue growth for our business"
I think Avid is going to grow faster than we imagined
I might add that the 500L reactor in the new viral vector PD lab could suffice for client clinical trials giving the new lab an output that we don't normally consider. Note this again from our 2019 PD Lab tour, speaking of their 200L reactor: "The product from the pilot-scale reactor belongs to the client to use as they need. The client can run preclinical trials with this pilot size batch."
Again, this 500L size within the lab could be a nice strategic move.
Wow! Viral Vector labs are up and running. Two lab PRs in two days! Makes me envision a boxer throwing a one-two punch. Smart move to have the PD lab running before the rest of the Viral Vector facility is ready. It comes first in the overall process so they can be making a few $$million before even manufacturing the real load. Also notice that this lab has an intermediate-scale-up reactor of 500L, more than twice what the existing mammalian PD lab has. From the 2019 lab tour: "The new PDL also has a 50L and 200L pilot plant (mid-range/intermediate scale)." I'm not sure if that is a strategic/progressive step up or if it is simply peculiar/necessary for viral vector development. Opinions?
Avid Bioservices Announces Launch of Analytical and Process Development Suites Within New, World-Class Viral Vector Development and Manufacturing Facility
June 14, 2022 at 8:05 AM EDT
Download PDF
AD/PD Laboratories Open Exactly Eight Months After Announced Plans to Build Viral Vector Facility
Build-Out of Facility’s CGMP Manufacturing Suites Continues, Expected to Come Online in Mid-Calendar 2023
TUSTIN, Calif., June 14, 2022 (GLOBE NEWSWIRE) -- Avid Bioservices, Inc. (NASDAQ:CDMO), a dedicated biologics contract development and manufacturing organization (CDMO) working to improve patient lives by providing high quality development and manufacturing services to biotechnology and pharmaceutical companies, today announced the opening of the analytical and process development (AD/PD) suites within the company’s new, world-class viral vector development and CGMP manufacturing facility. The launch of the AD/PD labs comes eight months to the day that Avid announced its intention to expand its CDMO service offering into the rapidly growing cell and gene therapy market. Build-out of the viral vector facility’s CGMP manufacturing suites is ongoing, with those capabilities expected to come online in mid-calendar 2023.
The company’s new AD/PD labs are equipped with complete upstream, downstream and analytical development capabilities and are able to support up to 500 liter suspension culture in single-use bioreactors and various adherent cell modes. The company’s viral vector AD/PD team, which is led by Elie G. Hanania, Ph.D., has deep expertise in developing and implementing state-of-the-art enabling technologies for viral vector production and purification processes focused on adeno-associated viruses (AAVs), lentiviruses, oncolytic viruses and other viruses for gene therapy and vaccine applications.
“Avid’s entry into the cell and gene therapy sector is a critical component of the company’s growth strategy over both the short- and long-term. We believe that we are uniquely positioned to leverage our deep expertise in the manufacturing of traditional biologics to address the growing demand for high quality manufacturing of cell and gene therapies,” said Nick Green, president and chief executive officer of Avid Bioservices. “Today’s launch of our AD/PD labs is a crucial first step in our expansion into the viral vector space as we remain laser focused on smart and sustainable revenue growth for our business. We now look forward to engaging in a meaningful way with potential viral vector clients and offering this latest tangible evidence of the quality of Avid’s facilities and the capabilities of our team.”
In October 2022, Avid announced plans to construct a purpose-built 53,000 square foot viral vector facility in Costa Mesa, CA, approximately five miles from Avid’s existing operations in Tustin, CA. The strategic decision was driven by continued strong growth in the cell and gene therapy market combined with the CDMO industry’s overall lack of proven, high-quality CGMP manufacturing expertise and capacity for viral vectors. With more than 17 years of experience in commercial manufacturing of biologics underpinned by a strong quality ethos and a customer-centric approach to doing business, Avid offers a strong value proposition to prospective customers in the cell and gene therapy market.
“The opening of our newly constructed viral vector process and analytical development laboratories within eight months of our announced investment is truly a testament to the capability and dedication of our Avid team, as well as our partners at CRB. We are excited to begin working on customer projects in this new facility and look forward to completing the ongoing construction of our new vial vector CGMP manufacturing suites,” said Drew Brennan, general manager of viral vector technologies of Avid Bioservices.
About Avid Bioservices, Inc.
Avid Bioservices is a dedicated contract development and manufacturing organization (CDMO) focused on development and CGMP manufacturing of biologics. The company provides a comprehensive range of process development, CGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With 28 years of experience producing monoclonal antibodies and recombinant proteins, Avid's services include CGMP clinical and commercial drug substance manufacturing, bulk packaging, release and stability testing and regulatory submissions support. For early-stage programs the company provides a variety of process development activities, including upstream and downstream development and optimization, analytical methods development, testing and characterization. The scope of our services ranges from standalone process development projects to full development and manufacturing programs through commercialization. www.avidbio.com
Forward-Looking Statements
Statements in this press release which are not purely historical, including statements regarding Avid Bioservices, Inc.'s intentions, hopes, beliefs, expectations, representations, projections, plans or predictions of the future, are forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements involve risks and uncertainties including, but not limited to, the risk that the company may experience delays in the construction of viral vector facility, the risk that expanding into a new biologics manufacturing segment may distract senior management’s focus on the company’s existing operations and/or its current expansion of the Myford facility, the risk that the company may experience delays in hiring qualified individuals into the viral vector business, the risk that the company may experience delays in engaging initial customers for the viral vector business, and the risk that the viral vector business may not become profitable for several years, if ever . Our business could be affected by a number of other factors, including the risk factors listed from time to time in our reports filed with the Securities and Exchange Commission including, but not limited to, our annual report on Form 10-K for the fiscal year ended April 30, 2021 and subsequent quarterly reports on Form 10-Q, as well as any updates to these risk factors filed from time to time in our other filings with the Securities and Exchange Commission. We caution investors not to place undue reliance on the forward-looking statements contained in this press release, and we disclaim any obligation, and do not undertake, to update or revise any forward-looking statements in this press release except as may be required by law.
Contacts:
Stephanie Diaz (Investors)
Vida Strategic Partners
415-675-7401
sdiaz@vidasp.com
Tim Brons (Media)
Vida Strategic Partners
415-675-7402
tbrons@vidasp.com
OK, posting here from London…lovely weather and a cityscape full of cranes….packed bars, pubs and restaurants….then I check out the markets and read CNBC and it seems the sky is falling(the market surely is)!
Is a recession in the cards? Not sure but certainly not apparent here…yet anyway!
As for Avid….Nick appears to be doing a great job on a solid growth path of revenue and profits and at one point we will all benefit from this…for now we are caught in very volatile, scared and skiddish market….our time is coming….we just need to wait this out.
If you don’t have to sell, I would advocate hanging in there. All IMO!
Well institutional holdings % over on fintel have remained unchanged for over a week plus...and its like a spigot has been turned off on the daily buy/sells by them since 6/1(even index driven ones?)...and the quarter ending in less the 3 weeks...along with the CC...r
Anyone else feel like we are screwed? The whole market is a mess. All markets are down.
No flight to quality, interest rates are up? Inflation high and gold is down?
BTC crashing.
How does this end? Not pretty I'm sure
It's hard to be optimistic
What's CDMO with future Rev of $370 million worth?
YES! This is a doubling of the then-brand-new PD suite that we toured in 2019. Very nice.... very strategic
See my post 332104.
"rapidly grow our revenue"
Those are beautiful words!!!
Avid Bioservices Announces Further Expansion of Process Development Capacity for Mammalian Cell Business
June 13, 2022 at 8:05 AM EDT
Download PDF
New Laboratories Have Potential to Support an Additional $20 Million in Annual Process Development Revenue, Doubling Current Mammalian Cell Process Development Capacity
Expansion to Cost Approximately $6 Million with Completion Anticipated by End of Calendar 2022
TUSTIN, Calif., June 13, 2022 (GLOBE NEWSWIRE) -- Avid Bioservices, Inc. (NASDAQ:CDMO), a dedicated biologics contract development and manufacturing organization (CDMO) working to improve patient lives by providing high quality development and manufacturing services to biotechnology and pharmaceutical companies, today announced that the company is further expanding its process development capacity for its mammalian cell business. As part of these efforts, Avid is expanding its state-of-the-art laboratories which could support an additional $20 million in annual process development revenue, doubling the company’s current process development capacity. The company estimates that this expansion will cost approximately $6 million and completion is anticipated by the end of calendar 2022. This expansion follows previous investments in increased process development capacity made in fiscal year 2019 and the most recent addition of high throughput capabilities in both upstream and downstream processing in fiscal year 2022.
Process development represents a vital CDMO function and one through which Avid provides a critical advantage to customers in both early and late stages of development. This planned expansion further strengthens the company’s capabilities in the areas of R&D support, process development, process characterization and validation, and pilot scale production for a broad range of mammalian cell-based biotechnology products. The new suites will further enhance Avid’s ability to develop and deliver cost-effective, robust, scalable and compliant processes and to drive efficient and rapid on-boarding of new customer programs progressing to CGMP manufacturing.
“Avid continues to diligently and strategically expand our capacity across all segments of our CDMO business in order to ensure that we are always able to meet the needs of current and future customers. This latest project will generate additional upstream and downstream process development capacity at the front-end of our mammalian cell business, which is critical for the efficient on-boarding of new customer projects,” said Nick Green, president and chief executive officer of Avid Bioservices. “With several expansion projects underway across various facilities and business segments, we feel that we are well positioned to take advantage of the increasing demand for high-quality CDMO services and continue to rapidly grow our revenue.”
About Avid Bioservices, Inc.
Avid Bioservices is a dedicated contract development and manufacturing organization (CDMO) focused on development and CGMP manufacturing of biologics. The company provides a comprehensive range of process development, CGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With 28 years of experience producing monoclonal antibodies and recombinant proteins, Avid's services include CGMP clinical and commercial drug substance manufacturing, bulk packaging, release and stability testing and regulatory submissions support. For early-stage programs the company provides a variety of process development activities, including upstream and downstream development and optimization, analytical methods development, testing and characterization. The scope of our services ranges from standalone process development projects to full development and manufacturing programs through commercialization. www.avidbio.com
Forward-Looking Statements
Statements in this press release which are not purely historical, including statements regarding Avid Bioservices, Inc.'s intentions, hopes, beliefs, expectations, representations, projections, plans or predictions of the future, are forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements involve risks and uncertainties including, but not limited to, the risk that the company may experience delays in the construction of new process development laboratories. Our business could be affected by a number of other factors, including the risk factors listed from time to time in our reports filed with the Securities and Exchange Commission including, but not limited to, our annual report on Form 10-K for the fiscal year ended April 30, 2021 and subsequent quarterly reports on Form 10-Q, as well as any updates to these risk factors filed from time to time in our other filings with the Securities and Exchange Commission. We caution investors not to place undue reliance on the forward-looking statements contained in this press release, and we disclaim any obligation, and do not undertake, to update or revise any forward-looking statements in this press release except as may be required by law.
Contacts:
Stephanie Diaz (Investors)
Vida Strategic Partners
415-675-7401
sdiaz@vidasp.com
Tim Brons (Media)
Vida Strategic Partners
415-675-7402
tbrons@vidasp.com
New tweet on twitter of progress in Costa Mesa Viral Vector facility
Avid Bioservices
@AvidBioservices
·
6h
Our #viralvector facility in #CostaMesa California is making great progress! Check out the latest timelapse of our #processdevelopment and office area being built
Our #viralvector facility in #CostaMesa California is making great progress! Check out the latest timelapse of our #processdevelopment and office area being built#cdmo #biotech #biopharma #cellandgenetherapy #genetherapy pic.twitter.com/bv2qFDooPu
— Avid Bioservices (@AvidBioservices) June 11, 2022
eb
Last time I drove past Myford end of April they had moved the two flat bed mounted generators that were behind the building and the blue equipment(don't know exactly what it was) The two generators were move to the side of the building. The side where we went into the building at the 2019 ASM
I assume they moved them to start building the new power plant?
In this weeks timelapse you'll see 30 scissor lifts working simultaneously within Myford South, our 42,000 sq. ft. #CGMP #drugsubstance manufacturing site.#biotech #biopharma #cdmo #contractmanufacturing pic.twitter.com/4FdcVlohx4
— Avid Bioservices (@AvidBioservices) June 9, 2022
Horselover, Ask him to go by, maybe into the parking lot, Myford and see if the walls and roof are up on the back addition (Central Utility Plant) to the building.
In my view/opinion, it is not just a “main raw floor.” Much of the construction is laid underneath/within that floor. Watch the beginning half of the video again. There are multiple, multiple pipes, conduits, and cables laid before the floor is poured. Moreover, the ductwork beyond this space (back into Myford North and South), along with the necessary cabling, conduit work, and piping would have been done when the clean rooms, labs, and other spaces were built out. The additional work needed in this space, beyond what is in the floor, is twofold. First is setting and connecting the power, refrigeration, air handling & purifying, and possibly water, etc. equipment. The connections are already at the specified equipment locations due to being laid under the floor. The delivery timeline is dependent on the equipment vendors and no more real construction is needed. Second, I doubt that there will be any internal wall/ceiling construction within the utility site because it is not needed. It should/could be just one big utility room. The only additional construction should be exterior walls and roof. I don’t see that taking much longer than, and concurrent with, receiving & setting up the equipment. This is only my opinion from my experience as a project engineer, although I haven’t been involved in this specific type of facility. I have, however, had 4 tours of the Myford site with the first one in 2015 when construction was not quite finished. Other opinions are welcome.
Thanks,
He is planning to go on Monday.
3030 Airways Ave Costa Mesa
The building backs up to the runway at John Wayne airport.
No mystery. The name was on the building in February.
Tell you're friend that you can drive along the right(west) side and if some of the big receiving doors are open you can see inside.
There is a picture of the building from February in post# 341389
I'll be out there next week I will try to get over there also, get a few shots
I'm sure you have the physical address in Costa Mesa.
Tried to look it up on their site but couldn't find it/. I even sent Avid an email asking about it but still no reply.
I have a friend who is also invested and lives out there. He wants to go by and take a couple of pictures.
Is there a mystery about this new place in Costa Mesa??
eb0783, thanks a lot.
I assumed that the construction should have been more advanced then just the main raw floor. I didn't think to analyze the title but your assumption makes sense.
And indeed, July 2022 is in the future, so it must be January 2022.
AIMO
eb, thanks. good timeline as to construction.
Protector, “Myford updated 7:1:2022.mp4” is the title of this video that was uploaded on 6/1/22. 7:1:2022 would be July 1st so that date in the title must be a European format (day.month.year). I will assume that the video was shot/finished on January 7th of this year, 5 months prior to today. This space where they are putting the Central Utility Plant was the back parking area before they built “Myford North.” This space, right behind the building, is where they did have the backup diesel, generator, and refrigeration units on trailers for the original Myford facility. This is also where employees walked back and forth to the PD Lab and the original Avid I facility. It makes sense that the new Central Utility Plant would/should be built there.....and it could well be finished.
djohn, Sorry, here is the link again.
Avid Ext. II
You're the best. I think it is good we keep an eye on this stuff personally vs. relying on "them" to feed us info!
I emailed Stephanie a few weeks ago and asked if there where any updates on the expansions at Myford or Costa Mesa VV. She said that they were in a "quite time" because of end of quarter and release of results. All updates will be released and discussed on the quarterly report and CC.
I will have some time to go by and get a few pictures and post them.
Thanks! now I see it. The construction area behind the blue fence looks pretty much the same as the last time I was there about 5 weeks ago. I remember the stairs going up to the top of building was there.
I for one would really appreciate that. I wonder, since you're going to be up that way, if calling them/Stephanie or..??? could get you a quick tour if your time allowed?? Sitting here in the bottom right corner makes doing that impossible for me but if you did it would be MOST APPRECIATED and thank you for sharing!
djohn, remove the "
The video link doesn't work for me.
I will be in Tustin next week and can do a drive by at both Myford and Costa Mesa see what construction looks like.
Those have to be early videos.
They wouldn't have all the new job openings if those were up to date constructions.
Question, is it just me or do these AVID construction works look early stage compared to where PII extensions need to be (were said to be) expected to be ready.
https://vimeo.com/716162683" rel="nofollow" target="_blank" >https://vimeo.com/716162683[tag]Myford updated 7:1:2022.mp4
6 days ago[/tag]
This is 6 days old, of course the video images can date from some months ago. Do some of you that sometimes drive by the site have any comments?
thx
The SP is quietly going upward by 0.01 increments and less than 2,000 shares.
I originally got into this stock because of the work to fight glio.
This is good news, for patients and maybe us down the road too.
https://www.yahoo.com/now/oncxerna-therapeutics-announces-upcoming-asco-120000707.html?guccounter=1&guce_referrer=aHR0cHM6Ly93d3cuZ29vZ2xlLmNvbS8&guce_referrer_sig=AQAAAFTGDDZk0x2hn9MuCYdIvq3snlfwHlaABV_RVVXNTuWrDH_K5_lj7iaT1LS3Yc3f4MVvWGjDutvufZakQ_pBgxJCNBPQDJzolDifOtY-QPSKjjczn-V1l-S0kiHX6vaab0CYmPRgMWUIQQwQR1aE47GhE6eosATWddeC_QlaS8RB
I think we're up 5% because of their employee appreciation lunch video... Lol
https://vimeo.com/716255459?ref=em-share
Saw this on the NYT site behind the paywall so i couldn't read the entire article but now out on dailymail site...very good news for the 5-10% of patients who have the marker... https://www.dailymail.co.uk/health/article-10890467/Doctors-left-shocked-clinical-trial-cancer-drug-cures-disease-participant.html ...r
Hello CP;
Thanks for the info. I thought it very odd that there were no posts for several days. I thought that perhaps there was an issue with the IHub website. I don't post very often but, I read this board on a daily bases. I also noted that there were no posts on the HALO board for a week now. But there, that's not that unusual.
Regards
golfho
P.S. I should have listened to you WRT HGEN...I took quite a haircut...Grrrrr
golfho, yesterday it was an holiday in Europe (=some less posters) and we went up (=even less posters :) and not much else happened. But you see all the posts.
I see no post after this one...
golfho
|
Followers
|
835
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
346385
|
|
Created
|
11/07/03
|
Type
|
Free
|
| Moderators Preciouslife1 4OurRetirement | |||

Avid Bioservices, Inc. (Nasdaq “CDMO”), Tustin CA. http://www.avidbio.com
President/CEO: Nicholas Green (eff. 7-30-20 https://tinyurl.com/yczapcc7 )
Four Avid Fact Sheets a/o 2-2020: https://avidbio.com/resources/fact-sheets/
Avid Bioservices is a CDMO committed to improving the lives of patients by manufacturing products derived from mammalian cell culture for the biotechnology & biopharmaceutical industries. Services include cGMP clinical & commercial product manufacturing, purification, bulk packaging, stability testing & regulatory strategy, submission, and support. The company also provides a variety of process development activities, including cell line development & optimization, cell culture & feed optimization, analytical methods development, and product characterization.
12-2019/Video: Tour of Myford Facility (2mins.): https://vimeo.com/380135562
7-2020: AVID’s Push Towards cv19 (I don’t capitalize things I hate) contracts: https://tinyurl.com/y8wzgneh
UPCOMING EVENTS: https://avidbio.com/events/
Sep1(Tue): Q1/FY21 (qe 7-31-20) Financials & Conf. Call - Transcript: https://tinyurl.com/y329llow
Sep17/Fireside Chat w/Nick Green: 18th Annual Morgan Stanley Global Healthcare Conf.: https://tinyurl.com/y4cbjjrc
Sep21-24/Virtual: Biotech Week Conf., Boston
Sep21-24/Virtual: BioProcess Intl. US Wes Conf., Santa Clara CA
...Time TBD: Presentation by Haiou Yang, PhD "Facility-Fit Driven Dev. for a mAb Production Process"
Oct20 10amPT/VIRTUAL: Annual Shareholders Mtg. https://tinyurl.com/y28blkjn 14A Proxy: https://tinyurl.com/y46ga9el
...ASM Replay (9:45): https://www.virtualshareholdermeeting.com/CDMO2020 (Control# not needed)
Apr20-22 2021/Booth#1159: INTERPHEX, NYC
Dec2(Wed) after mkt: Q2/FY21 (qe 10-31-20) Financials & Conf. Call - Transcript: https://tinyurl.com/y66xk888
Dec14-17: Antibody Engineering & Therapeutics Conf., San Diego
Quotes: Yahoo: Yahoo.com/CDMO Nasdaq: http://www.nasdaq.com/symbol/cdmo RT: http://www.nasdaq.com/symbol/cdmo/real-time
• IR: Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com, Tim Brons (Media) 415-675-7402 tbrons@vidasp.com
SEC:
Latest 10Q 10-31-20 iss. 12-2-20 https://tinyurl.com/y3ournzr (Cash 10-31-20=$35.7mm)
Latest 10K 4-30-20 iss. 6-30-20 https://tinyurl.com/yak25nco (Cash 4-30-20=$36.3mm)
Latest DEF14A/Proxy iss. 8-27-20 (re: 10-20-20 ASM): https://tinyurl.com/y46ga9el
ALL SEC filings for CDMO: https://tinyurl.com/yc4zjyzv
CDMO's Corp. Bylaws (a/o 11-14-14): http://tinyurl.com/y8hsppea
Poison Pill adopted 3-16-06 for 10yrs: http://tinyurl.com/yvypvh 44-pg SEC filing: http://tinyurl.com/5m57ut BUNGLER explains in plain language: http://tinyurl.com/mft4nd6
...3-17-16 Poison Pill extended until 3-16-2021 https://tinyurl.com/y74glo2n (Form8A Amendment #1)
...Poison Pill TERMINATED eff. 9-23-2019 https://tinyurl.com/y2mzx3xo
SHARES:
Shares O/S as of 11-23-20=56,726,334 - history since 4-2006: https://tinyurl.com/y66xk888 (at bottom)
...A/O 10-31-20: 3,451,000 stock options outstanding at a wgt.avg. exercise price of $6.41 (pg.16 10Q).
...MarketWatch.com for CDMO (shows Float): https://www.marketwatch.com/investing/stock/cdmo
1-12-18: S-3 Shelf Registration filed for up to $125mm https://tinyurl.com/y9qtewpw
...12-14-20: Avid raises ~$34M gross, selling 3,833,335@9.00/sh. (underwriter: RBC Capital) https://tinyurl.com/y92yr9g4
...2-20-18: Avid Raises ~$21.8M net, selling 10,294,445@$2.25 (underwriter: Wells Fargo) 8-K: https://tinyurl.com/ya3nenth 424B5: https://tinyurl.com/ycpshgxl
Total INST.+LARGE Holdings now 41,617,965 73.5% a/o 9-30-20 https://tinyurl.com/y652nxpr
13 LARGEST SHAREHOLDERS a/o 9-30-20:
1. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A/Proxy: https://tinyurl.com/y9c972fa – orig. 13G filed 11-2015)
2. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
3. Blackrock Inc. (Larry Fink): 3,834,816 6.8% (-111,172 a/o 9-30-20 Nasdaq Inst.)
4. IsZo Capital Mgt. (Brian Sheehy): 3,459,888 6.1% (-88,144 a/o 9-30-20 Nasdaq Inst.)
5. Millennium Mgt. 2,678,984 4.7% (+633,918 a/o 9-30-20 Nasdaq Inst.)
6. Vanguard Group 2,659,738 4.7% (-19,553 q/e 9-30-20 Nasdaq Inst.)
7. Snyder Capital 2,302,621 4.1% (-2,258 a/o 9-30-20 Nasdaq Inst.)
8. Altravue Capital 1,973,349 3.5% (+39,534 a/o 9-30-20 Nasdaq Inst.)
9. Wellington Mgt. 1,503,162 2.7% (+236,947 a/o 9-30-20 Nasdaq Inst.)
10. Sargent Invest. Grp. 1,052,099 1.9% (-11,000 a/o 9-30-20 Nasdaq Inst.)
11. State Street 992,280 1.8% (-65,922 a/o 9-30-20 Nasdaq Inst.)
12. Portolan Capital 963,374 1.7% (+528,662 a/o 9-30-20 Nasdaq Inst.)
13. Silvercrest Asset Mgt. 878,805 1.6% (-16,746 a/o 9-30-20 Nasdaq Inst.)
Inst. Holdings (Nasdaq) - updated 45-days after each qtr-end: http://www.nasdaq.com/symbol/cdmo/institutional-holdings
INSIDER-Trans: https://tinyurl.com/ycpw4j9z (formerly PPHM thru 2017: http://tinyurl.com/ypkow8 )
Short Interest, updated twice a month: https://www.nasdaq.com/symbol/cdmo/short-interest
A-T-M (At-The-Market) Sales 3/2009 - 7/14/17 ($272,500,000gr./34,869,747sh=$7.81/sh): http://tinyurl.com/yagmu2on
PPHM shares were 1:7 Reverse Split eff. 7-10-17 (315mm/$.606=>45mm/$4.24) http://tinyurl.com/yymtzcm9
PPHM shares were 1:5 Reverse Split eff. 10-19-09 (~237mm/$.64=>~47.4mm/$3.20) http://tinyurl.com/ykuw588
Potential Value: 9-19-2017: Catalent acquires CDMO Cook Pharmica for $950M ($179Msales x 5.3; EV/EBITDA=17.3) http://tinyurl.com/yd46m8py
AVID TEAM: https://avidbio.com/leadership/ - Open Jobs: https://avidbio.applicantpro.com/jobs
7-30-20: Nicholas Green (ex-Therapure Biopharma) becomes President/CEO of Avid - 6-25-20 PR: https://tinyurl.com/yczapcc7
4-13-20: Avid Hires 2 Senior Directors of Bus. Dev: Jason C. Brady & Sylvia Hinds https://tinyurl.com/sq2679g
1-8-20: Avid Appoints Timothy Compton as Chief Commercial Officer (repl. Tracy Kinjerski) https://tinyurl.com/ydww58sn
7-26-19: Catherine Mackey, PhD (ex Pfizer VP) joins Avid's BOD, replacing Joel McComb https://tinyurl.com/y3xhqmvm
5-8-19: Richard (Rick) B. Hancock named Interim President/CEO; Roger Lias resigns https://tinyurl.com/y4pqdwyc
7-16-18: Daniel Hart (ex-ENO Holdings) joins Avid as CFO https://tinyurl.com/ychz45p2
7-16-18: Michael Faughnan (ex-Lonza/WuXi Biologics) joins Avid as Sr.Dir./BusDev/WestC https://tinyurl.com/yaozdggz (Left 5-2019)
5-29-18: Sandra C. Carbonneau (ex-Lonza) joins Avid as Dir./BusDev./EastCoast https://tinyurl.com/ybvrwn2l (left 12-2019)
5-8-18: Magnus Schroeder (ex-AGC Biologics) joins Avid as VP/Process Sciences https://tinyurl.com/y7tpswaw
2-23-18: CFO Paul Lytle voluntarily resigns eff. 5-24-18 (2-23-18 +90days), "CFO search underway" https://tinyurl.com/ybnjzbv6
9-11-17: Roger J. Lias (ex-Allergan) to become Avid’s CEO and join PPHM’s BOD eff. 9-25-17 https://tinyurl.com/yddufw4s (left 5-8-19)
11-29-17: Tracy L. Kinjerski joins Avid as VP/Bus.Operations https://tinyurl.com/yc4zenkc (Left 1-8-2020)
Profiles of all 7 BOD members: http://ir.avidbio.com/corporate-governance/board-of-directors
...Joseph Carleone/Chair, Nicholas Green(CEO), Richard Hancock, Gregory Sargen, Mark Bamforth, Patrick Walsh, Catherine Mackey

10-24-17: Peregrine Adds Patrick D. Walsh to BOD, "An Industry Veteran with 30+yrs Experience Leading Successful CDMO Org’s” http://tinyurl.com/y3jo2rv7
10-19-17: Peregrine Adds Mark R. Bamforth to BOD, "10yrs/Genzyme; 30yrs of biologics leadership experience, incl. founding 2 CDMOs" http://tinyurl.com/y6ydcaof
ANALYST COVERAGE: http://ir.avidbio.com/analyst-coverage
Also see: https://www.marketbeat.com/stocks/NASDAQ/CDMO/price-target
Janney Montgomery Scott - Paul Knight PT=$10
H.C. Wainwright & Co. - Joseph Pantginis PT=$9
Craig-Hallum Capital Group - Matt G. Hewitt PT=$10
First Analysis Securities - Joseph Munda PT=$7.50
Stephens Inc. - Jacob Johnson PT=11 (10=>11 9-2-20)
FINANCIALS & BUSINESS DEV.:
2-3-21: Avid to Mfg. CV19 Therapeutic Lenzilumab for Humanigen https://tinyurl.com/1hzzksxa
8-26-20: Mapp Biopharmaceutical signs w/Avid for ClinDev of Antiviral Antibody (MBP091) https://tinyurl.com/yxwvr949 BARDA #HHSO100201900018C($16.5M+Opt/$30M), see: https://tinyurl.com/yye8t5nx
8-20-20: Oragenics signs Dev/Mfg Agreement with Avid for COVID-19 Vaccine “TerraCoV2” https://tinyurl.com/yxqg3w4v
8-6-20: Iovance Biotherapeutics signs w/Avid(+Aragen) to Dev+Mfg. IOV-3001 (IL-2 Analog) https://tinyurl.com/y2lgzh6x
7-28-20: Avid Teams with Argonaut to add “Parenteral Fill-Finish Services” https://tinyurl.com/y2lgzh6x
5-6-20: Avid Teams with Aragen to speed up drug dev./delivery timelines https://tinyurl.com/ycuu2m5r
4-23-20: Avid receives $4.4M PPP/CV19 Loan (can apply for forgiveness) 8K: https://tinyurl.com/yaltupxa
...5-12-20: Avid Pays Back $4.4M PPP/CV19 Loan (“New SBA Guidance/abundance of caution”) 8K: https://tinyurl.com/y993tkk6
3-10-20: CDMO's Revs & Burns By Qtr Table, FY07/Q1 thru FY20/Q3 (q/e 1-31-20): https://tinyurl.com/s9cmzmf
......Avid FY20 (fye 4-30-20) revenues guidance: $55-59M; committed backlog=$58mm at 1-31-2020.
7-8-19: Avid Signs New Top10 Global Pharma and Expands Existing Cust. Relationship https://tinyurl.com/yyq8zgb9
6-27-19: "The 5 new clients signed in late FY2018 contributed significantly to revenue diversification in FY2019." SEE CUST SPLITS for FY19: http://tinyurl.com/y5j4dlsv
4-24-18: ”In recent weeks, Avid has signed new agreements with 3 addl. undisclosed intl. drug dev. companies (that’s 4 thus far in CY2018)” https://tinyurl.com/y89whc8d
3-1-18: Acumen Pharm. selects Avid for Process Dev. & Mfg. of ACU193 (Alzheimer’s) https://tinyurl.com/y8jvwleq http://www.acumenpharm.com
2-21-18: Enzyvant selects Avid to commercially mfg. RVT-801 (Farber disease) https://tinyurl.com/yd5xhcx8 http://www.enzyvant.com
2-12-18: Peregrine’s Legacy PS-Targeting IP Sold to ONCOLOGIE INC. (Boston, CEO: Laura E. Benjamin) for $8M/upfront, $95M/milestones https://tinyurl.com/yam8gb3h
...NOTE: 2-13-18: Oncologie Licenses Mologen’s immunotherapeutic ‘lefitolimod’ (TLR9 agonist) https://tinyurl.com/y9z54f4x “ONCOLOGIE is backed by top-tier intl. investors and has the objective to dev. novel personalized medicines in the field of immuno-oncology."
2-28-17: Avid & Cook (acq. by Catalent 12-2017 for $950M; $179Msales x 5.3; EV/EBITDA=17.3) remain Halozyme's 2 CMO's (“working to scale-up/validate/qualify Avid II/Myford for Roche collab.”): http://tinyurl.com/h75teta
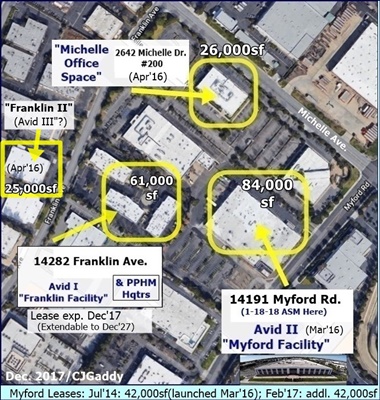
FACILITIES: https://avidbio.com/facilities/
10-8-19: Avid Announces Completion of Expansion of Process Dev. Capabilities & Labs https://tinyurl.com/y3hfwst9
...will “significantly accelerate Avid’s ability to drive efficient & rapid on-boarding of new cust. pgms progressing to mfg.”
4-24-18: Avid adding Process Dev. Labs, upgrading capabilities (in phases; total will be >6000sq; 1st one ready Q3CY18/Jul-Sep'18) https://tinyurl.com/y89whc8d
5-2017: Avid II (Myford) adds 2 MilliporeSigma Mobius 2,000L single-use bioreactors; total mfg. capacity now ">11,000L". http://tinyurl.com/ky7bmu4
5-10-17: Halozyme comments on Avid II(Myford) expansion in their 3-31-17/10Q pub. 5-9-17: http://tinyurl.com/mrl34uk
..."validation of the new facility is scheduled to end in Q2/2017… Once this new facility is approved, it will become the primary source for Roche of bulk rHuPH20.”
6-2-16: Corp.Update – Avid Expansion & Drug Development - http://tinyurl.com/zvmhqmr
3-7-16/Avid II: Formal Commissioning of Avid's New 40,000sq "Myford" Facility, “single-use/fully disposable” (potentially $40M addl revs) http://tinyurl.com/y5jmfpo3
12-10-15/Avid II: Avid Expansion into MYFORD Facility now GMP-run ready (potential +$40mm sales) - contemplating further expansion http://tinyurl.com/y539yut4
12-10-14: Avid to Double Mfg. Capacity(“Myford Expansion”) http://tinyurl.com/y2sqoy2u & http://tinyurl.com/kmdgq8t

PRESENTATIONS & ARTICLES: https://avidbio.com/events/
Sep17/Fireside Chat w/Nick Green: 18th Annual Morgan Stanley Global Healthcare Conf.: https://tinyurl.com/y4cbjjrc
5-18-20: Avid Listed In Top CMO Awards Article https://tinyurl.com/yb5cnh6m
...”Avid received 5 CMO Leadership Awards categories, incl. 2 Champion designations: Expertise & Service.”
10-25-18/BioPharma: Roger Lias Interview, “Avid expansion to Triple Process Dev. Capacity” https://tinyurl.com/ybtuk29m
Sept2018/BioTechWEEK: Tracy Kinjerski (VP/BusOP) interview: Avid’s differentiators (6mins.) https://www.youtube.com/watch?v=x3tzo4pZivk
1-29-18: NobleCon14 - 14th Annual Inv. Conf., Ft.Laud. => Roger Lias' webcast/slideshow https://tinyurl.com/yanwk9yo
11-15-17/Outsourcing-Pharma: “How This R&D Company (Peregrine) is Transitioning to a Pure-Play CDMO: ‘Opportunities Are Almost Endless’" - Recap of interviews with Steve King & Avid Pres. Roger Lias http://tinyurl.com/y7dv9faz
CONFERENCE CALLS & ASM's:
12-2-20: Qtly. Conf. Call (Green/Hart/Compton) PR & Transcript https://tinyurl.com/y66xk888
10-20-20 Annual Shareholders Meeting - Replay (9:45): https://www.virtualshareholdermeeting.com/CDMO2020 (Control# not needed)
9-1-20: Qtly. Conf. Call (Green/Hart/Compton) PR & Transcript https://tinyurl.com/y329llow
...CEO N.Green: ”My first few weeks have done nothing but confirm my view that I believe Avid to be a strong and state-of-the-art company with significant opportunity for growth."
6-30-20: Qtly. Conf. Call (Hancock/Hart/Compton) PR & Transcript https://tinyurl.com/y9zbgmos
...RickH: "Based on our customers' growing demand and our expanding business development activity, we believe that we will significantly increase capacity utilization in 2021 and beyond. Accordingly, we have entered into a new phase of planning for the expansion that will support our growing business in the years to come.”
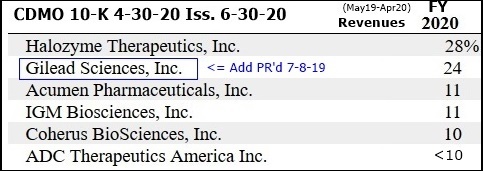
...”Top10 Pharma customer added 7-2019 named in 10-K: GILEAD SCIENCES.”
3-10-20: Qtly. Conf. Call (Hancock/Hart/Compton) PR & Transcript https://tinyurl.com/s9cmzmf
12-9-19: Qtly. Conf. Call (Hancock/Hart/Kinjerski) PR & Transcript https://tinyurl.com/tfewuzc
9-5-19: Qtly. Conf. Call (Hancock/Hart/Kinjerski) PR & Transcript https://tinyurl.com/y6t8zfur
6-27-19: Qtly. Conf. Call (Hancock/Hart/Kinjerski) PR & Transcript https://tinyurl.com/y6zdjyu2
...Rick Hancock, "Most importantly, during Q4, the Company achieved positive income, generating cash from dev. & mfg. operations for the 1st time since the beginning of the CDMO transition (Jan'18)."
3-11-19: Qtly. Conf. Call (Lias/Hart) PR & Transcript http://tinyurl.com/yy6dvjvc
12-3-18: Qtly. Conf. Call (Lias/Hart//Kinjerski) PR & Transcript https://tinyurl.com/y9n374kp
10-4-18: ASM/2018 (@Myford Facility) - Roger Lias’ Slideshow & Attendee Report https://tinyurl.com/yctfzhlb
9-10-18: Qtly. Conf. Call (Lias/Hart) PR & Transcript https://tinyurl.com/y8oc6hx8
...Roger Lias, "During the qtr, we advanced the projects of our existing active clients and continue to engage with numerous potential new customers."
7-16-18: Qtly. Conf. Call (Lias/Kinjerski) PR & Transcript https://tinyurl.com/yaozdggz
...Roger Lias, “In a short period, we’ve established a targeted business dev. operation that is actively providing visibility for Avid Bioservices within our fast growing but competitive marketplace.”
3-12-18: Qtly. Conf. Call (Lias/Kinjerski/Lytle) PR & Transcript https://tinyurl.com/yakdl4wj
...Roger Lias, ”I'm pleased to be able to report that in a very short period of time we've generated significant interest from both emerging & growth biopharmaceutical players and from pharmaceutical multinationals. I'm confident that the plan we're executing will drive a considerable increase in backlog and the opportunity to further enhance capacity utilization in the future.”
......From the 3-12-18 PR: “At present, we are in late-stage negotiations with several potential new customers and expect to announce the executed agreements before the end of the FY[4-30-18].”
1-18-18: ASM/2017 (@Myford Facility) - Roger Lias’ Slideshow & Attendee Reports https://tinyurl.com/yca6enbr 12-7-17 PROXY/14A: https://tinyurl.com/y7qprpg9
12-11-17: Qtly. Conf. Call (Lias/Lytle) Transcript https://tinyurl.com/ybycb2s6
...Roger Lias, "the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
9-11-17: Qtly. Conf. Call (King/Lias/Lytle) Transcript http://tinyurl.com/y9y8qdac
...Steve King: “For this reason, we have concluded that in order to best position Peregrine’s R&D assets for successful development, they should be advanced by a partner with the appropriate expertise and ample resources to invest in the necessary clinical trials. To that end, we have been working diligently towards the transformation of the overall business to becoming a pure-play CDMO, while assessing the best strategic options for the R&D assets that would allow stockholders to directly see the future value from their continued developments. By partnering & eliminating future R&D expenditures, we believe we are best positioning Avid for future growth. Through reinvestment & expansion, we believe we will attract new customers and extend current contracts that will help position Avid as a leading U.S. CDMO. We are moving forward expeditiously with strategic discussions as we recognize the need to move quickly both from the R&D & CDMO standpoints. We hope to bring this process to completion over the coming months and will update you on our progress.”
7-14-17: Qtly. Conf. Call (King/Shan/Lytle) Transcript http://tinyurl.com/yb4wulvu
...Steve King: “We are seriously considering the possibility of separating our 2 distinct businesses, Avid and R&D/PS-Targeting.”
3-13-17: Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
...Steve King: “We believe the recent improvement of stock price is a growing recognition of the value of Avid, and having the full value of the Avid business reflected in our stock price is a top priority.”
12-12-16: Qtly. Conf. Call (King/Shan/Hutchins/Lytle) Transcript http://tinyurl.com/hhn4gga
...Steve King: “Our goal is to bring the overall company to profitability within the next 18mos. We believe just the value of Avid Bioservices is far greater than our current market cap and is only growing in value."
10-13-16 ASM/2016: ATTENDEE Reports & Link to CEO Steve King's 35min/45slide webcast: http://tinyurl.com/jx7ouay
Feb. 2018: Peregrine Pharmaceuticals completed its Mid’17-Early’18 Transition to a Pure-Play CDMO
(Contract Development & Manufacturing Organization), Avid Bioservices, Inc. - CEO: Roger Lias.
**FULL PPHM=>CDMO Transition History (Ronin/SWIM): https://tinyurl.com/ybqvzwhg
1-8-17: Peregrine chgs. name to “Avid Bioservices, Inc.”; new ticker: “CDMO” (+CDMOP) https://tinyurl.com/y8vhjow4
2-12-18: Peregrine’s Legacy PS-Targeting IP Sold to ONCOLOGIE INC. (Boston, CEO: Laura E. Benjamin) for $8M/upfront, $95M/milestones https://tinyurl.com/yam8gb3h
...12-13-18/8-K: Overview of Oncologie sale: https://tinyurl.com/yab9c6cr
NOTE: “PS-targeting Exosome tech. not included; back to UTSW", see: https://tinyurl.com/yakdl4wj
Click here for an ARCHIVE of the History of Peregrine’s Anti-PS/Bavituximab Platform - MOA, Trials, and Activity over the years, from early 2000’s thru 2017, prior to being Sold to ONCOLOGIE:
https://tinyurl.com/y8pq4rhc
Of Interest (post Oncologie Sale):
Oncologie's website: https://oncologie.com/true-home-v2/ NEWS: https://oncologie.com/newsroom/
4-20-18/AACR’18: MSKCC(LudwigCC) Tweets about 2 WolchokLAB/”PPHM” Anti-PS Posters https://tinyurl.com/ycgjhvqa
4-26-18: New Bavi+Keytruda/LIVER Ph2 IST Trial, Sponsor=UTSW, Collab=MERCK https://tinyurl.com/y7fd9vdb
6-7-18: Oncologie Obtains $16.5M Seed Funding Led by Pivotal bioVenture Partners China Fund https://tinyurl.com/ybrrbgg7
6-7-18/BioCentury: Laura Benjamin states, “Oncologie plans to begin a trial mid-summer '18 evaluating Bavi to treat HCC and a P-O-C trial in Gastric cancer in 1Q19/2Q19.” https://tinyurl.com/ycb8r7sm
12-3-18: During q/e 10-31-18, Avid sold remaining legacy R&D asset, r84(anti-VEGF), to Oncologie for $1.0M upfront. Avid is eligible to receive up to an addl. $21M in dev/reg./comm. milestones, and low to mid-single digit royalties on net sales upon commercialization of products utilizing r84 https://tinyurl.com/y9n374kp
11-4-19/PharmaBoardroom: Interview with Oncologie CEO Laura Benjamin discussing the 2 ongoing Bavi Trials w/Keytruda (USA UK Taiwan S.Korea) https://tinyurl.com/ydf6zhsv
...Dr. Benjamin, "We have high expectations of bavituximab and have obtained the global rights for this compound."
...Ongoing Trial #1 (N=80): Open Label, Bavi+Keytruda Adv. Gastric/GEJ Cancer https://clinicaltrials.gov/ct2/show/NCT04099641
......1-17-2020 ASCO/GI Poster: https://oncologie.com/wp-content/uploads/2020/01/2020-01-17-ASGO-GI-poster-final-draft.pdf
...Ongoing Trial #2 (N=34 UTSW): Open Label, Bavi+Keytruda Adv. Hepatocellular Carcinoma https://clinicaltrials.gov/ct2/show/NCT03519997
6-11-20/Fiercebiotech: Oncologie Reels In $80M To Push Clinical Pgms/Build Pipeline https://tinyurl.com/yany8f34 & https://tinyurl.com/y8p9artk
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
