Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Yeah haha
What a squeeeze
Lol great minds think alike
Here we go:)
* * $ARDM Video Chart Analysis 9/21/17 * *
Link to Video- watch analysis by clicking HERE
Ask is so thinnnn
I really like the tight float
Could exblode
Almost 60 days for FDA response
Finaly Running
Aradigm Awarded NIH Grant to Investigate the Treatment of Pulmonary Non-Tuberculous Mycobacterial (PNTM) Infections with Linhaliq
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=1037303
LYTTON LAURENCE W has filed a new 13G, reporting 5.1% ownership in $ARDM - https://fintel.io/so/us/ardm and https://fintel.io/i/lytton-laurence-w
* * $ARDM Video Chart 05-18-17 * *
Link to Video - click here to watch the technical chart video
Bulls in full control last 2 days - wonder how long they can keep the higher lows
Thanks. I didn't cut my losses quick enough. At this point probably just best to wait it out.
I cut my losses on this one, but might be a great entry point. They got some bad data on treatment tests, people want profits "now" so it took a dump.
Good news is, I've listened to conference calls and read up a lot of DD. They just need a positive PR and this thing will POP!
Any thoughts? ARDM is at a 52 week low. Haven't seen any positive signs lately. Thanks.
Next ARDM update? I've done a lot of DD here and really like this company. Looks like some bad/skewed data in one of their trials. I have high hopes for them in the future, just a waiting game now.
Ah...but listen to the research presentation. The market misunderstood semantics. And, that's why First Eagle swooped in and bought more. The CEO basically implied that no one cares about minor lung events--the major ones are pains in the arse and cause the most damage and cost the most money. The inhaled antibiotics provided a buffer of an extra two months between these ICU events.
Also, the federal govt is in talks with $ARDM to protect our soldiers from bio warfare. Govt contracts are ALWAYS a good idea. And, with Trump in the WH, I foresee big spending in anything defense.
https://event.webcasts.com/starthere.jsp?ei=1130387
You can register as independent investor.
How old is the data of this article. The results of the two phase 3 studies have been posted for a month.
https://www.dvidshub.net/news/219852/changing-tide-antibiotic-resistant-infections
Pulmaquin is currently undergoing two Phase 3 clinical studies to determine its safety and effectiveness as an inhaled formulation for the treatment of patients with non-cystic fibrosis bronchiectasis, cystic fibrosis or non-tuberculous mycobacteria. However, Pulmaquin and an alternate formulation, Lipoquin, both demonstrated proof-of-concept efficacy in rodent models of inhalational tularemia, plague and Q-fever.
2 month reduction in SEVERE pulmonary events.
In other words, severe costs more money. Preventing them SAVES Money.
I listened to webcast and learned a lot.
Been sitting on sidelines for years. I joined in today. I sold Grifols and put $$$ in $ARDM.
https://event.webcasts.com/viewer/event.jsp?ei=1130387
Changing the Tide of Antibiotic-Resistant Infections
https://www.dvidshub.net/news/219852/changing-tide-antibiotic-resistant-infections
Does somebody know the reason?
Mayby this:
"We will review the next steps towards an application for approval of Pulmaquin in the U.S. at an upcoming meeting scheduled with FDA and are planning to engage soon in discussions of these results with the EMA as well," said Dr. Juergen Froehlich, MD, Aradigm's Chief Medical Officer.
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=1001671
Insider acquired 40900 shares (SEC Form 4)
The shares were purchased in multiple transactions at prices ranging from $2.21 to $2.32, inclusive.
https://www.sec.gov/Archives/edgar/data/1013238/000112329216003039/xslF345/edgar.xml
STATEMENT OF CHANGES IN BENEFICIAL OWNERSHIP
First Eagle Investment Management, LLC
https://www.sec.gov/Archives/edgar/data/1013238/000114036116088916/xslF345/doc1.xml
Aradigm to Present at the Stifel 2016 Healthcare Conference on November 16
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=998741
Multi-million Dollar DTRA Funding for Biodefense Research with Aradigm's Inhaled Ciprofloxacin
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=994073
Aradigm Announces Australian Research Council Funding to Develop Nanotechnologies Targeting Bacterial and Fungal Biofilms
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=992622
Aradigm Announces Last Patient Dosing Visit in ORBIT-3 and ORBIT-4 Phase 3 Studies of Pulmaquin in Non-Cystic Fibrosis Bronchiectasis Patients
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=990452
Aradigm (ARDM) Shares March Higher, Can It Continue?
http://www.nasdaq.com/article/aradigm-ardm-shares-march-higher-can-it-continue-cm682434
Aradigm to Host Analyst Meeting and Webcast on September 26 in New York City
HAYWARD, Calif. --(BUSINESS WIRE)-- Aradigm Corporation (Nasdaq: ARDM) (the "Company") announced today that it will host an Analyst Lunch on the topic of Non-Cystic Fibrosis Bronchiectasis (non-CF BE) on Monday, September 26, 2016 from 12:00-1:30 pm Eastern Time in New York, NY . The event will feature presentations by two key opinion leaders (KOLs): David E. Griffith , MD and Gregory Tino , MD.
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=990263
Will ARDM Bounce Last Just a Few Days?
Wait for release of Phase 3 (ORBIT 3 and 4) clinical trials results very soon.
See https://clinicaltrials.gov/ct2/show/NCT02104245?term=aradigm
bullish signal
Ownership Summary as reported in the most recent 13F filings
Read more: http://www.nasdaq.com/symbol/ardm/institutional-holdings#ixzz477VsWs9r
Today ARDM can fly over 6 $$ ( I think )
First Closing of Private Placement Offering of $23 Million Senior Convertible Notes Due 2021
http://ih.advfn.com/p.php?pid=nmona&article=71249128
Today will be a great day.
Fantastic chart and best news.
Private Placemente of 23.000.000 $ Senior Notes.
http://www.barchart.com/chart.php?sym=ARDM&t=BAR&size=M&v=1&g=1&p=WO&d=X&qb=1&style=technical&template=
Positive Stock Sentiment
http://www.standardtrib.com/aradigm-corp-nasdaqardm-positive-stock-sentiment.html
nice chart.
Ardm is ready for another stretch upward
http://stockcharts.com/freecharts/gallery.html?ARDM
Excelent news:
Aradigm Announces Decision for Centralized Review of Pulmaquin by the European Medicines Agency
Aradigm Corporation (Nasdaq:ARDM) (the "Company") today announced that the European Medicines Agency (EMA) has approved its request to review Pulmaquin®, Aradigm’s investigational inhaled liposomal ciprofloxacin product, under the Centralised Authorisation Procedure drug review process. This procedure results in a single marketing authorization that is valid in all 28 European Union countries, as well as three European Economic Area countries. Aradigm requested, and was granted, the centralized pathway on the basis that Pulmaquin represents a significant technical innovation for the potential treatment of non-cystic fibrosis bronchiectasis (non-CF BE) associated with chronic Pseudomonas aeruginosa (P. aeruginosa) infection.
“We are pleased that the EMA has agreed to review Pulmaquin through the centralized review process,” said Dr. Juergen Froehlich, Aradigm’s Chief Medical Officer. “This will enable us to streamline the process of gaining a license in all member states of Europe, with the opportunity of providing those living with non-CF BE and chronic lung infections with P. aeruginosa quicker access to Pulmaquin. Registration through the Centralized Procedure is typically reserved for products with significant therapeutic, scientific or technical innovation. This favorable decision reflects positively on the innovative approach we are taking investigating Pulmaquin for the treatment of this current unmet medical need in non-CF BE patients.”
Its going higher and higher but its still dirt cheap with a ridiculous market cap of $60 million .
“The completion of the enrollment of our pivotal Phase 3 clinical program for bronchiectasis last year was a critical achievement for Aradigm. All our efforts are now focused on having the key data analyses for both trials completed this year to enable us to file the US NDA early in 2017, followed by submission for market authorization in the European Union,” said Igor Gonda, CEO and President of Aradigm.
"Aradigm's Lipoquin and Pulmaquin inhalable antibiotics are potential best-in-class treatments designed for chronic use and to be effective at treating bacterial infections in patients' lungs, while also minimizing lung irritation that is common in cystic fibrosis and especially bronchiectasis patients," said Andrew I. McDonald, Ph.D., Founding Partner at LifeSci Advisors. "The recent partnership with Grifols provides adequate funding to bring the program through Phase III trials in bronchiectasis and may allow Aradigm to pursue additional indications."
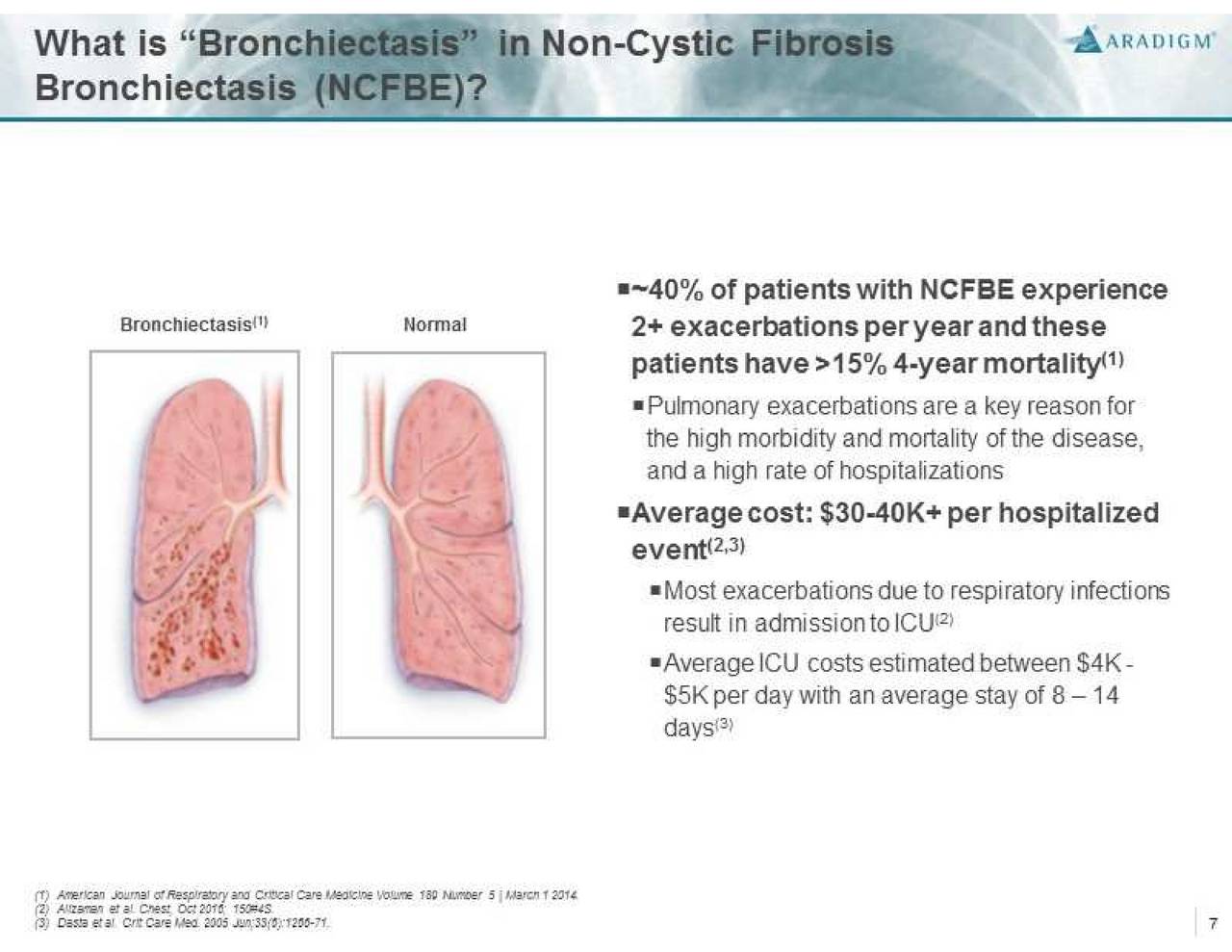
About Non-Cystic Fibrosis Bronchiectasis
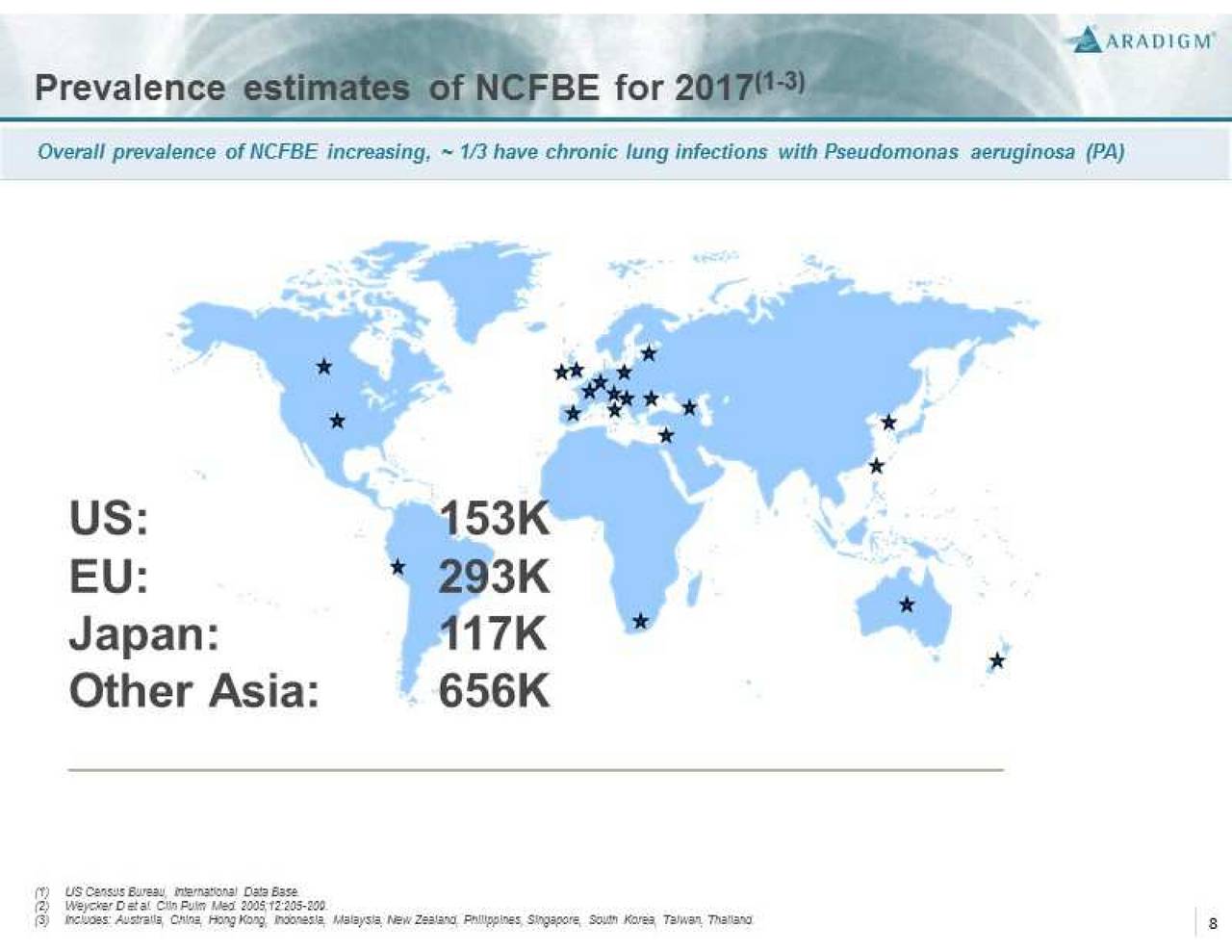
Non-CF BE is a severe, chronic and rare disease characterized by abnormal dilatation of the bronchi and bronchioles, frequently associated with chronic lung infections. It is often a consequence of a vicious cycle of inflammation, recurrent lung infections, and bronchial wall damage. Non-CF BE represents an unmet medical need with high morbidity and mortality that affects more than 110,000 people in the U.S. and over 200,000 people in Europe. There is currently no drug approved for the treatment of this condition.
They have a long history of failure to make it to market, RS splits and insiders stock manipulation.
Hopefully this time they will succeed.
Undiscovered ARDM is one of the cheapest Biotechs out there which has MONSTER Upside Potential . If Pulmaquin is successful in Phase 3 in 2H 2016 then this low float rocket has the Potential to move Close to $100 per Share .Pulmaquin has QIDP,Orphan Drug and Fast Track Status and there is no approved Drug for Bronchiectasis .
Market Cap: $47 Million
Cash Balance : $39.9 Million
Shares Out : 14.73 Million
Pipeline
http://www.aradigm.com/products_pipeline.html
Aradigm Corporation Completes Enrollment in the Second Phase III Study of Pulmaquin in Non-Cystic Fibrosis Bronchiectasis (ORBIT-3)
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=936283
Aradigm Corporation Completes Enrollment in Phase III Study of Pulmaquin in Non-Cystic Fibrosis Bronchiectasis (ORBIT-4)
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=932534
Grifols and Aradigm Corporation Complete the Closing of Worldwide Licensing Agreement to Develop and Commercialize Pulmaquin®
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=787555
Full Analysis
https://www.baystreet.ca/articles/research_reports/lifesci/266497b2-3c17-408a-91b8-f40e7d3ad3cf.pdf
Licensing Agreement with Grifols Validates Aradigm’s Inhaled Antibiotic Approach. In May 2013, Aradigm announced an exclusive worldwide licensing agreement with Grifols (NasdaqGS:GRFS) covering both of Aradigm’s proprietary, liposomal formulations of ciprofloxacin, Lipoquin and Pulmaquin.The deal provides up to $65 million for developmental and clinical expenses related to the BE indication. Aradigm can receive an additional $25 million in development milestones. Grifols is responsible for commercialization upon FDA and EU approvals. Aradigm will receive tiered royalties on worldwide sales of 12.5% on the first $300 million annually, and increasing to 20% above that. Aradigm will also receive a transfer price mark-up on the supplied product. Grifols also received an option to license Aradigm’s AERx® pulmonary drug delivery platform for another drug, and made a $26 million investment in Aradigm. Grifols is a Spanish multinational pharmaceutical company with over $5 billion in sales for 2012, and almost 75 years of experience selling, promoting, and distributing healthcare products. Grifols is an ideal partner with experience in respiratory indications in the US, and has a successful 25 year track record selling Prolastin-C for patients with emphysema due to deficiency of alpha1-proteinase inhibitor. Grifols, along with existing and new investors, also led a $41.4 million round of financing that greatly extends Aradigm’s cash runway, and will allow the Company to focus on the execution of its clinical trials for BE and other indications.
Pulmaquin's Potential
Bronchiectasis is observed in patients with cystic fibrosis on a frequent basis. However, it is less common in patients without cystic fibrosis. According to Aradigm, non-cystic fibrosis bronchiectasis affects more than 110,000 people in the U.S. As a result, Pulmaquin was granted orphan drug designation by the FDA in June 2011. The orphan drug status means that if approved, Pulmaquin can command a higher price. Also, in the U.S., there are no drugs approved right now for the treatment of bronchiectasis. The incidence of bronchiectasis is growing at 9% a year in the U.S. In Europe, the disease affects 210,000 patients. According to Igor Gonda, president and CEO of Aradigm, Pulmaquin for bronchiectasis is a billion-dollar plus opportunity globally.
Aradigm to Present at the Ladenburg Thalmann 2015 Healthcare Conference on September 29
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=932731
Aradigm Corporation Completes Enrollment in Phase III Study of Pulmaquin in Non-Cystic Fibrosis Bronchiectasis (ORBIT-4)
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=932534
Results from Pulmonary Non-Tuberculous Mycobacteria (PNTM) Study Using Aradigm's Liposomal Ciprofloxacin to Be Presented at ICAAC/ICC 2015
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=930955
Brokerages Set $26.00 Price Target for Aradigm (NASDAQ:ARDM)
http://www.dakotafinancialnews.com/brokerages-set-26-00-price-target-for-aradigm-nasdaqardm/403364/
Phase 3 Study With Dual Release Ciprofloxacin for Inhalation in Non-CF Bronchiectasis (ORBIT-4) is ongoing, but not recruiting participants.
Last updated: August 12, 2015
https://clinicaltrials.gov/ct2/results?term=aradigm
Golden Cross
Golden Croos THIS WEEK !!
Only the volume is bad but the free float is very short.
Back in,trading activity looks interesting.hopefully we'll get some updates soon. Look at AXON,similar float size,was 4.00 six months ago. GL
Prices below $7 per share was great opportunity to buy.
https://clinicaltrials.gov/ct2/show/NCT02104245?term=Aradigm&rank=1
https://clinicaltrials.gov/ct2/show/NCT01515007?term=Aradigm&rank=2
|
Followers
|
41
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1434
|
|
Created
|
11/23/04
|
Type
|
Free
|
| Moderators | |||





NEW YORK, NY--(Marketwired - Oct 29, 2013)
"Aradigm's Lipoquin and Pulmaquin inhalable antibiotics are potential best-in-class treatments designed for chronic use and to be effective at treating bacterial infections in patients' lungs, while also minimizing lung irritation that is common in cystic fibrosis and especially bronchiectasis patients," said Andrew I. McDonald, Ph.D., Founding Partner at LifeSci Advisors. "The recent partnership with Grifols provides adequate funding to bring the program through Phase III trials in bronchiectasis and may allow Aradigm to pursue additional indications."
http://www.marketwired.com/press-release/lifesci-advisors-initiates-coverage-of-aradigm-corporation-otcbb-ardm-1845844.htm
Phase III Program in Bronchiectasis Planned to Launch in 2014; Recent Partnership With Grifols Worth Up to $90 Million Plus Tiered Royalties;
ALBANY, New York, February 25, 2014 /PRNewswire/
Pulmonary Drug Delivery Systems Market Expected to Reach USD 28.7 Billion Globally in 2019: Transparency Market Research
According to a new market report published by Transparency Market Research "Pulmonary Drug Delivery Systems Market(Products- Metered Dose Inhalers, Dry Powder Inhalers and Nebulizers; Applications- Asthma, COPD and Cystic Fibrosis) - Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2013 - 2019," the global pulmonary drug delivery systems market was valued at USD 21.03billion in 2012 and is expected to grow at a CAGR of4.5% from 2013 to 2019, to reach an estimated value of USD 28.70billion in 2019.
http://www.prnewswire.com/news-releases/pulmonary-drug-delivery-systems-market-expected-to-reach-usd-287-billion-globally-in-2019-transparency-market-research-247046411.html
HYATTSVILLE, Maryland Tue Apr 1, 2014 7:49pm EDT
U.S. FDA advisers back MannKind's inhaled diabetes drug Afrezza
http://www.reuters.com/article/2014/04/01/us-mannkind-fda-diabetes-idUSBREA301Z620140401
ARDM has huge potential with AERx iDMS (inhalable insulin) right now.
The drug was in the last lap of clinical trials before halting by Novo Nordisk in past.
http://www.aradigm.com/products_iDMS.html
http://www.aradigm.com/products_1700.html
https://www.visiongain.com/Report/1252/Drug-Delivery-Technologies-Market-Forecast-2014-2024
Sept 22, 2017
Aradigm to Present at the Ladenburg Thalmann 2017 Healthcare Conference on September 26
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=1041356
https://seekingalpha.com/article/4109924-aradigm-corporation-ardm-presents-ladenburg-thalmann-2017-healthcare-conference-slideshow
Sept 27, 2017
Aradigm Announces FDA Acceptance of NDA for Linhaliq with Priority Review Status
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=1041475
Oct 10, 2017
Aradigm Regains Compliance with NASDAQ Listing Requirements
http://investor.aradigm.com/releasedetail.cfm?ReleaseID=1043285
SEC Company Filings - Aradigm Corporation
https://searchwww.sec.gov/EDGARFSClient/jsp/EDGAR_MainAccess.jsp?search_text=*&sort=Date&formType=1&isAdv=true&stemming=true&numResults=10&queryCo=Aradigm&numResults=10
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
