Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=152943733
BXDX Meloxicam IV formulation incorporates ALKS Nanocrystal technology
Bravo FDA ... challenging efficacy not just rubber stamping ALKS new drug idea to market another bottle of miracle cure... looking at ALL phase 3 not just the ones that showed statistically significant results that the company said validated their drug... companies are notorious for choosing the studies that validate their thesis and dropping the ones that don't...oh and the CEO's breathless comment on the CC call yesterday that there have been no pharmacological advances for major depression for the past 30 years as a reason for the FDA to rush another drug to market belies the fact that studies have shown that medication and CBT therapy are equally effective after 6 months but that CBT effectiveness lasts years after completion while medication effectiveness diminishes rapidly after the medication is stopped .. as for pharmacological interventions.. take a look at this link form UCSD on alternatives ... https://health.ucsd.edu/news/features/Pages/2018-01-03-q-and-a-ketamine-for-depression.aspx
ALKS: FDA RTF letter
https://ih.advfn.com/p.php?pid=nmona&article=77078502
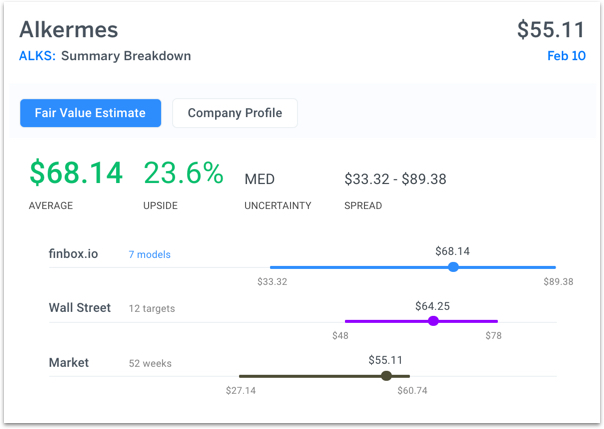
Alkermes is expected to report on Wednesday and seven separate valuation analyses imply a fair value of $68.14, slightly above Wall Street's target:
Six Healthcare Stocks with Nice Upside Potential
Here’s Our Take On Alkermes’s Latest Hit
http://marketexclusive.com/heres-take-alkermes-plc-nasdaqalkss-latest-hit/34712/?icd1
ALKS: We have held ALKS shares since 6/21/2002, current basis of $16.24, closed yesterday @ $43.51.
AVXL: With AVXL, we have held 5x shares of ALKS since 4/23/2015, split-adjusted basis of $1.80, closed yesterday @ $3.36.
There is a market...neither has penetrated. I speculate there will be room for both medications, when and if FDA approves both. I do not know[have an informed opinion] whether AVXL has "a better depression drug."
AVXL has a better depression drug and working both BIIB and Michael J Fox Foundation.
http://www.businesswire.com/news/home/20161020006475/en/
AH +, very active.
Will this Crack $50 if Hilliary & Co get in the WH?
Municipalities now supporting Vivitrol at there narcan training.
Check out this link from Nassau county on Long Island. http://www.nassaucountyny.gov/shotatlife Way to go Nassau if every county in the country followed you we might end this addiction .
I'm happy that it closed green. Hopefully Monday we keep going up from here. This low pps is unbelievable All I can say buy the fear and sell the hype.
ALKS price cut from 65 to 36 $ : UBS posted on Yahoo >>>
http://finance.yahoo.com/marketpulse/ALKS
I didnt think it would break under $35 maybe $30 is the Bottom before it starts bouncing?
Volume is insane!
Hopefully it opens a few dollars up and go from there back up.
Do you it keeps going down tomorrow?
Brutal today. Hopefully it starts to go back up too recover some of the losses. It's crazy looking at the portfolio today.
$33.65 GETTING PUNISHED EVEN MORE....
ALKS
NLOD 35.82
ALKS
$ALKS recent news/filings
bullish 72.96
breakout soon
## source: finance.yahoo.com
Tue, 15 Dec 2015 16:20:10 GMT ~ Massachusetts biotechs behind one-in-six drugs approved by FDA this year
read full: http://www.bizjournals.com/boston/blog/bioflash/2015/12/massachusetts-biotechs-behind-one-in-six-drugs.html?ana=yahoo
*********************************************************
Tue, 08 Dec 2015 18:16:40 GMT ~ ALKERMES PLC. Financials
read full: http://finance.yahoo.com/q/is?s=alks
*********************************************************
Wed, 02 Dec 2015 13:59:59 GMT ~ Is Alkermes Plc (ALKS) A Good Stock To Buy?
read full: http://www.insidermonkey.com/blog/is-alkermes-plc-alks-a-good-stock-to-buy-397634/
*********************************************************
Mon, 16 Nov 2015 19:45:00 GMT ~ No Snake Oil in These Four Pharmaceutical Stocks
read full: http://www.thestreet.com/story/13367430/1/no-snake-oil-in-these-4-pharmaceutical-picks.html?puc=yahoo&cm_ven=YAHOO
*********************************************************
Wed, 11 Nov 2015 21:06:07 GMT ~ Alkermes’ Corporate Presentation to be Webcast at the Jefferies 2015 London Healthcare Conference
[at noodls] - DUBLIN--(BUSINESS WIRE)--Nov. 11, 2015-- Alkermes plc (NASDAQ: ALKS) announced today that its corporate presentation will be webcast live at the Jefferies 2015 London Healthcare Conference on Wednesday, ...
read full: http://www.noodls.com/view/67CE9D3B788BD7C750C34564513763E937631DA5
*********************************************************
$ALKS charts
basic chart ## source: stockcharts.com
basic chart ## source: stockscores.com
big daily chart ## source: stockcharts.com
big weekly chart ## source: stockcharts.com
$ALKS company information
## source: otcmarkets.com
Link: http://www.otcmarkets.com/stock/ALKS/company-info
Ticker: $ALKS
OTC Market Place: Not Available
CIK code: 0000874663
Company name: Alkermes plc
Incorporated In: PA, USA
$ALKS share structure
## source: otcmarkets.com
Market Value: $10,748,658,480 a/o Dec 14, 2015
Shares Outstanding: 150,058,055 a/o Oct 26, 2015
Float: Not Available
Authorized Shares: Not Available
Par Value: 0.01
$ALKS extra dd links
Company name: Alkermes plc
## STOCK DETAILS ##
After Hours Quote (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/after-hours
Option Chain (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/option-chain
Historical Prices (yahoo.com): http://finance.yahoo.com/q/hp?s=ALKS+Historical+Prices
Company Profile (yahoo.com): http://finance.yahoo.com/q/pr?s=ALKS+Profile
Industry (yahoo.com): http://finance.yahoo.com/q/in?s=ALKS+Industry
## COMPANY NEWS ##
Market Stream (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/stream
Latest news (otcmarkets.com): http://www.otcmarkets.com/stock/ALKS/news - http://finance.yahoo.com/q/h?s=ALKS+Headlines
## STOCK ANALYSIS ##
Analyst Research (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/analyst-research
Guru Analysis (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/guru-analysis
Stock Report (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/stock-report
Competitors (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/competitors
Stock Consultant (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/stock-consultant
Stock Comparison (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/stock-comparison
Investopedia (investopedia.com): http://www.investopedia.com/markets/stocks/ALKS/?wa=0
Research Reports (otcmarkets.com): http://www.otcmarkets.com/stock/ALKS/research
Basic Tech. Analysis (yahoo.com): http://finance.yahoo.com/q/ta?s=ALKS+Basic+Tech.+Analysis
Barchart (barchart.com): http://www.barchart.com/quotes/stocks/ALKS
DTCC (dtcc.com): http://search2.dtcc.com/?q=Alkermes+plc&x=10&y=8&sp_p=all&sp_f=ISO-8859-1
Spoke company information (spoke.com): http://www.spoke.com/search?utf8=%E2%9C%93&q=Alkermes+plc
Corporation WIKI (corporationwiki.com): http://www.corporationwiki.com/search/results?term=Alkermes+plc&x=0&y=0
## FUNDAMENTALS ##
Call Transcripts (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/call-transcripts
Annual Report (companyspotlight.com): http://www.companyspotlight.com/library/companies/keyword/ALKS
Income Statement (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/financials?query=income-statement
Revenue/EPS (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/revenue-eps
SEC Filings (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/sec-filings
Edgar filings (sec.gov): http://www.sec.gov/cgi-bin/browse-edgar?action=getcompany&CIK=0000874663&owner=exclude&count=40
Latest filings (otcmarkets.com): http://www.otcmarkets.com/stock/ALKS/filings
Latest financials (otcmarkets.com): http://www.otcmarkets.com/stock/ALKS/financials
Short Interest (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/short-interest
Dividend History (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/dividend-history
RegSho (regsho.com): http://www.regsho.com/tools/symbol_stats.php?sym=ALKS&search=search
OTC Short Report (otcshortreport.com): http://otcshortreport.com/index.php?index=ALKS
Short Sales (otcmarkets.com): http://www.otcmarkets.com/stock/ALKS/short-sales
Key Statistics (yahoo.com): http://finance.yahoo.com/q/ks?s=ALKS+Key+Statistics
Insider Roster (yahoo.com): http://finance.yahoo.com/q/ir?s=ALKS+Insider+Roster
Income Statement (yahoo.com): http://finance.yahoo.com/q/is?s=ALKS
Balance Sheet (yahoo.com): http://finance.yahoo.com/q/bs?s=ALKS
Cash Flow (yahoo.com): http://finance.yahoo.com/q/cf?s=ALKS+Cash+Flow&annual
## HOLDINGS ##
Major holdings (cnbc.com): http://data.cnbc.com/quotes/ALKS/tab/8.1
Insider transactions (yahoo.com): http://finance.yahoo.com/q/it?s=ALKS+Insider+Transactions
Insider transactions (secform4.com): http://www.secform4.com/insider-trading/ALKS.htm
Insider transactions (insidercrow.com): http://www.insidercow.com/history/company.jsp?company=ALKS
Ownership Summary (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/ownership-summary
Institutional Holdings (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/institutional-holdings
Insiders (SEC Form 4) (nasdaq.com): http://www.nasdaq.com/symbol/ALKS/insider-trades
Insider Disclosure (otcmarkets.com): http://www.otcmarkets.com/stock/ALKS/insider-transactions
## SOCIAL MEDIA AND OTHER VARIOUS SOURCES ##
PST (pennystocktweets.com): http://www.pennystocktweets.com/stocks/profile/ALKS
Market Watch (marketwatch.com): http://www.marketwatch.com/investing/stock/ALKS
Bloomberg (bloomberg.com): http://www.bloomberg.com/quote/ALKS:US
Morningstar (morningstar.com): http://quotes.morningstar.com/stock/s?t=ALKS
Bussinessweek (businessweek.com): http://investing.businessweek.com/research/stocks/snapshot/snapshot_article.asp?ticker=ALKS
$ALKS DD Notes ~ http://www.ddnotesmaker.com/ALKS
Funny for me to come upon your months ago post ...
Yes I follow the board, I am as well invested and hold quite a bit of ALKS.
Today is NOT a good day at all.!!!
very scary...!??
Anybody follow this board? Alkermes is my favorite company and I think it could be nice to get a discussion going. I have a large options position and am thinking about buying some stock as well.
Irish address and promising pipeline make Alkermes an attractive buyout target.
http://lastfinancier.com/amarin-amrn-and-alkermes-alks-tax-inversion-targets/
7:03AM Alkermes announces notice of allowance of U.S. Patent for a novel fumarate prodrug for Multiple Sclerosis; expects to file an Investigational New Drug (IND) application in mid-2014 (ALKS) 40.07 : Co announced that the United States Patent and Trademark Office (USPTO) has issued a Notice of Allowance for U.S. Patent Application 14/032,736, entitled "Prodrugs of fumarates and their use in treating various diseases."
The allowed composition of matter claims will cover Alkermes' MMF prodrug, ALKS 8700, a proprietary, small-molecule prodrug of monomethyl fumarate (MMF) for the treatment of multiple sclerosis.
The Notice of Allowance resulted from a prioritized examination granted by the USPTO, and the allowed claims are directed toward a novel MMF prodrug that is designed to rapidly and efficiently convert to MMF in the body.
Alkermes expects to file an Investigational New Drug (IND) application with the U.S. Food and Drug Administration (FDA) and initiate a phase 1 study of ALKS 8700 in mid 2014.
7:05AM Alkermes receives Fast Track Designation for ALKS 5461 for major depressive disorder (ALKS) 30.17 : Co announced that the FDA has granted Fast Track status for ALKS 5461 for the adjunctive treatment of major depressive disorder (MDD) in patients with an inadequate response to standard therapies. Fast Track is a process designed to facilitate the development, and expedite the review of drugs to treat serious conditions and with the potential to address an unmet medical need.
7:03AM Alkermes announces alignment with FDA on plans for pivotal program for ALKS 5461 for Major Depressive Disorder; program to begin in early 2014 (ALKS) 30.29 : Co has successfully completed its End-of-Phase 2 interactions with the U.S. Food and Drug Administration (FDA), and the company plans to advance ALKS 5461 into phase 3 development in early 2014. Alkermes is developing ALKS 5461 for the treatment of patients with major depressive disorder (MDD) who have inadequate response to standard therapies. The company and the FDA agreed on key elements of the development program, including preclinical and clinical requirements for the New Drug Application, the confirmatory study plans, the incorporation of innovative study designs that include the use of sequential parallel comparison design (SPCD), the primary endpoint and the statistical methodology. In September 2013, in advance of a planned End-of-Phase 2 meeting, Alkermes submitted a written briefing document detailing design elements of the proposed development program. The FDA's written responses aligned with the company's proposals such that the End-of-Phase 2 meeting was deemed unnecessary.
7:16AM On The Wires (WIRES) :Alkermes (ALKS) announced completion of patient enrollment in the pivotal, multinational phase 3 study evaluating aripiprazole lauroxil in patients with schizophrenia. Co continues to expect topline results from the phase 3 study in 1H2014. Interim analysis of sample size indicated that sample size of 540 patients or more would have sufficient statistical power to evaluate the primary endpoint.
7:09AM Alkermes beats by $0.08, beats on revs (ALKS) 29.17 : Reports Q1 (Jun) earnings of $0.30 per share, $0.08 better than the Capital IQ Consensus Estimate of $0.22; revenues fell 8.9% year/year to $138.6 mln vs the $134.29 mln consensus.
The company reiterated its financial expectations for the nine-month period ending Dec. 31, 2013, which were originally provided on May 23, 2013.
Oh. That explains the drop in pps. Geeeeezzzz
7:00AM Alkermes presents positive results from phase 2 clinical study of ALKS 5461 in major depressive disorder at 53rd Annual NCDEU Meeting; ALKS 5461 met its primary endpoint (ALKS) 33.58 : Co announced the presentation of positive phase 2 data for ALKS 5461, a novel opioid modulator, in patients with major depressive disorder and inadequate response to standard therapies. In the phase 2 study, ALKS 5461 met its primary endpoint, met key secondary endpoints and demonstrated significant reduction in depressive symptoms versus placebo. The study is being presented in an oral session at the 53rd Annual New Clinical Drug Evaluation Unit Meeting in Hollywood, Fla., by Maurizio Fava, M.D., of Massachusetts General Hospital and Harvard Medical School.
The phase 2 study of ALKS 5461 utilized a sequential parallel comparison design, designed to reduce the impact of clinically meaningful response to treatment with placebo. The study included two four-week, randomized, double-blind stages run in sequence: an Initial Study Stage and a Successive Study Stage. The Successive Study Stage randomized only those patients who were non-responders to placebo in the Initial Study Stage. Both stages of the phase 2 study evaluated two doses of ALKS 5461, a lower dose and a higher dose.
In the phase 2 results for the overall study population, including both the Initial Study Stage and the Successive Study Stage, patients who received either dose of ALKS 5461 for a treatment period of four weeks showed a significant reduction in depressive symptoms from baseline in HAM-D17 and MADRS scores, compared to placebo. The primary endpoint of the phase 2 study was the change from baseline in depressive symptoms over a four-week treatment period in the overall study population, as measured by HAM-D17, compared to placebo. Data from the study showed that ALKS 5461 was generally well tolerated. The most common adverse events observed in the study were nausea, headache and dizziness.
7:01AM Alkermes presents positive results from Phase 1 study of ALKS 3831 as a broad spectrum treatment for Schizophrenia; difference between the ALKS 3831 treatment group and the control group receiving olanzapine alone was statistically significant (ALKS) 32.84 : Co presented positive results from a phase 1 study of ALKS 3831, a novel drug candidate for the treatment of schizophrenia, in an oral session at the 53rd Annual New Clinical Drug Evaluation Unit Meeting in Hollywood, Fla. By combining a novel opioid modulator, ALKS 33, and olanzapine, an antipsychotic drug commercially available as ZYPREXA?, ALKS 3831 is designed to be a broad spectrum treatment for schizophrenia with the potential benefits of reduced weight gain associated with olanzapine and expanded utility in patients with schizophrenia and comorbid substance abuse.
The phase 1, multicenter, randomized, double-blind, placebo- and active-controlled study was designed to compare the mean change from baseline in body weight following three weeks of oral administration of ALKS 3831 in a study that included 106 healthy, normal-weight male volunteers. The safety and tolerability results for ALKS 3831 were overall similar to those observed with the olanzapine-only treatment group. Healthy volunteers who received ALKS 3831 gained an average of 2.5 kg (5.5 lbs), while subjects who received olanzapine alone gained an average of 3.4 kg. The difference between the ALKS 3831 treatment group and the control group receiving olanzapine alone was statistically significant over the three-week study period, with a trend indicating the potential for even greater differentiation over longer study periods.
Based on the positive results of the phase 1 study, Alkermes plans to initiate a phase 2 dose-ranging study of ALKS 3831 in mid calendar 2013.
Has anyone looked at CEYY as an alternative? Better results for treating alcohol and a penny stock as opposed to $22/ share :P
I have ALKS on my radar. This company appears to be getting lots of attention. And nobody here to post about it!
7:11AM Alkermes beats by $0.07, beats on revs; raises FY13 EPS, rev guidance above consensus (ALKS) 18.52 : Reports Q2 (Sep) earnings of $0.17 per share, $0.07 better than the Capital IQ Consensus Estimate of $0.10; revenues rose 72.2% year/year to $124 mln vs the $122.49 mln consensus.
Co raises guidance for FY13, sees EPS of $0.88-1.02 vs. $0.77 Capital IQ Consensus Estimate; sees FY13 revs of $510-540 mln vs. $519.09 mln Capital IQ Consensus Estimate.
"In September, we updated our expectations for improved non-GAAP net income and free cash flow for fiscal 2013, reflecting the impact of the successful refinancing of the term loans used to fund the merger. Today, we are further improving our financial expectations for fiscal 2013 based on strong operational performance in the first six months, and we now expect Alkermes to generate between $120 million and $140 million in non-GAAP net income this fiscal year."
Alkermes, Plc (ALKS)
ZacksBy Zacks Equity Research | Zacks – 8 hours ago
We are upgrading Alkermes Plc (ALKS) to Outperform following the upwardly revised adjusted earnings guidance provided by the company for fiscal 2013. The guidance was boosted following the successful completion of the refinancing of Alkermes' previously outstanding senior secured bank debt.
Alkermes has been performing well since the purchase of Elan's EDT unit last year, thanks to the expanded portfolio. Moreover, the US approval of type II diabetes drug, Bydureon, is a major positive for Alkermes as the drug offers significant commercial potential. We are also pleased by the pipeline progress at Alkermes.
We believe that there is significant scope for stock price appreciation from current levels. Our price target of $24.00 is based on 6.1x our fiscal 2013 revenue estimate.
7:12AM Alkermes beats by $0.23, beats on revs; reaffirms FY13 EPS guidance, revs guidance (ALKS) 18.79 : Reports Q1 (Jun) earnings of $0.39 per share, excluding non-recurring items, $0.23 better than the Capital IQ Consensus Estimate of $0.16; revenues rose 145.9% year/year to $152.2 mln vs the $126.69 mln consensus. Co reaffirms guidance for FY13, sees EPS of $0.62-0.77 vs. $0.70 Capital IQ Consensus Estimate; sees FY13 revs of $490-530 mln vs. $510.74 mln Capital IQ Consensus Estimate.
7:22AM On The Wires (WIRES) Alkermes (ALKS) announced that Janssen Research & Development has initiated a phase 3 clinical research program for a three-month formulation of INVEGA SUSTENNA, an injectable medicine for the treatment of schizophrenia.
12:55AM Alkermes and Amylin Pharmaceuticals (AMLN) reports BYDUREON associated with significant and sustained glycemic control at four years (ALKS) 15.39 : Cos announce results from the long-term extension of the DURATION-1 study, which showed that BYDUREON, the first and only once-weekly treatment for type 2 diabetes, was associated with clinically significant and sustained improvements in glycemic control during four years of treatment in adults with type 2 diabetes. In the study, patients completing four years of BYDUREON treatment experienced clinically significant improvements in A1C (1.7 percentage points) and fasting plasma glucose from baseline. A1C is a measure of average blood sugar over three months. Although BYDUREON is not indicated for weight loss, patients treated with BYDUREON also lost an average of 5.5 pounds from baseline. Patients treated with BYDUREON also experienced statistically significant reductions in certain cardiovascular risk markers, including systolic blood pressure, total cholesterol, LDL cholesterol and triglycerides (-13%). No unexpected safety findings were observed with long-term BYDUREON therapy, and incidence of mild nausea, the most common adverse event during the controlled portion of the study, decreased over time.
4:01PM Alkermes announces ALKS 37 product profile did not satisfy their pre-specified criteria for advancing into phase 3 clinical trials (ALKS) 16.06 -0.52 : Co announces topline results from a phase 2b dose-ranging clinical study of ALKS 37 in the treatment of opioid-induced constipation. The multicenter, randomized, double-blind, placebo-controlled, repeat-dose study was designed to assess the safety, tolerability, pharmacokinetic profile and efficacy of ALKS 37 in approximately 150 patients. ALKS 37 was generally well tolerated at all dose levels, and while subjects taking ALKS 37 demonstrated an increase in bowel movements compared to baseline, the product profile did not satisfy pre-specified criteria for advancing into phase 3 clinical trials. Based on this evaluation, Alkermes has decided not to advance ALKS 37 and will consider out-licensing opportunities. "We had predetermined the product profile we needed to observe in order to continue to advance ALKS 37 into phase 3 clinical studies. Based on the results of this study, we will focus our future clinical development efforts on our other development programs, including ALKS 9070 for schizophrenia and ALKS 5461 for major depressive disorder, we will continue to maintain a disciplined approach to R&D and focus our resources on clinical candidates that show the most promise." A second phase 2b study of ALKS 37 for the treatment of opioid-induced constipation is concluding, and no additional clinical studies for ALKS 37 are planned.
Alkermes to Host Conference Call to Discuss Financial Results for Fiscal Year 2012
Business WirePress Release: Alkermes plc – Thu, May 10, 2012 4:00 PM EDT
DUBLIN--(BUSINESS WIRE)--
Alkermes plc (NASDAQ: ALKS - News) will host a conference call at 8:30 a.m. EDT (1:30 p.m. BST) on Thursday, May 17, 2012, to discuss the company’s financial results for its fiscal year ended March 31, 2012. Management will also provide financial expectations for fiscal year 2013.
The conference call will be webcast on the Investors section of Alkermes’ website at www.alkermes.com or may be accessed by dialing +1 888 424 8151 for U.S. callers and +1 847 585 4422 for international callers. The conference call ID number is 6037988.
A replay of the conference call will be available from 11:30 a.m. EDT (4:30 p.m. BST) on Thursday, May 17, 2012, through 5:00 p.m. EDT (10:00 p.m. BST) on Thursday, May 24, 2012, and may be accessed by visiting Alkermes’ website or by dialing +1 888 843 7419 for U.S. callers and +1 630 652 3042 for international callers. The replay access code is 6037988.
9:19AM Alkermes prices secondary offering of 21 mln ordinary shares held by Elan (ELN) at a price to the public of $16.50/share (ALKS) 17.30 :
|
Followers
|
9
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
176
|
|
Created
|
05/09/08
|
Type
|
Free
|
| Moderators | |||
http://www.alkermes.com/
http://finance.yahoo.com/q/ks?s=ALKS
http://www.form4oracle.com/company?cik=0000874663&ticker=alks
Alkermes, Inc., a biotechnology company, develops products based on proprietary, extended-release and pulmonary drug delivery technologies. It has two commercial products that include RISPERDAL CONSTA, a long-acting atypical antipsychotic medication approved for use in schizophrenia, marketed worldwide by Janssen-Cilag; and VIVITROL, a once-monthly injectable medication approved for the treatment of alcohol dependence, marketed in the U.S. primarily by Cephalon, Inc. The company's pipeline comprises extended-release injectable, pulmonary, and oral products for the treatment of prevalent and chronic diseases, such as central nervous system disorders, addiction, and diabetes. Its development programs also include AIR Insulin, which is in Phase III clinical development stage for the treatment of diabetes; Exenatide LAR, a Phase II clinical development stage product for the treatment of type 2 diabetes; a Phase I/II clinical study product, ALKS 29, for the treatment of alcohol dependence; a Phase IIa clinical study product, ALKS 27, for the treatment of COPD, which is a serious, chronic disease characterized by a gradual loss of lung function; and AIR parathyroid hormone, a Phase I clinical study product for the treatment of osteoporosis. In addition, the company has an early-stage pipeline focused on various disease areas. Alkermes has collaborations with Eli Lilly and Company; Amylin Pharmaceuticals, Inc.; Cephalon, Inc.; Indevus Pharmaceuticals, Inc.; and Janssen-Cilag. The company was founded in 1987 and is headquartered in Cambridge, Massachusetts.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
