10-2-2011: HeartWare Intl. EACTS/Lisbon Breakout Session
(U.S. BTT/CAP Update n=241)
Dr. Mark Slaughter (Univ.Louisville) & Heartware CEO Doug Godshall
“Results of the Heartware ADVANCE Bridge-to-Transplant Trial & CAP Update”
Data Cutoff: 7-15-2011, DB Lock: 8-25-2011
Webcast replay (1:28)…
HTWR Presentations: http://ir.heartware.com/phoenix.zhtml?c=187755&p=irol-presentations
Direct/EACTS_10-2-2011: http://www.media-server.com/m/acs/75a9a0b9282ffc35bd9a0eb06778c07b
Or: http://edge.media-server.com/m/p/5ax6sj23/lan/en
DR. SLAUGHTER’S 10-2-2011/EACTS SLIDES:


















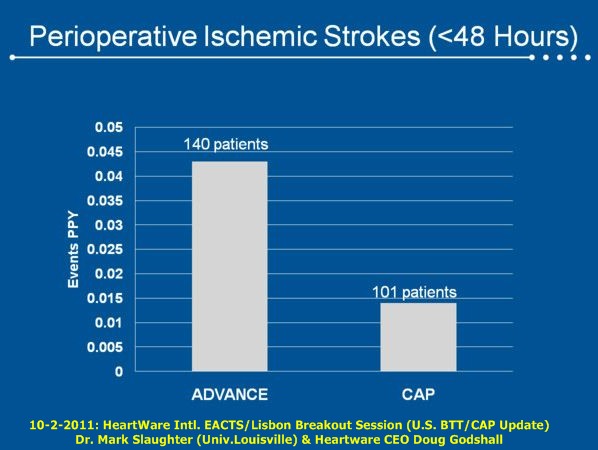











= = = = = = = = = = =
10-2-2011: HVAD/BTT Data Update given by Dr. Slaughter at 25th EACTS/Lisbon. “180-day survival of 93%” for 241 pts in ADVANCE trial & 3 CAP’s…
HeartWare Reports Additional Clinical Data From Bridge-to-Transplant and Continued Access Patients at the 25th European Association for Cardio-Thoracic Surgery (EACTS) Meeting in Lisbon
• Webcast Investor Meeting today at 9:00amET
SEE: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=67606997
. . .
OTHER OBSERVATIONS REPORTED DURING THE PRESENTATION INCLUDE:
• Only 21.6% of study patients had received a transplant at the 6-month endpoint, reflecting longer waiting times for heart transplantation;
• Reduction in the rate of pump exchange for suspected thrombus to less than 0.03 events per patient year, since an adjustment in anticoagulation in March;
• Reoperations for bleeding and sepsis, and driveline infections were notably infrequent.
- - - - -
Dr. Mark Slaughter, 10-2-11: "Consistent with previously presented data from this study, we observed high survival rates for patients through the 6-month endpoint, and particularly low observed rates for bleeding & infection. Adjustments in anticoagulation are contributing to reduced thrombus events, and overall adverse event rates remain low despite increased exposure to the device due to higher survival and a lower transplant rate."
- - - - -
CEO Doug Godshall. 10-2-11: "This data from our ADVANCE Bridge-to-Transplant clinical trial reaffirms the potential utility of the HVAD Pump in improving treatment outcomes for end-stage heart failure patients. We appreciate the strong support for this new generation of mechanical circulatory assist device from our international customers and U.S. clinical investigators as we continue to optimize implantation techniques and post-operative care."
FEATURED Cannabix Technologies Launches New Compact Breath Logix Workplace Series and Prepares for Delivery to South Africa • May 7, 2024 8:51 AM
Moon Equity Holdings, Corp. Announces Acquisition of Wikolo, Inc. • MONI • May 7, 2024 9:48 AM
NanoViricides Reports that the Phase I NV-387 Clinical Trial is Completed Successfully and Data Lock is Expected Soon • NNVC • May 2, 2024 10:07 AM
ILUS Files Form 10-K and Provides Shareholder Update • ILUS • May 2, 2024 8:52 AM
Avant Technologies Names New CEO Following Acquisition of Healthcare Technology and Data Integration Firm • AVAI • May 2, 2024 8:00 AM
Bantec Engaged in a Letter of Intent to Acquire a Small New Jersey Based Manufacturing Company • BANT • May 1, 2024 10:00 AM









