Monday, September 22, 2014 9:23:25 AM
Posted: Sep 22, 2014
Curcumin nanoparticles as innovative antimicrobial and wound healing agents
(Nanowerk Spotlight) Despite significant progress in medical treatments of severe burn wounds, infection and subsequent sepsis persist as frequent causes of morbidity and mortality for burn victims. This is due not only to the extensive compromise of the protective barrier against microbial invasion, but also as a result of growing pathogen resistance to therapeutic options.
Innovative therapies are urgently needed that overcome mechanisms of pathogen resistance – not only for thermal injuries but in general – and are easily administered without concerning systemic side effects (read more: "Nanotechnology solutions to combat superbugs").
"Antimicrobial resistance continues to be a growing crisis, highlighted by the FDA's Generating Antibiotic Incentives Now (GAIN) program, through which three new antibiotics with the indication for acute bacterial skin and skin structure infections were rapidly approved in unprecedented succession. All three however are systemically administered, and we have yet to see new topical antimicrobials emerge," Dr Adam Friedman, Assistant Professor of Dermatology and Director of Dermatologic research at the Montefiore-Albert Einstein College of Medicine, tells Nanowerk. "For me, this gap fuels innovation, serving as the inspiration for my research with broad-spectrum, multi-mechanistic antimicrobial nanomaterials."
In new work, Friedman and a team of researchers from Albert Einstein College of Medicine and Oregon State University have explored the use of curcumin nanoparticles for the treatment of infected burn wounds, an application that resulted in reduced bacterial load and enhancing wound healing.
The findings are available as an Article in Press in Nanomedicine ("Curcumin-encapsulated nanoparticles as innovative antimicrobial and wound healing agent").
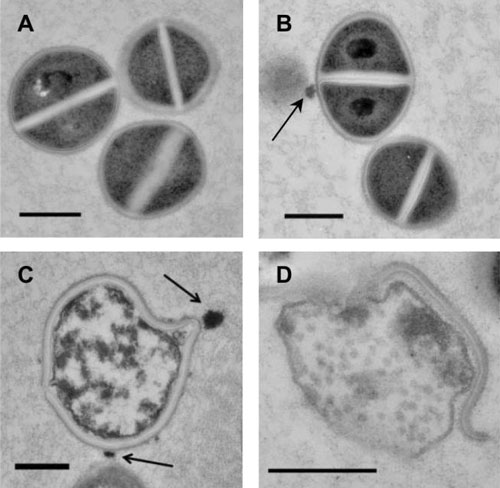
Curcumin nanoparticles induce cellular damage of MRSA. High-power transmission electron microscopy demonstrated interaction of nanoparticles (arrows) with MRSA cells. (A) Untreated MRSA showed uniform cytoplasmic density and central cross wall surrounding a highly contrasting splitting system. (B) After 24 hours, cells incubated with control nanoparticles 5 mg/ml did not exhibit changes in cellular morphology compared to untreated control. (C) After 6 hours, cells incubated with curcumin nanoparticles 5 mg/ml exhibited distortion of cellular architecture and edema, followed by lysis and extrusion of cytoplasmic contents after 24 hours (D). All scale bars=500 nm. (© Elsevier)
Turmeric (Curcuma longa L.) is the shining star among the cornucopia of traditional medicinal plants. It has a long history of usage in traditional medicine in India and China. Ancient Indians have known the medicinal properties of turmeric – i.e. curcumin – for several millennia.
In the scientific literature there is a large body of evidence showing that curcuminoids exhibit a broad spectrum of biological and pharmacological activities including anti-oxidant, anti-inflammatory, anti-bacterial, anti-fungal, anti-parasitic, anti-mutagen, anti-cancer and detox properties. Curcumin's unique ability to work through so many different pathways with its extraordinary antioxidant and anti-inflammatory attributes can have a positive influence in combating almost every known disease (read more: "Nanotechnology-enhanced curcumin: Symbiosis of ancient wisdom with modern medical science" and, if you are really into details, this: "Nanotechnology-enhanced curcumin - literature and patent analysis").
"There has been tremendous excitement regarding curcumin in multiple fields of medicine, most prominently in Oncology," Friedman points out. "Here, for the first time, we demonstrated that curcumin nanoparticles were more effective at both accelerating thermal burn wound closure and clearing infection with Methicillin Resistant S. aureus (MRSA) as compared to curcumin in its bulk size."
Friedman and his team utilized an innovative sol-gel-based polymerization technique to create silane composite nanoparticles that incorporate curcumin within a highly structured porous lattice. The versatility of the resulting nanoformulation allows for loading of different active ingredients, with therapeutic efficacy when applied topically, intradermally, and intravenously.
"While so much is known about curcumin's therapeutic potential, there have been numerous limitations with respect to clinical translation resulting from its poor solubility, instability at physiology pH and unsightly yellow-orange color," says Friedman. "Nanotechnology can and has overcome many of these impediments. At the nanoscale, the likelihood of curcumin interfacing with its intended target is much greater."
To sum it up, this work nicely demonstrates that curcumin nanoparticle technology circumvents the difficulties inherent in curcumin administration, enabling delivery of this therapeutic substance. Unlike currently used treatments, curcumin nanoparticles are less likely to select for resistant bacterial strains or delay wound healing.
"We believe our technology has the potential to serve as a novel topical agent for burn wound infection and possibly other cutaneous injuries," Friedman concludes.
By Michael Berger. Copyright © Nanowerk
http://www.nanowerk.com/spotlight/spotid=37448.php#ixzz3E3AvLQCj
Recent NNVC News
- NanoViricides Reports that the Phase I NV-387 Clinical Trial is Completed Successfully and Data Lock is Expected Soon • InvestorsHub NewsWire • 05/02/2024 02:07:42 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/16/2024 09:53:32 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 02/14/2024 09:55:35 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/19/2024 09:44:48 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 12/04/2023 09:08:49 PM
- Form 10-K - Annual report [Section 13 and 15(d), not S-K Item 405] • Edgar (US Regulatory) • 10/13/2023 08:30:41 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 09/28/2023 08:44:54 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 09/01/2023 08:46:45 PM
- Clinical Trial of Broad-Spectrum Antiviral Drug NV-CoV-2 is Progressing Well, Says NanoViricides - NV-CoV-2 is Positioned to Fulfill Many Unmet Medical Needs • InvestorsHub NewsWire • 07/06/2023 03:16:37 PM
VAYK Discloses Strategic Conversation on Potential Acquisition of $4 Million Home Service Business • VAYK • May 9, 2024 9:00 AM
Bantec's Howco Awarded $4.19 Million Dollar U.S. Department of Defense Contract • BANT • May 8, 2024 10:00 AM
Element79 Gold Corp Successfully Closes Maverick Springs Option Agreement • ELEM • May 8, 2024 9:05 AM
Kona Gold Beverages, Inc. Achieves April Revenues Exceeding $586,000 • KGKG • May 8, 2024 8:30 AM
Epazz plans to spin off Galaxy Batteries Inc. • EPAZ • May 8, 2024 7:05 AM
Moon Equity Holdings, Corp. Announces Acquisition of Wikolo, Inc. • MONI • May 7, 2024 9:48 AM










