Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Three out of four: Protlix also received marketing approval for its medicine for Fabry disease
The approval comes just after the company's stock was delisted from trading in Tel Aviv.
(Article published in Hbrew - Google translated, a non perfect translation.)
• MediWound and Gamida Cell also received marketing approvals for their drugs this year, after many
years of drought in obtaining approvals for Israeli drugs through new mechanisms
• BiolineRx is also awaiting the FDA's response.
Gali Vinrab 10.05.2023
The company Protelix, which produces drugs for rare diseases by engineering plant cells so that they express human proteins, has received marketing approval in the US for its drug for Fabry disease. Protelix is traded on the New York Stock Exchange at a value of 191 million dollars and in pre-opening trading the stock is up 17%. In March it was deleted The company's stock from trading in Tel Aviv. Even before that, it managed to rise nicely, in preparation for the possibility of receiving the approval. Its return in the last year on the New York Stock Exchange is 190%.
It is one of four Israeli companies that submitted innovative drugs to the FDA, the American Food and Drug Administration, for approval this year. This, after many years in which no innovative Israeli drug reached this status. Two companies have already received a positive answer: Gamida Sel, which is developing a drug that improves treatments for transplanting a new immune system in blood cancer patients, and Medivand, which has developed a drug against burns. Bioline, which is also developing a blood cancer treatment, is also waiting for the Authority's answer until the end of the year.
Protelix's drug is the only one that enters an existing market where it will compete head-to-head with Genzyme's drug Favrezyme. Genzyme's drug is produced in animal cells, while Protelix is the only one that produces it in plant cells. The entire market is estimated at about 2.2 billion dollars. Protelix has a marketing agreement in the US with the Chiesi company, which specializes in the field of rare diseases.
Protlix previously developed a drug for Gaucher's disease based on the same technology, and commercialized it to Pfizer. This drug generated revenue for the company, but due to delays in entering the market, it led to the fact that when it entered the market it was very competitive, and it was unable to take a significant place in the market, and Pfizer lost interest in it, and in the end the drug was not successful in the world market.
After learning this lesson, Protlix stated that it would only go to market with products that were superior to those already on the market. The Fabri product was designed to be better than existing products, and the company does state in its press release that it has a longer half-life. The studies conducted by the company were "non-inferiority" studies, and they did not establish that the half-life results in superiority of the product in terms of efficacy or safety.
That's why Protlix is conducting two follow-up trials for marketing purposes and obtaining insurance indemnity at the desired price. One trial was designed to support the claim that the product improves kidney function compared to Genzyme's product, and another trial was designed to support a treatment protocol according to which the injection is given once a month instead of once every two weeks.
The companies have not yet disclosed what the product's pricing will be compared to Genzyme's, but Protlix will probably not try to compete with an extremely low price, but will price it around the market price.
Amicus also competes in the American febrile market with the Galfold product, which can be swallowed and is only suitable for some patients. Outside of the USA, Shire (Takeda) also operates, which for a decade has not been able to get FDA approval.
The maximum revenue Protlix expects from the product is 150-200 million dollars per year. Protlix will produce the drug at the plant in Karmiel, where it also produces the drug for Gosha.
In the first quarter of this year, Protlix recorded revenues of 5.1 million dollars from the Ghosha product. At the end of the quarter, it had $33 million in cash. Upon approval, she is expected to receive a milestone payment from Casey.
The other two companies that received approvals from the beginning of the year, Medivand and Gamida Sel, did not, as a result, register significant increases in their shares over time. There is a difference between these two companies and Protelix: both companies market their products independently to markets where there are currently no other products, and they now need to invest in building a marketing system and educating the market, so they may require more time and significant investment before they reap the benefits of marketing their drugs . Protelix may have an easier path forward because it has already signed the deal with Casey. However, it is possible that some of these differences are already priced in the share price.
Pray To GOD - Trump did not know this -
https://truthpress.com/news/fauci-gave-scientist-2m-in-grant-money-after-pressuring-him-to-stay-quiet-about-covid-lab-theory/
NEW EPIDEMIC PEOPLE DROPPING DEAD FROM VAXX! SO MANY DEATHS MEDIA
CAN NO LONGER HIDE TRUTH!

https://www.bitchute.com/video/WKWqNMQ1qgzJ/
MAGA Tucker Carlson Today | Sudden Death Epidemic with Ed Dowd
2839 views
https://www.brighteon.com/a8d63027-dde1-43c8-845d-6cbb64d21fe2
THE FALL OF THE CABAL: 'ARRESTS AND EXECUTIONS'. "WE HAVE IT ALL" ~Q!
WATCH

https://www.bitchute.com/video/urarRSy3BosY/
WASHINGTON DC - SEX ORGIES AND DRUGS - EXPOSED -
WATCH

https://www.bitchute.com/video/176mrgQYS1RS/
How To Stop The War Monglers? - NATO PREPARES DRAFT FOR WORLD WAR 3...
https://www.brighteon.com/9051995e-2be8-4886-a4a7-84b82cdb0feb
How to stop a war -
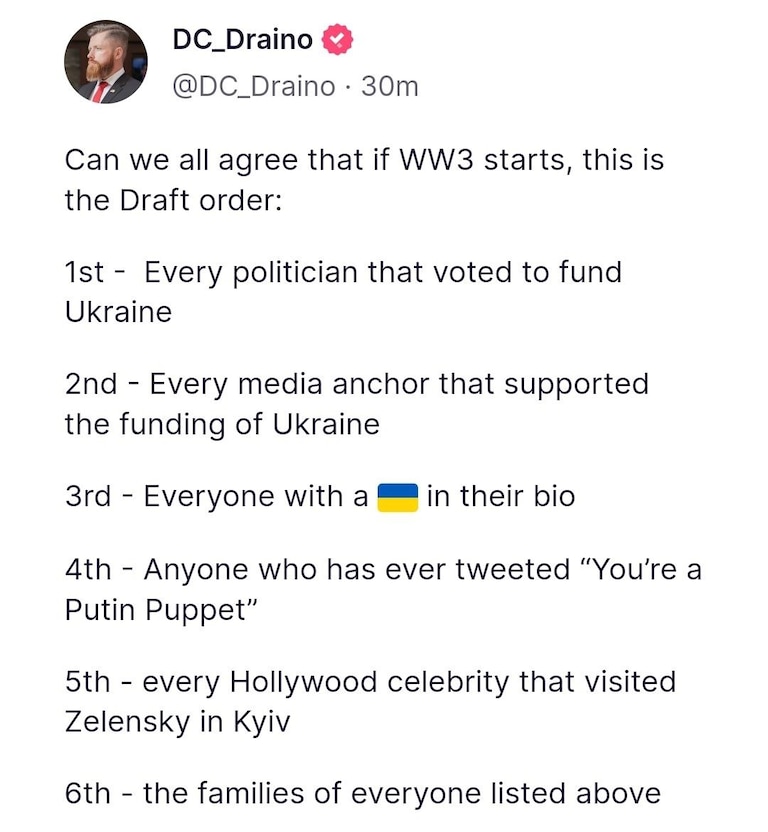
NATO and U.S. issue red alert against China and Putin | Redacted
with Clayton Morris















 Good Evening My Friends - it would be very interesting to hear
Good Evening My Friends - it would be very interesting to hear 





BREAKING: Fully Vaccinated Adults Account for Over 90% of COVID-Related Deaths!
Posted on February 25, 2023 by Constitutional Nobody
Key Points:
https://ussanews.com/2023/02/25/breaking-fully-vaccinated-adults-account-for-over-90-of-covid-related-deaths/
JESUS PRAYERS Needed Against - NWO = OWG KHZARIAN DEEPSTATE EVIL - THE ILLUMINATI AND ONE WORLD
GOVERNMENT WARMONGLERS NECON NAZIS =
DEPOPULATION AGAINST ALL HUMANITY -
WATCH

The Illuminati and One World Government
From Genesis to the Tower of Babel, to the
Egyptian Pharaohs, to Goat God's, to the World Bank, and the
FreeMasons, Secret Societies, Rosicrucians,
Rothschild bloodline, sexual perversions,
Marxism, Communism, Illuminati, Evil 666 -
https://www.bitchute.com/video/wzXLOeEAGm1d/
JESUS PRAYERS THANKS - SITUATION UPDATE GCR REPORT 2/20: JFK JR DECODE! HARRIS STARTS WW3?! EPSTEIN/CHASE BANK REVEAL! WOW!
WATCH

https://www.bitchute.com/video/IdhwLuCqaUg6/
https://www.bitchute.com/video/JrOIjbfGtLKs/
TRUMP LIFTS SPIRITS IN EAST PALESTINE, OHIO, WHILE BIDEN RESTS FROM
BANGING THE WAR DRUMS IN UKRAINE
WATCH

https://www.bitchute.com/video/YST1BSDWdsRw/
WATCH = NWO KHAZARIANS MAFIA MURDER OUR BROTHERS & SISTERS AND THE HUMANITY -
SECRET ISRAELI AND PFIZER CONTRACT EXPOSED BY STEVE KIRSCH AND STEW
PETERS
WATCH

https://www.bitchute.com/video/0eh6vANp9pOj/
Pp Watch - STEW PETERS: MARXISTS HIJACKED AMERICA, WHO IS RUNNING THE UNITED STATES - 2/20/23
WATCH

https://www.bitchute.com/video/wTE2J0hekgIK/
Evil eugenics MAID in Canada: Socialist left's massive genocide disguised as medical care
Mirror. Source
Evil Eugenics MAID In Canada: Socialist Left's MASSIVE Genocide Disguised As Medical Care https://odysee.com/@TimTruth:b/canada-maid-genocide:1

https://www.bitchute.com/video/K4zArax8Es6v/
Quote: "Evil Eugenics MAID In Canada: Socialist Left's MASSIVE Genocide Disguised As Medical Care Want more videos? Join https://GroupDiscover.com to find the best videos from across the free speech internet platforms like Odysee, Rumble, Bitchute & Brighteon all in one huge video repository. Add me on these great platforms: https://rokfin.com/timtruth https://odysee.com/@TimTruth:b/ https://rumble.com/timtruth https://bitchute.com/timtruth/ https://GroupDiscover.com Support links (thank you to all the supporters!): Easy to do one time tips via https://rokfin.com/timtruth or https://odysee.com/@TimTruth:b https://timtruth.substack.com/subscribe https://subscribestar.com/timtruth "
-
5G is a weapon system - Don't be fooled by the fake narrative https://tinyurl.com/tf38xs3d ~ The agenda - They are destroying human kind https://tinyurl.com/2p82r3j9 ~ 60GHz in schools - Lena Pu and Mark Steele https://tinyurl.com/2c67ep66 ~ 5G target acquiring weapon system - This is not for control but an extermination technology https://tinyurl.com/4hetn32u ~ UK Government hacked https://tinyurl.com/337zjb4s ~ Report #133: David Noakes on GcMAF cancer treatments, FDA/MHRA/Pharma corruption, & wrongful charges https://tinyurl.com/ev8kms8n ~ BitChute { noakes falconscafe https://tinyurl.com/2h7z47ve } ~ The disciples of Ra: The deception of "medicine", viruses & vaccines https://tinyurl.com/2p8uc7as ~ Viruses don't exist https://is.gd/E4li0z ~ If you don't know what causes what they call a virus you will never know unless you read the science https://tinyurl.com/yj8j9pd2 ~ Assembling the kill grid ~ Excerpt: Mark Steele https://tinyurl.com/4cethr4b ~ Prof. Francis Boyle "The British must not take these frankenshots"! Interview https://tinyurl.com/3cbrwts2 ~ The MAC phenomenon in people "vaccinated" from COVID-19 https://tinyurl.com/2p8xhjz3 ~ Video summary of La Quinta Columna that shows evidence of genocide based on injectable analysis https://tinyurl.com/43bdk4na
Illegal organ trafficking of homeless people in Texas? Same thing happened during Hurricane Katrina https://tinyurl.com/ym7uyt3e ~ Homeless vet killing society https://tinyurl.com/y2ycpn6m ~ NATO satanism, testimony, Kay Griggs: Colonel's wife tell-all, oppression, deception, secret society https://tinyurl.com/2p8ybsjv ~ Horus matrix at Normandy Omaha Beach Overlord D-Day 666 Cemetery satanic ritual sacrifice https://tinyurl.com/yckjeu8r ~ The cover up continues - Share this with all vaccinated, who have been lied to by their doctors https://tinyurl.com/3w65f9ny ~ Whistleblower: Hospitals killing for organs, "This is absolutely evil and a crime against humanity!" https://tinyurl.com/4mp7h8vy ~ The world must know #PureEvil #HellOnEarth https://tinyurl.com/2p93msb3 ~ Bombshell: Pfizer vaccine study's massive list of "Adverse events of interest" https://tinyurl.com/yc7tyu2r
Did he just say snake venom? - Dr Bryan Ardis talks to Right Now https://tinyurl.com/4chrmwy8 ~ World premiere: Watch the Water https://tinyurl.com/3ybuwhxv ~ Part 1/3 - Dr. Bryan Ardis reveals bombshell origins of COVID, mRNA vaccines and treatments https://tinyurl.com/38earx4a ~ Biological weapons; Is there a link between the water supply system and the pandemic? https://tinyurl.com/2p8pvuze ~ Professor Darrel Hamamoto on persecution and inquisition at UC Davis https://tinyurl.com/4wkcjcu3 ~ The China-NHS lateral flow test, massive fraud, for those that lost work.. or murdered on COVID ward https://tinyurl.com/2mbamwmf ~ COVID-19 test fraud, also carcinogenic https://tinyurl.com/2p99uwws ~ Your future The SPARS pandemic 2025 - 2028 https://is.gd/kCajO1
Snuff Hill https://tinyurl.com/573ufnvj ~ Blood Hill https://tinyurl.com/ymckkptu ~ Fitzwilliam military cult https://tinyurl.com/bdhz7529 ~ Troy River https://tinyurl.com/2p86hv66 ~ Tent City https://tinyurl.com/56hfw4kf ~ 18 Brickyard Troy Depot, Troy School, Cemetery, Discount Tire, satanic stalking, ritual sacrifice https://tinyurl.com/2jvdutm2 ~ Bohemian Grove Jr, Bridgewater Associates - CIA corporate front, CIA role in snuff and pornography https://tinyurl.com/2p8v8yr5 ~ Hebron Coven ~ Part 1 to 4 of 9 https://tinyurl.com/yw952bnn ~ Body Organs Of Over 18,000 Syrian Children Sold in Six Years https://tinyurl.com/djarv3w8 ~ I saw kids in cages outside a masonic lodge being loaded into trucks https://tinyurl.com/46uxrvs5 ~ Amazon USB key - Part 1 to 2 - CYM Adrenochrome https://tinyurl.com/yckfvnn2
The men on the moon https://tinyurl.com/42dh2ejv ~ Moon truth https://tinyurl.com/mrxx5sks ~ 7 rockets hit dome ! https://tinyurl.com/46rd63v5 ~ Who shot the moon landing, classroom bloopers https://tinyurl.com/mw7xwh39
Oil is abundant and cheap https://tinyurl.com/3e2nkbbm
MAGA Prayers To Father GOD Needed - THE WORLD VS. THE KHAZARIAN MAFIA -- JIM WILLIE
WATCH

https://www.bitchute.com/video/PSIN2glcoaju/
Watch MAGA Thanks - Pray To Get Father GOD'S Remedies Back To US - ROCKEFELLER INFLUENCED THE WORLD TO REMOVE ALL THE NATURAL
REMEDIES FROM PRACTICE FOR HIS BENEFIT
WATCH

https://www.bitchute.com/video/uD36QiNCxC8O/
China Says Ready To "Join Forces With Russia" To "Defend National Interests" As Putin Confirms Xi Visit
Tyler Durden's Photo
BY TYLER DURDEN
WEDNESDAY, FEB 22, 2023 - 10:00 AM
Despite all latest among Washington's repeat warnings to Beijing against strategic or military cooperation with Moscow, China is now pledging to "join forces" with "like-minded" partner Russia to defend national interests. The statement came by the close of the first day of the director of the Office of the Foreign Affairs Commission of the Communist Party of China’s Central Committee Wang Yi's trip to Moscow.

https://www.zerohedge.com/geopolitical/china-says-ready-join-forces-russia-defend-national-interests-putin-confirms-xi-visit
CH - TOXIC FUMES FROM OHIO MAY SERIOUSLY AFFECT EVERYONE EAST OF THE MISSISSIPPI RIVER 250 MILLION PEOPLE
WATCH

https://www.bitchute.com/video/hCb5IvRRkL7G/
$500 TRILLION LAWSUIT AGAINST THE FEDERAL GOVT AND OVER 140 MONOPOLISTS (REMOVED BY YOUTUBE IN 5HRS)
WATCH

https://www.bitchute.com/video/QFcfDjifRl6u/
Even the doctors fell for it
https://rumble.com/v2aenx2-more-than-130-canadian-doctors-have-died-suddenly-since-c19-killshots-began.html
5G – Microwave as a weapon Part 2 - Dr. Reiner Fuellmich and Barrie Trower https://rumble.com/v2a3th8-5g-microwave-as-a-weapon-part-2- dr.-reiner-fuellmich-and-barrie-trower.html | Towards The Light channel https://www.bitchute.com/cha…

https://www.bitchute.com/video/RaEZsuhWr2G0/
NWO Biden like a 3 year old -

MAGA JESUS PRAYERS THANKS - SITUATION UPDATE GCR REPORT 2/20: JFK JR
DECODE! HARRIS STARTS WW3?! EPSTEIN/CHASE BANK REVEAL! WOW!
WATCH

https://www.bitchute.com/video/IdhwLuCqaUg6/
https://www.bitchute.com/video/JrOIjbfGtLKs/
TRUMP LIFTS SPIRITS IN EAST PALESTINE, OHIO, WHILE BIDEN RESTS FROM
BANGING THE WAR DRUMS IN UKRAINE
WATCH

https://www.bitchute.com/video/YST1BSDWdsRw/
WATCH = NWO KHAZARIANS MAFIA MURDER OUR BROTHERS & SISTERS AND THE HUMANITY -
SECRET ISRAELI AND PFIZER CONTRACT EXPOSED BY STEVE KIRSCH AND STEW
PETERS
WATCH

https://www.bitchute.com/video/0eh6vANp9pOj/
Pp Watch - STEW PETERS: MARXISTS HIJACKED AMERICA, WHO IS RUNNING THE UNITED STATES - 2/20/23
WATCH

https://www.bitchute.com/video/wTE2J0hekgIK/
Evil eugenics MAID in Canada: Socialist left's massive genocide disguised as medical care
Mirror. Source
Evil Eugenics MAID In Canada: Socialist Left's MASSIVE Genocide Disguised As Medical Care https://odysee.com/@TimTruth:b/canada-maid-genocide:1

https://www.bitchute.com/video/K4zArax8Es6v/
Quote: "Evil Eugenics MAID In Canada: Socialist Left's MASSIVE Genocide Disguised As Medical Care Want more videos? Join https://GroupDiscover.com to find the best videos from across the free speech internet platforms like Odysee, Rumble, Bitchute & Brighteon all in one huge video repository. Add me on these great platforms: https://rokfin.com/timtruth https://odysee.com/@TimTruth:b/ https://rumble.com/timtruth https://bitchute.com/timtruth/ https://GroupDiscover.com Support links (thank you to all the supporters!): Easy to do one time tips via https://rokfin.com/timtruth or https://odysee.com/@TimTruth:b https://timtruth.substack.com/subscribe https://subscribestar.com/timtruth "
-
5G is a weapon system - Don't be fooled by the fake narrative https://tinyurl.com/tf38xs3d ~ The agenda - They are destroying human kind https://tinyurl.com/2p82r3j9 ~ 60GHz in schools - Lena Pu and Mark Steele https://tinyurl.com/2c67ep66 ~ 5G target acquiring weapon system - This is not for control but an extermination technology https://tinyurl.com/4hetn32u ~ UK Government hacked https://tinyurl.com/337zjb4s ~ Report #133: David Noakes on GcMAF cancer treatments, FDA/MHRA/Pharma corruption, & wrongful charges https://tinyurl.com/ev8kms8n ~ BitChute { noakes falconscafe https://tinyurl.com/2h7z47ve } ~ The disciples of Ra: The deception of "medicine", viruses & vaccines https://tinyurl.com/2p8uc7as ~ Viruses don't exist https://is.gd/E4li0z ~ If you don't know what causes what they call a virus you will never know unless you read the science https://tinyurl.com/yj8j9pd2 ~ Assembling the kill grid ~ Excerpt: Mark Steele https://tinyurl.com/4cethr4b ~ Prof. Francis Boyle "The British must not take these frankenshots"! Interview https://tinyurl.com/3cbrwts2 ~ The MAC phenomenon in people "vaccinated" from COVID-19 https://tinyurl.com/2p8xhjz3 ~ Video summary of La Quinta Columna that shows evidence of genocide based on injectable analysis https://tinyurl.com/43bdk4na
Illegal organ trafficking of homeless people in Texas? Same thing happened during Hurricane Katrina https://tinyurl.com/ym7uyt3e ~ Homeless vet killing society https://tinyurl.com/y2ycpn6m ~ NATO satanism, testimony, Kay Griggs: Colonel's wife tell-all, oppression, deception, secret society https://tinyurl.com/2p8ybsjv ~ Horus matrix at Normandy Omaha Beach Overlord D-Day 666 Cemetery satanic ritual sacrifice https://tinyurl.com/yckjeu8r ~ The cover up continues - Share this with all vaccinated, who have been lied to by their doctors https://tinyurl.com/3w65f9ny ~ Whistleblower: Hospitals killing for organs, "This is absolutely evil and a crime against humanity!" https://tinyurl.com/4mp7h8vy ~ The world must know #PureEvil #HellOnEarth https://tinyurl.com/2p93msb3 ~ Bombshell: Pfizer vaccine study's massive list of "Adverse events of interest" https://tinyurl.com/yc7tyu2r
Did he just say snake venom? - Dr Bryan Ardis talks to Right Now https://tinyurl.com/4chrmwy8 ~ World premiere: Watch the Water https://tinyurl.com/3ybuwhxv ~ Part 1/3 - Dr. Bryan Ardis reveals bombshell origins of COVID, mRNA vaccines and treatments https://tinyurl.com/38earx4a ~ Biological weapons; Is there a link between the water supply system and the pandemic? https://tinyurl.com/2p8pvuze ~ Professor Darrel Hamamoto on persecution and inquisition at UC Davis https://tinyurl.com/4wkcjcu3 ~ The China-NHS lateral flow test, massive fraud, for those that lost work.. or murdered on COVID ward https://tinyurl.com/2mbamwmf ~ COVID-19 test fraud, also carcinogenic https://tinyurl.com/2p99uwws ~ Your future The SPARS pandemic 2025 - 2028 https://is.gd/kCajO1
Snuff Hill https://tinyurl.com/573ufnvj ~ Blood Hill https://tinyurl.com/ymckkptu ~ Fitzwilliam military cult https://tinyurl.com/bdhz7529 ~ Troy River https://tinyurl.com/2p86hv66 ~ Tent City https://tinyurl.com/56hfw4kf ~ 18 Brickyard Troy Depot, Troy School, Cemetery, Discount Tire, satanic stalking, ritual sacrifice https://tinyurl.com/2jvdutm2 ~ Bohemian Grove Jr, Bridgewater Associates - CIA corporate front, CIA role in snuff and pornography https://tinyurl.com/2p8v8yr5 ~ Hebron Coven ~ Part 1 to 4 of 9 https://tinyurl.com/yw952bnn ~ Body Organs Of Over 18,000 Syrian Children Sold in Six Years https://tinyurl.com/djarv3w8 ~ I saw kids in cages outside a masonic lodge being loaded into trucks https://tinyurl.com/46uxrvs5 ~ Amazon USB key - Part 1 to 2 - CYM Adrenochrome https://tinyurl.com/yckfvnn2
The men on the moon https://tinyurl.com/42dh2ejv ~ Moon truth https://tinyurl.com/mrxx5sks ~ 7 rockets hit dome ! https://tinyurl.com/46rd63v5 ~ Who shot the moon landing, classroom bloopers https://tinyurl.com/mw7xwh39
Oil is abundant and cheap https://tinyurl.com/3e2nkbbm
MAGA Prayers To Father GOD Needed - THE WORLD VS. THE KHAZARIAN MAFIA -- JIM WILLIE
WATCH

https://www.bitchute.com/video/PSIN2glcoaju/
Watch MAGA Thanks - Pray To Get Father GOD'S Remedies Back To US - ROCKEFELLER INFLUENCED THE WORLD TO REMOVE ALL THE NATURAL
REMEDIES FROM PRACTICE FOR HIS BENEFIT
WATCH

https://www.bitchute.com/video/uD36QiNCxC8O/
China Says Ready To "Join Forces With Russia" To "Defend National Interests" As Putin Confirms Xi Visit
Tyler Durden's Photo
BY TYLER DURDEN
WEDNESDAY, FEB 22, 2023 - 10:00 AM
Despite all latest among Washington's repeat warnings to Beijing against strategic or military cooperation with Moscow, China is now pledging to "join forces" with "like-minded" partner Russia to defend national interests. The statement came by the close of the first day of the director of the Office of the Foreign Affairs Commission of the Communist Party of China’s Central Committee Wang Yi's trip to Moscow.

https://www.zerohedge.com/geopolitical/china-says-ready-join-forces-russia-defend-national-interests-putin-confirms-xi-visit
CH - TOXIC FUMES FROM OHIO MAY SERIOUSLY AFFECT EVERYONE EAST OF THE MISSISSIPPI RIVER 250 MILLION PEOPLE
WATCH

https://www.bitchute.com/video/hCb5IvRRkL7G/
$500 TRILLION LAWSUIT AGAINST THE FEDERAL GOVT AND OVER 140 MONOPOLISTS (REMOVED BY YOUTUBE IN 5HRS)
WATCH

https://www.bitchute.com/video/QFcfDjifRl6u/
Even the doctors fell for it
https://rumble.com/v2aenx2-more-than-130-canadian-doctors-have-died-suddenly-since-c19-killshots-began.html
5G – Microwave as a weapon Part 2 - Dr. Reiner Fuellmich and Barrie Trower https://rumble.com/v2a3th8-5g-microwave-as-a-weapon-part-2- dr.-reiner-fuellmich-and-barrie-trower.html | Towards The Light channel https://www.bitchute.com/cha…

https://www.bitchute.com/video/RaEZsuhWr2G0/
NWO Biden like a 3 year old -

INSIGHTEC ANNOUNCES POSITIVE COVERAGE DECISION BY CIGNA FOR FOCUSED ULTRASOUND TO TREAT ESSENTIAL TREMOR
Cigna is the fourth national payor to cover the outpatient procedure.
HAIFA, Israel & MIAMI, Jan. 31, 2023 /PRNewswire/ -- Insightec, a global healthcare company dedicated to using acoustic energy to transform patient care, today announced that Cigna is the fourth national payor to cover focused ultrasound for treating medication-refractory essential tremor.
"We are so excited to have another national payor providing access to focused ultrasound to non-Medicare patients," said Dee Kolanek, Insightec Vice President of Market Access and Reimbursement. "We want to extend our thanks to leading insurers like Cigna for their decision to cover this game-changing outpatient treatment for essential tremor patients."
Essential tremor, a movement disorder that causes uncontrollable shaking, affects an estimated ten million Americans. For patients whose tremor does not respond to medications, focused ultrasound is performed in a single, outpatient procedure with many patients showing immediate improvement of tremor in the treated hand with minimal complications.
"This is important progress towards our goal of expanding access to this transformative treatment option and we want to thank Cigna for their positive coverage decision," added Maurice R. Ferré MD, Insightec's CEO and Chairman of the Board of Directors. "Now, more patients with essential tremor can get back their ability to live independently, return to work, and improve their mental and physical health."
Cigna becomes the fourth national payor to cover the treatment after Anthem, Aetna, and Blue Cross Blue Shield Association gave positive coverage decisions in December 2022, April 2021, and August 2018, respectively. There are currently more than 125 treatment centers around the world using Insightec's Exablate® Neuro platform to treat essential tremor. Focused ultrasound treatment for medication-refractory essential tremor is covered by Medicare in all 50 states.
A sound you can’t hear but may one day change your life
By Kristen Rogers, CNN
Published 10:17 AM EST, Sun January 15, 2023
https://edition.cnn.com/2023/01/15/health/focused-ultrasound-tremor-depression-ocd-wellness/index.html
Brenda Hric consults with neurosurgeon Dr. Jeff Elias, who conducted her focused ultrasound.
Undergoing clinical trials around the world is a brain surgery that doesn’t need an incision or produce any blood yet drastically improves the lives of people with essential tremor, depression and more. The procedure, known as a focused ultrasound, aims sound waves at parts of the brain to disrupt faulty brain circuits causing symptoms.
“Focused ultrasound is a noninvasive therapeutic technology,” said Dr. Neal Kassell, founder and chairman of the Focused Ultrasound Foundation. “We’ve said that focused ultrasound is the most powerful sound you will never hear, but sound that someday could save your life.”
Kassell describes the way it works as “analogous to using a magnifying glass to focus beams of light on a point and burn a hole in a leaf.”
“With focused ultrasound, instead of using an optical lens to focus beams of light,” he added, “an acoustic lens is used to focus multiple beams of ultrasound energy on targets deep in the body with a high degree of precision and accuracy, sparing the adjacent normal tissue.”
The procedure has been significantly beneficial for people with essential tremor, a neurological disorder that causes involuntary and rhythmic shaking. The disorder can affect almost any body part, but the tremors typically occur in hands — even during simple tasks such as eating, drinking or writing.
Essential tremor is usually more prominent on one side of the body and can worsen with movement. It’s most common in people 40 and older, and it affects nearly 25 million worldwide, according to a 2021 study.
Such was the case with Brenda Hric, 80, who recently underwent focused ultrasound at the University of Virginia, a pioneering institution of the procedure.
Hric’s tremors made her uncomfortable in social situations because she was afraid of spilling or knocking something over, she told CNN.
But just 44 seconds of focused ultrasound waves got rid of her tremor.
“I looked at my hand, and I could see that it was not moving, and that was the first time I had been able to see my fingers still in about 20 years,” Hric said. “I think it’s definitely a miracle, and I thank the Lord for it.”
How it works
Focused ultrasound is a form of functional neurosurgery, the targeting of precise structures deep in the brain to change it, to restore function or, in this case, to stop a tumor. It’s an alternative treatment for those who, like Hric, don’t respond to or stop being affected by conventional medication treatment, experts said.
“In a simplistic sense, you can imagine that there’s a bunch of abnormal neurons in this one target that are firing away uncontrollably, causing the tremor, the shaking,” Kassell said.
Focused ultrasound technology uses a transducer to force beams of sound waves to converge at one point to raise the temperature and destroy tissue.
Before receiving high-intensity focused ultrasound, the one necessary for treating essential tremor, patients need to have their heads shaved since air can sometimes get trapped in hair follicles.
The patient then undergoes MRI and CT scans so doctors can use the resulting images to map the structure of the brain and the target.
Pictured are scans of Hric's brain. Focused ultrasound signficantly improved the 80-year-old's tremors.
The Insightec Exablate Neuro, a focused ultrasound platform, instructs how many beams should be used to do the treatment, then neurosurgeons might do what Dr. Jeff Elias calls “test shots, just to make sure we’re focused right at the bull’s-eye.”
A UVA Health neurosurgeon who treated Hric, Elias is a pioneer of treating essential tremors using ultrasound waves. In 2011, he led the clinical trials critical for gaining regulatory approval of this procedure in the United States.
“These (test shots) are really low energy, but we want to see if our treatment is exactly where we want it,” he said. “This is our chance to kind of sight the rifle.”
Four 11-second treatment doses significantly improved Hric’s tremor. The entire procedure lasted less than two hours, with most of it spent mapping the brain and testing the target.
Beforehand, Hric had trouble drawing inside the lines of circles. Focused ultrasound helped her color inside the lines.
Pros and cons
Generally, anyone with an essential tremor diagnosis not responding to medications would be eligible for focused ultrasound treatment, said Dr. Nir Lipsman, a scientist at Sunnybrook Health Sciences Centre in Toronto and director of Sunnybrook’s Harquail Centre for Neuromodulation.
People who can’t undergo MRI scans due to claustrophobia or having metal inside their body aren’t eligible for focused ultrasound, said Dr. Noah Philip, a professor of psychiatry and human behavior at Brown University’s Alpert Medical School. Philip is also lead for mental health research at the VA RR&D Center for Neurorestoration and Neurotechnology.
Ideally, the benefits of focused ultrasound are permanent, Lipsman said. “If you’re able to destroy the part of the brain responsible for the tremor, it should be a permanent effect,” he said. “At one year, however, some of these patients will have a rebound or recurrence of their tremor, and we don’t know why that is.”
MICE
Old mice grow young again in study. Can people do the same?
Such a return can happen with medication treatment, too, though — which is why some essential tremor patients turn to focused ultrasound in the first place.
But some patients have experienced the benefits five years after undergoing focused ultrasound, according to a 2022 study by Elias.
Potential side effects of focused ultrasound are why the mapping and testing parts of the procedure are so important. If the wrong area is targeted or treated excessively, a patient’s balance and stability can be harmed long term.
“The most common risks that we encounter in patients is a temporary numbness or tingling that can sometimes happen in the treated arm or in the lip area,” Lipsman said. “The vast majority of the time that goes away with time.”
Other common, but usually temporary, risks include slight unsteadiness on one’s feet after the procedure. But doctors don’t use a general anesthetic or hospitalize patients for this procedure, he added.
What’s next in the field
Today, focused ultrasound technology is used globally in various stages, including clinical trials and approved regulatory use. There are more than 170 clinical uses — including for neurodegenerative disorders and tumors of the brain, breast, lung, prostate and more — and the field is growing, Kassell said.
“You can watch the effect of the ultrasound treatment in real time while the treatment’s being administered, whereas with radiation, the effect of the treatment is invisible while it’s being administered,” Kassell said. “And it takes weeks or months for the effect of radiation to become apparent.”
Background design of human profile and fractal forms on the subject of inner reality, mental health, imagination, thinking and dreaming
Severe depression eased by single dose of synthetic 'magic mushroom'
Use for depression and obsessive-compulsive disorder is on the table, according to a small 2020 study by Lipsman and a team of researchers. They found focused ultrasound was safe and effective in improving symptoms for people with major depression and OCD. But further studies are needed.
One limitation of focused ultrasound is that not every person’s skull is made equal, Lipsman said.
“The density of the skull has a major impact on the ability of ultrasound to travel through it,” he added. “It’s rare, but there are some patients that, try as we might, we cannot make an effective lesion in the brain. The skull does not allow the passage of ultrasound. So that’s a technical limitation of the technology, something that we’re actively working on.”
Focused ultrasound isn’t available for every condition, but experts said they are hopeful that “medicine’s best-kept secret” will one day become a standard treatment.
“My belief is that in 10 years,” Kassell said, “focused ultrasound will be a mainstream therapy that is affecting millions of patients every year around the world. It’ll be widely accepted.”
CNN’s Adeline Chen contributed to this report.
Four Israeli cos expect FDA approvals in 2023
29 Dec, 2022 22:00
Gali Weinreb
Treatments for blood cancer and rare diseases are the target markets, but investors are well aware of the challenges that await after approval is received.
You can count the number of drugs of independent Israeli companies that have been approved by the US Food and Drug Administration (FDA) on the fingers of two hands. Teva Pharmaceutical Industries (TASE: TEVA; NYSE: TEVA) takes four, with MS treatment Copaxone, Parkinson’s Disease treatment Azilect, Austedo, for chorea associated with Huntington’s Disease, and Ajovy, for preventive treatment of migraine. Another goes to Interpharm, which registered Rebif but which is no longer Israeli. They are joined by Kamada (TASE: KMDA; Nasdaq: KMDA), RedHill Biopharma (Nasdaq: RDHL), Protalix Biotherapeutics (TASE: PLX; NYSE: PLX), Purple Biotech (Nasdaq: PPBT), Chiasma, and more recently UroGen Pharma (Nasdaq: URGN), in 2020. These latter companies produced drugs that all underwent challenging clinical trials, but all of them were variations of known substances. In fact, since 2002, when Rebif was approved, there has been no approval of a drug not based on an existing product, apart from one drug by Teva.
In 2023, four more approvals could be obtained by Israeli companies MediWound (Nasdaq: MDWD), Gamida Cell (Nasdaq: GMDA), BioLineRx (TASE: BLRX; Nasdaq: BLRX), and (again) Protalix, in addition to a drug from Teva, a delayed release version of a treatment for schizophrenia. Three of the products concerned have completely new action mechanisms.
What led to four companies reaching the finishing line at the same time, after years of drought? One explanation lies in a change in exit patterns in the biomed industry. These companies didn’t necessarily want to reach the finishing line while still responsible for the development of their drugs, but because they have received no worthwhile acquisition offers (so far) they have had to do so.
There is also a positive explanation. All these companies managed to raised money on Nasdaq or the New York Stock Exchange during the years of plenty. Each raised hundreds of millions of dollars in fairly small rounds over a period of years, as the stock exchanges in the US have allowed in recent years.
In addition, in the case of three of the four companies, Clal Biotechnology Industries, which is traded on the Tel Aviv Stock Exchange, is a substantial shareholder.
The four companies have something else in common: they all address fairly small markets, and have received certain relaxations in their development tracks, something else that has become widespread in recent years. This is what has enabled Israeli companies to reach the approval application stage at an investment of just a few hundred million of dollars, and not the many hundreds of millions, or even billions, required to develop drugs for large markets.
Share prices not responding
If the FDA does approve the drugs, the companies will be able to start marketing them in a few months’ time, at the same time as they try to obtain insurance coverage for them. There are also intermediate statuses between approval and no approval, such as requests for further information, for small supplementary trials, for another expensive efficacy trial (especially since most of the companies have carried out just one Phase 3 trial), for another factory audit, and so on. Any such partial response will be liable to put back approval by between a few months and several years, depending on what the FDA requires. Sometimes, providing the required additional information is such a costly process that it makes the product uneconomic.
In advance of an important trial or the possibility that a drug will be approved, the share price of the company concerned will generally rise, but in these four cases the past two years have been hard for their stocks. They are down 70-80% from their peaks. Even though possible approval is near, all of these stocks are a long way from correcting their declines, and, apart from Protalix, have seen no significant rise.
Perhaps that will yet happen as the time for receiving the FDA’s response approaches, but it could be that this year the market forces depressing the biomed sector are stronger than the desire to gamble on a positive outcome. Perhaps those who might invest in small independent companies fear that such an outcome will lead to further fund raising, putting pressure on the share price, besides which, they know that approval is just the start.
The day after approval
The tough life of an independent drug company becomes even tougher the day after approval. Each of these companies will come to a market in which there is already competition, even if not necessarily a similar product. Each will face the challenges of positioning, pricing, marketing, and production, and each has geared up for these challenges differently.
MediWound: Topical treatment for burns that will compete with surgeons
Founded: 2000
CEO: Ofer Gonen
Field: Treatment of wounds and burns
Market cap: $67m
MediWound, which appointed Ofer Gonen as CEO in May this year, could obtain marketing approval within the next few days. The company has developed a product to treat burns based on the pineapple plant, and it could be one of the first companies to receive FDA approval for a botanical drug, although the emphasis is more on the way that the plant is processed.
The product removes dead tissue from burns (and potentially from wounds as well, but the forthcoming approval is only for burns). It mainly competes with surgery to remove burns, which requires more expensive manpower and removes more healthy tissue. The product is already on sale in Europe. The US market fro treatment of burns in adults is estimated at $200 million, and MediWound has signed a marketing agreement with Vericel.
At the end of September, MediWound had $35 million cash. In October, it raised $30 million, and it will receive $7.5 million as a milestone payment, which means that it will have enough cash to last it until 2025. Meanwhile, there could also be developments in the product for treating wounds, which has a larger potential market, and the company has already said that it is examining options for a further strategic agreement.
Gamida Cell: Improving success rates for bone marrow transplants
Founded: 1998
CEO: Abigail Jenkins
Field: Improving bone marrow transplants for cancer patients
Market cap: $90m
Gamida Cell has developed a product designed to improve the success of stem cell transplants to treat blood cancers. In a clinical trial, the company demonstrated that its product cut the time taken for absorption of the transplanted immune system from 22 days to 12. This is a significant reduction, as the time saved is a period precious to the transplant center and dangerous for the patient, who is without an active immune system. The trial also showed a reduction in patient infections and in the time spent in hospital.
The original date for FDA approval was January 30, but after the documents were filed the FDA asked for further information. Investors reacted by sending the share price down 20%, but a request for further information before the final approval date is generally preferable to one that comes after it.
Gamida Cell is gearing up to market its product itself, and although it has other interesting products in its pipeline, at present it is devoting most of it resources to this process. Its advantage and disadvantage is its small market. It believes that its product could be relevant to about 10,000 patients annually, in 70 medical centers, that can be covered by 25 salespeople. 19 of these centers have already tried the product.
The company will have to price its product high. Will the insurance companies accept that? The card it holds vis-à-vis them is the saving in hospitalization time and in complications. In any event, it will take time to obtain coverage, and the company will have to be prepared to finance some of the procedures itself at first, to help the product gain momentum.
Gamida Cell intends to produce at a Jerusalem plant. Production is complex, and the company will have to ensure that it is profitable.
Protalix: Head-to-head with a dominant treatment for Fabry Disease
Founded: 1993
CEO: Dror Bashan
Field: Extraction of plant cell proteins for treating rare diseases
Market cap: $60m
Protalix’s drug for treating the rare genetic condition Fabry Disease will enter a competitive market dominated by Genzyme (Sanofi) with Fabrazyme. Amicus Therapeutics is also in this market with Galafold, which is administered orally and is only suitable for some of the patients. Outside the US, Shire (Takeda) has been trying without success for a decade to obtain FDA approval for a treatment.
Fabry Disease stems from a deficiency in the Alpha Galactosidase A enzyme, and all these products are actually a protein that the body fails to produce. Genetic editing treatments are currently undergoing trials. These are meant to make the body produce the protein by itself. One company developing such a treatment had a well-publicized failure in a trial.
Protalix carried out three trials to demonstrate the efficacy and safety of its product, among them a trial head-to-head against the Genzyme product. In order to obtain FDA approval, it had to show that its product was not inferior to that of Genzyme, which it did. The company is now carrying out two further trials for marketing purposes and to obtain insurance coverage at the desired price.
Protalix has signed a marketing agreement with Italian company Chiesi, which will save direct costs and the price of inexperience, but will make it wholly dependent on the company. Protalix has already experienced that dependence, on Pfizer, with which it signed a marketing agreement for its previous product, for Gaucher Disease but which lost enthusiasm for the market. Chiesi is a mid-size company, and Protalix’s product is apparently important in its plans.
Protalix projects maximum annual revenue from the product of $150-200 million. It will manufacture it at its plant in Karmiel, where it produces its Gaucher treatment.
The company has $20 million cash, which will last it until the final quarter of 2023. If it obtains FDA approval, it will apparently receive a milestone payment from Chiesi, and will start producing for it. If not, and if it does not raise capital, it will be liable to get into financial difficulties, even though it has additional drugs in its pipeline. In its conference call, the analysts showed greater interest in the unique products in the pipeline than in the product about to be launched.
BioLineRx: Saving hospitalization and money in treating blood cancer
Founded: 2003
CEO: Philip Serlin
Field: Buying drugs and developing them
Market cap: $40m
Like Gamida Cell, BioLine too is aiming at the blood cancer treatments market, but its product is designed for patients undergoing transplants of their own bone marrow. The product assists in mobilization of cells from the patient, and will enter a market in which there are already two similar products. Most patients receive a generic product called GCSF, but this requires more than the single treatment that BioLine offers, so that BioLine’s product saves hospital time and money. In an economic benefit study, BioLine showed a $19,000 advantage versus GCSF, and a $30,000 advantage versus a combined treatment of GCSF with another product made by Genzyme that reduces the number of treatments to two but costs more. This is before taking into account the price of BioLine’s product, which has yet to be set.
Since BioLine believes that the advantage of its product is clear, it has decided to produce and market it independently. The company estimates potential sales in the US at $360 million annually. Like Gamida Cell, it is aiming at the 70 transplant centers where 80% of the treatments are carried out, and so will presumably need the same marketing manpower, 20-30 salespeople. Like the other companies, it will need insurance company cover before it sees significant revenue from the product.
BioLine recently raised $55 million, which should enable it to launch the product once it obtains approval.
Published by Globes, Israel business news - en.globes.co.il - on December 29, 2022.
MERRY CHRISTMAS ~ To You And Your Family & With LOVE To All ~
https://theexoduseffect.com/
Christmas offer us a wonderful message Emmanuel
GOD with us alive He breath our air, felt our pain -
knew our sorrows with the Father and the Spirit King.








.jpg)
.jpg)
.jpg)
.jpg)
.jpg)






















FDA grants new approval for essential tremor treatment from Insightec
DECEMBER 20, 2022 BY SEAN WHOOLEY
The Exablate Neuro system. [Image from Insightec’s website]
Insightec announced today that it received additional FDA approval for treating essential tremor with its Exablate Neuro platform.
Essential tremor commonly affects both sides of the body, Insightec noted in a news release. The new approval allows appropriate patients to receive treatment on their second side at least nine months after treatment of the first side.
Exablate Neuro uses focused sound waves safely guided by MRI. It provides tremor treatment to patients with medication-refractory essential tremor and Parkinson’s disease.
“This FDA approval is a very important milestone for us and demonstrates our unwavering commitment to expanding the treatment options for people living with essential tremor,” said Dr. Maurice R. Ferré, CEO and chair of the board of directors at Insightec. “It’s very common for patients who’ve benefited from tremor reduction from the first side treatment to ask about having the second side treated. This approval paves the way for them to do that.”
How Insightec garnered its latest FDA approval
Insightec said its sponsored study included 51 patients across seven U.S. centers. It evaluated Exablate Neuro for safety and efficacy endpoints out to six months.
Data demonstrated a highly significant reduction in tremor following treatment of the patients’ second side. Results proved immediate and sustained through at least six months of follow-up. According to Insightec, this falls in line with results from the treatment of the first side.
Insightec said the study demonstrated a significant improvement in functional disability. This suggests a clinically meaningful impact on activities of daily living. That includes eating, drinking and writing.
Patients reported the majority of adverse events in the study as mild. Exablate Neuro demonstrated a similar safety profile in treating the second side as it did for the first side.
“We are happy to have an option for patients with essential tremor to ultimately receive focused ultrasound treatment on both sides of the brain,” said Dr. Michael G. Kaplitt, professor and executive vice-chair of neurological surgery, New York Presbyterian Hospital-Weill Cornell Medicine, and principal investigator of this study. “The investigators are working with Insightec to share the full results of the study with the professional community in an academic publication to further help practitioners guide their patients going forward.”
Insightec Announces Milestone of First Patients Enrolled in the Pivotal LIBERATE Clinical Trial (Liquid Biopsy with Low Intensity Ultrasound in Brain Tumors)
https://finance.yahoo.com/news/insightec-announces-milestone-first-patients-140000780.html
Breakthrough therapy uses focused ultrasound to advance diagnosis and monitoring in glioblastoma
HAIFA, Israel & MIAMI, Dec. 15, 2022 /CNW/ -- Insightec, a global healthcare company dedicated to using acoustic energy to transform patient care, announced today the first patients have enrolled in the pivotal LIBERATE clinical trial (Liquid Biopsy with Low Intensity Ultrasound in Brain Tumors). Enrollment in LIBERATE marks the start of an international effort to assess safety and efficacy of using Insightec's focused ultrasound (FUS) platform to temporarily disrupt the blood brain barrier (BBB) to enable liquid biopsies in patients with glioblastoma, the most common primary malignant brain tumor in adults.
The first patients were enrolled in the US and Canada at Mayo Clinic (Drs. Terry Burns MD PhD and Timothy Kaufmann MD MS) and at Sunnybrook Health Sciences Centre (Dr. Nir Lipsman MD PhD).
"Our team at Mayo Clinic is excited to have enrolled the first patient in this pivotal trial," said neurosurgeon Terry Burns MD PhD. "If successful, this work has the potential to substantially decrease the risk of obtaining the initial diagnosis. Importantly, FUS can be repeated non-invasively, allowing a rare molecular window into individual patient's brain tumors as they evolve during treatment."
"We are thrilled to take part in this first global pivotal trial of FUS as a novel, less invasive approach for brain tumor liquid biopsy," said neurosurgeon Nir Lipsman MD PhD. "This milestone represents a key expansion in applications of this FUS platform, with therapeutic potential ranging from improved diagnostics to enhanced drug delivery."
"Liquid biopsy is a new approach for noninvasive cancer diagnosis and monitoring in brain tumors," said Dr. Achal Singh Achrol MD, Chief Medical Officer of Insightec. "Despite remarkable progress in other cancers, liquid biopsy has had limited success in brain tumors due to the presence of the blood brain barrier (BBB). This pivotal trial is designed to assess for the first time the clinical benefit of using low-intensity focused ultrasound (LIFU) to transiently increase the permeability of the BBB and allow passage of region-specific biomarkers to enable incisionless liquid biopsy in brain tumors."
In the study, blood samples are collected before and after disruption of the BBB to assess for FUS-enabled increases in circulating free DNA (cfDNA). The study also assesses correlation between mutation profiles in the released cfDNA, i.e. circulating tumor DNA (ctDNA), with the mutation profiles in the tumor tissue obtained during standard-of-care invasive surgical resection and/or biopsy.
"This is a potential game-changer in neuro-oncology and would represent a paradigm shift in the treatment and evaluation of patients with brain tumors," said study Principal Investigator Dr. Manmeet Ahluwalia, MD, MBA, Chief of Solid Tumor Medical Oncology, Deputy Director and Chief Scientific Officer at Miami Cancer Institute, part of Baptist Health South Florida.
"Insightec is committed to supporting these leading physician-scientists at these world-renowned institutions as they seek to advance the field of incisionless neurosurgery," said Dr. Maurice R. Ferré MD, CEO and the Chairman of the Board of Directors at Insightec. "All of us at Insightec are proud to be supporting the LIBERATE investigators and launch of this pivotal trial is an important step forward in the fight against glioblastoma. Our ultimate goal is, as always, to positively impact patient lives."
For further information about LIBERATE and to find enrolling locations please visit: https://clinicaltrials.gov/ct2/show/NCT05383872
In addition to the LIFU investigational device in the LIBERATE trial, Insightec also develops and manufactures FDA-approved platforms using high-intensity focused ultrasound (HIFU). These include the Exablate Neuro, approved for the treatment of Essential Tremor and Parkinson's Disease, and Exablate Prostate, which received FDA 510K clearance for ablation of prostate tissue. (For detailed information about approved indications, see our website at https://insightec.com/regulatory-approvals/). Insightec systems are treating patients in commercially-approved indications at medical centers in over 30 countries. In addition, Insightec continues to support clinical trials with partner institutions across a range of other conditions, including Alzheimer's disease, opioid abuse, neuropsychiatry, epilepsy and other cancers.
About Insightec
Insightec is a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy. The company's Exablate Neuro platform focuses sound waves, safely guided by MRI, to provide tremor treatment to patients with medication-refractory Essential Tremor and Parkinson's Disease. Research for future applications in the neuroscience space is underway in partnership with leading academic and medical institutions. Insightec is headquartered in Haifa, Israel, and Miami, with offices in Dallas, Shanghai, and Tokyo.
Follow us on Facebook, LinkedIn and Twitter or visit www.insightec.com for more information.
jedijazz Thanks Prayers Wanted; /; Is this what the libs American Dream is all about???
https://newspunch.com/klaus-schwab-god-is-dead-and-the-wef-is-acquiring-divine-powers/
https://www.zerohedge.com/geopolitical/mask-wefs-klaus-schwab-declares-china-role-model
20 MILLION DEAD FROM VAX SHOTS
WATCH

:https://www.bitchute.com/video/9uMnzp6yMVXt/
HUMAN SEX CHILD TRAFFICKING
WATCH

https://www.bitchute.com/video/BWDGTM6M6sbw/
BRICS NATIONS STOCKING UP ALL THE GOLD AND PREPPING FOR AMERICAS COLLAPSE PART 2
WATCH

https://investorshub.advfn.com/secure/post_reply.aspx?message_id=170533123
BEWARE; BREAKING: New Vaccine data changes everything | Redacted with Clayton Morris Redacted
1.43M subscribers










jedijazz Thanks & Happy Thanksgiving To All
xxxx xxxx
20 MILLION DEAD FROM VAX SHOTS
WATCH

:https://www.bitchute.com/video/9uMnzp6yMVXt/
HUMAN SEX CHILD TRAFFICKING
WATCH

https://www.bitchute.com/video/BWDGTM6M6sbw/
BRICS NATIONS STOCKING UP ALL THE GOLD AND PREPPING FOR AMERICAS COLLAPSE PART 2
WATCH

https://investorshub.advfn.com/secure/post_reply.aspx?message_id=170533123
BEWARE; BREAKING: New Vaccine data changes everything | Redacted with Clayton Morris Redacted
1.43M subscribers









$ASPI 11M Float #Shortages of global supplies of molybdenum-99 (Mo-99) and lutetium-177 (Lu-177) may be inevitable.
MAJOR ADVANTAGES OF PRODUCTION OF ISOTOPES by ASP

FACT: There are only five nuclear reactors in the world capable of producing the Mo-99, an isotope used to produce technetium-99 (Tc-99m). Tc-99m is used in approximately 50,000 diagnostic medical procedures every day in the U.S. for advanced diagnostic imaging tests and limits in production directly impact patient care and access.
$ASPI Website: https://aspisotopes.com/medical-isotopes/
https://www.floatchecker.com/stock?float=ASPI
https://finviz.com/quote.ashx?t=ASPI&p=d
Non-invasive brain disease treatment center established in Shanghai
https://www.shine.cn/news/metro/2211213122/
By: Cai Wenjun
A center for non-invasive brain disease treatment was established at Shanghai International Medical Center on Monday, offering innovative treatment for patients with nervous system disorders like essential tremor or Parkinson's disease.
By introducing Israeli pharmaceutical company Insightec's MR-guided focused ultrasound (MRgFUS) system, the brain center said patients can enjoy a quick, safe, precise and non-invasive treatment and will be able to see the effects on site.
Medical experts said the treatment, which was approved by China's National Medical Products Administration in 2021, has a strong potential to be introduced to more fields like mental disease, degenerative disease and pain by relieving symptoms and discussed its future development during Monday's ceremony.
Wang Yizheng, a leading neurologist and an academic at the Chinese Academy of Sciences, said nervous system disease is highly complicated and patients usually suffer long-term effects.
"With the rising number of elderly people in China, there will be rising demand and burden for diseases like Parkinson's. The introduction and promotion of innovative therapy is very meaningful to patients by offering low-risk and high-precision treatment," he said.
Dr Sun Bomin, chief expert of the new brain disease center, said MRgFUS is a new-generation medical equipment for neuromodulation, which combines therapeutic acoustic ultrasound waves with continuous magnetic resonance imaging (MRI) guidance to guide physicians to accurately visualize, plan, guide, monitor and control treatment while the ultrasound acoustic energy destroys the targeted tissue in the brain by thermal ablation.
"The patient is awake and able to give response to doctors during the process. Usually, they can leave the hospital on the day after finishing the treatment," Sun said.
Officials from Shanghai Zhangjiang Science City said Shanghai International Medical Center is one of the first to join the zone and the new brain disease center can boost the high-tech park's ability regarding medical practice and scientific innovation.
Insightec's Focused Ultrasound for Essential Tremor Study Results Demonstrated Sustained and Significant Tremor Improvement Maintained Out to 5 Years
The results, published in the Journal of Neurosurgery, show improvement without any progressive or delayed complications.
HAIFA, Israel and MIAMI, Nov. 7, 2022 /PRNewswire/ -- Insightec, a global healthcare company dedicated to using acoustic energy to transform patient care, today announced the publication of the largest prospective, long-term follow-up study of unilateral MRI-guided focused ultrasound (MRgFUS) thalamotomy for essential tremor to date.
This randomized, controlled, multi-center study, recently published in the Journal of Neurosurgery, describes the long-term safety and efficacy of unilateral MRI-guided focused ultrasound (MRgFUS) thalamotomy for medication-refractory essential tremor using the Exablate System.
The data showed the durability of tremor reduction in patients' (n=40) treated hand, which remained significantly improved at five years. This 73.1% improvement in tremor severity (CRST Part A) is considered clinically meaningful and is consistent with previously published data from this cohort.
Importantly, quality of life (QUEST) and functional disability (CRST Part C) also remained sustained from baseline. In addition, no new adverse events related to the procedure were reported from the 12-month timepoint to the last follow-up at 5-years. The publication shows that unilateral Exablate MRgFUS thalamotomy shows sustained and significant tremor improvement at five years with an overall improvement in quality-of-life measures and without any progressive or delayed complications.
"We are happy to see the results from this study. The sustained tremor improvement over five years is a testament to the tireless work of our doctors and partner treatment centers," said Maurice R. Ferré, MD, CEO and the Chairman of the Board of Directors at Insightec. "This data shows that using MRI-guided focused ultrasound to treat essential tremor is safe and effective. We will continue future progress in finding other treatment options for patients for whom focused ultrasound can be a life-altering option."
The study was sponsored by Insightec and conducted at eight centers in the US, Canada, South Korea, and Japan.
About Insightec
Insightec is a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy. The company's Exablate Neuro platform focuses sound waves, safely guided by MRI, to provide tremor treatment to patients with medication-refractory Essential Tremor and Parkinson's Disease. Research for future applications in the neuroscience space is underway in partnership with leading academic and medical institutions. Insightec is headquartered in Haifa, Israel, and Miami, with offices in Dallas, Shanghai, and Tokyo.
NEWS THE RETURN OF NETANYAHU, ELON'S TWITTER CENSORS, DEFINITION OF ANTI-SEMITISM, KYRIE CONDEMNED | KMN
WATCH

https://www.bitchute.com/video/JcpJIA9wDx2z/
GREAT WATCH LIVE: PRESIDENT DONALD J. TRUMP HOLDS RALLY IN MIAMI, FL – 11/6/22
by RSBN

Sunday, November 6, 2022: Join the RSBN broadcast crew LIVE from Miami, FL
for all day coverage of President Donald J. Trump’s “Get Out the Vote Rally” with
Senator Marco…
https://www.rsbnetwork.com/category/video/donald-trump/
NWO The blame game -



GENETIC ENGINEERING BIOWEAPON EXPOSED: NANO-STRUCTURES IN THE DEATH JAB
WATCH

https://www.bitchute.com/video/xs7kfJ9w1Dbw/
BEWARE; A number of faith leaders have insisted that Jesus himself would get the jab, were
He here today.
But not once did Jesus advise people to rely on the tools — or
word — of man. Instead, He warned of a future time when even the elect will fall
prey to a grand deception
Hindsight, scientific evidence and statistical data allow us to conclude that many
faith leaders led their flocks astray.
The COVID shot does not protect you from
infection and death, nor does it protect anyone else. It was all a lie, and excess
death rates skyrocketed after the rollout of the COVID shots
https://articles.mercola.com/sites/articles/archive/2022/10/31/faith-leaders-promotes-covid-vaccine.aspx?ui=f31c52a3f8937693fc4daf3324ea789acdff1bba8274a31b5972577e0424a49d&sd=20150920&cid_source=dnl&cid_medium=email&cid_content=art1HL&cid=20221031&cid=DM1276919&bid=1633441104
Russia and Iran, New "Axis of Evil," Emboldened by Biden Administration
by Majid Rafizadeh
October 29, 2022 at 5:00 am
https://www.gatestoneinstitute.org/19051/russia-iran-evil-emboldened
ROUBINI WARNS OF IMMINENT DOLLAR CRASH
WATCH
https://www.bitchute.com/video/FuUBZ2VNzBeh/
BRICS | Is the Collapse of the Dollar Around the Corner? 15 Questions We All
Need to Be Asking About the Future Value of the U.S. Dollar (Andrew Sorchini
with Clay Clark)
https://www.brighteon.com/111d6525-f68c-4df1-9186-cc4a586af0f4
DOLLAR TO COLLAPSE! - SAUDI ARABIA TO LEAVE DOLLAR FOR BRICS! - MASSIVE POWER SHIFT!
WATCH

https://www.bitchute.com/video/O9ky2nStDghK/
Thanks Al44
ARIS Great Gold Producer;
https://seekingalpha.com/article/4527727-aris-gold-gcm-mining-merger-makes-sense
Aris Mining Corporation (Aris Mining) (TSX: ARIS) (OTCQX: TPRFF)
Great Gold, Global Population And Propaganda

https://kingworldnews.com/gold-global-population-and-propaganda/
$370 Silver Target, Plus Major Economic Trend Forecasts Issued By Gerald Celente
October 26, 2022
https://kingworldnews.com/370-silver-target-plus-major-economic-trend-forecasts-issued-by-gerald-celente/
Aris Mining Corporation (Aris Mining) (TSX: ARIS) (OTCQX: TPRFF) Highest
Close in 2 Weeks on the ARIS;
The $3.04 close is the highest since October 13th. Maybe the jaded seller
lamenting the loss of the dividend is sold out? With the US dollar index closing
below its 50 dma today, Gold may continue higher and bring a lift to the PM sector.
There are 556,000 shares short as of October 14th (per yahoo finance), which is 3
days of average trading volume. If they were smart, the covered under $3.
We should see the 3Q earnings report next month. I am hoping for a definitive
gold production forecast for 2023 and 2024. Marmato underground operations
and the mill capacity up grade at Segovia from 1500 to 2000 tpd, are near term
drivers for the company.[/b[
by tobinator01 (sth)
Aris Mining appoints Mónica de Greiff as a member of the Board of Directors
October, 04, 2022
VANCOUVER, BC, Oct. 4, 2022 /PRNewswire/ -
Aris Mining Corporation (Aris Mining) (TSX: ARIS) (OTCQX: TPRFF) announces
that, effective October 1, 2022, Mónica de Greiff was appointed as an
independent member of the Board of Directors, and as Chair of the Sustainability
Committee of the Board.
ARIS Mining (CNW Group/Aris Mining Corporation)
Mónica de Greiff was a member of the GCM Mining board of directors from 2018 to 2020, when she left to accept the position of Colombian Ambassador to Kenya. She has held positions in both the public and private sectors, including as Minister of Justice for the Republic of Colombia and Vice Minister of Mines and Energy. Ms. de Greiff is also a former member of the Board of Directors of the United Nations Global Compact, the world's largest corporate sustainability initiative.
Ian Telfer, Chair of Aris Mining, stated "I am delighted to welcome Mónica to our Board of Directors. She brings considerable experience within Colombia and in the highly valued and important area of sustainability. We look forward to her contributions to our business as we continue to grow and enhance our commitment towards ESG."
About Aris Mining
Aris Mining is a Canadian company led by an executive team with a track record
of creating value through building globally relevant mining companies.
In Colombia, Aris Mining operates several high-grade underground mines at its
Segovia Operations and the Marmato Mine, which together produced 230,000
ounces of gold in 2021.
Aris Mining also operates the Soto Norte joint venture, where environmental
licensing is advancing to develop a new underground gold, silver and copper
mine. In Guyana, Aris Mining is advancing the Toroparu Project, a gold/copper
project with expected average gold production of 225,000 per year over the life of
mine. Aris Mining plans to pursue acquisition and other growth opportunities to unlock value creation from scale and diversification.
Aris Mining promotes the formalization of small-scale mining as this process
enables all miners to operate in a legal, safe and responsible manner that protects
them and the environment.
Additional information on Aris Mining can be found at
http://www.aris-mining.com
and www.sedar.com.
https://www.aris-mining.com/operations/operating-mines/segovia/overview/default.aspx
https://www.aris-mining.com/investors/events-and-presentations/default.aspx
https://www.aris-mining.com/news/news-details/2022/Aris-Mining-appoints-Mnica-de-Greiff-as-a-member-of-the-Board-of-Directors/default.aspx
CisionView original content to download multimedia:https://www.prnewswire.com/news-releases/aris-mining-appoints-monica-de-greiff-as-a-member-of-the-board-of-directors-301640112.html
SOURCE Aris Mining Corporation
VIEW ALL NEWS
GCM Mining Corp. (formerly Gran Colombia Gold)
(TSX: GCM / OTCQX: TPRFF)
Accretive Acquisition to Drive Valuation Higher
BUY
https://www.researchfrc.com/wp-content/uploads/2022/08/GCM-Aug-2022-Update-1.pdf?vgo_ee=TMO54WfXmKl1gA%2FTEb1O1%2FlMy%2BOWWuyaZunZiCXh6gI%3D
Current Price: C$3.35
Fair Value: C$10.09
Risk: 3
GCM Mining Corp. PowerPoint Presentation
https://s28.q4cdn.com/389315916/files/doc_downloads/2022/07/Aris-Gold-GCM-presentation-25-July-2022.pdf
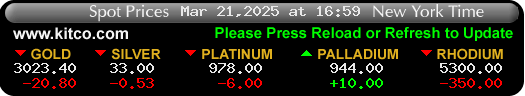
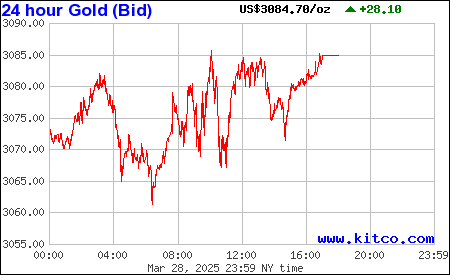
$In GOD We Trust - Real Money - AU Safety 6000yrs ![]() )
)

https://www.kitconet.com/images/quotes_7a.gif?1493417496003




https://www.kitco.com/images/live/silver.gif?0.8344882022363285
Gold & Silver is the only REAL Legal Tender -
by The Founding Fathers for your -
Rights, Liberty and Freedom -
http://www.biblebelievers.org.au/monie.htm
GOLD STANDARD; THE REAL LEGAL MONEY:
https://www.cs.mcgill.ca/~rwest/wikispeedia/wpcd/wp/g/Gold_standard.htm#:~:text=Advocates%20of%20a%20variety%20of,basis%20for%20a%20monetary%20system.
https://www.usdebtclock.org/
https://www.worldometers.info/coronavirus/country/us/
https://www.whatdoesitmean.com/index.htm
God Bless.America
https://www.silverdoctors.com/headlines/world-news/u-s-dollar-to-collapse-saudi-arabia-to-leave-the-dollar-for-brics-in-massive-power-shift/
CT Follow The Patriot Base;
https://rumble.com/c/KPB
Supreme Court Blocks Congress From Getting Trump's Tax Returns
Tyler Durden's Photo
BY TYLER DURDEN
TUESDAY, NOV 01, 2022 - 11:00 AM
Authored by Zachary Stieber via The Epoch Times,
The U.S. Supreme Court on Nov. 1 blocked Congress from obtaining former President Donald Trump’s tax returns.
Chief Justice John Roberts, a George W. Bush appointee, stayed an appeals court order that had ruled a congressional panel could gain access to the documents.
https://www.zerohedge.com/markets/supreme-court-blocks-congress-getting-trumps-tax-returns
Aris Mining Corporation (Aris Mining) (TSX: ARIS) (OTCQX: TPRFF)
Low Bargain Close -
We should see the 3Q earnings report Nov. I am hoping for a definitive
gold production forecast for 2023 and 2024.
Marmato underground operations and the mill capacity up grade at Segovia
from 1500 to 2000 tpd, are near term
drivers for the company.
Aris Mining appoints Mónica de Greiff as a member of the Board of Directors
October, 04, 2022
VANCOUVER, BC, Oct. 4, 2022 /PRNewswire/ -
Aris Mining Corporation (Aris Mining) (TSX: ARIS) (OTCQX: TPRFF) announces
that, effective October 1, 2022, Mónica de Greiff was appointed as an
independent member of the Board of Directors, and as Chair of the Sustainability
Committee of the Board.
ARIS Mining (CNW Group/Aris Mining Corporation)
Mónica de Greiff was a member of the GCM Mining board of directors from 2018 to 2020, when she left to accept the position of Colombian Ambassador to Kenya. She has held positions in both the public and private sectors, including as Minister of Justice for the Republic of Colombia and Vice Minister of Mines and Energy. Ms. de Greiff is also a former member of the Board of Directors of the United Nations Global Compact, the world's largest corporate sustainability initiative.
Ian Telfer, Chair of Aris Mining, stated "I am delighted to welcome Mónica to our Board of Directors. She brings considerable experience within Colombia and in the highly valued and important area of sustainability. We look forward to her contributions to our business as we continue to grow and enhance our commitment towards ESG."
About Aris Mining
Aris Mining is a Canadian company led by an executive team with a track record
of creating value through building globally relevant mining companies.
In Colombia, Aris Mining operates several high-grade underground mines at its
Segovia Operations and the Marmato Mine, which together produced 230,000
ounces of gold in 2021.
Aris Mining also operates the Soto Norte joint venture, where environmental
licensing is advancing to develop a new underground gold, silver and copper
mine. In Guyana, Aris Mining is advancing the Toroparu Project, a gold/copper
project with expected average gold production of 225,000 per year over the life of
mine. Aris Mining plans to pursue acquisition and other growth opportunities to unlock value creation from scale and diversification.
Aris Mining promotes the formalization of small-scale mining as this process
enables all miners to operate in a legal, safe and responsible manner that protects
them and the environment.
Additional information on Aris Mining can be found at
http://www.aris-mining.com
and www.sedar.com.
https://www.aris-mining.com/operations/operating-mines/segovia/overview/default.aspx
https://www.aris-mining.com/investors/events-and-presentations/default.aspx
https://www.aris-mining.com/news/news-details/2022/Aris-Mining-appoints-Mnica-de-Greiff-as-a-member-of-the-Board-of-Directors/default.aspx
CisionView original content to download multimedia:https://www.prnewswire.com/news-releases/aris-mining-appoints-monica-de-greiff-as-a-member-of-the-board-of-directors-301640112.html
SOURCE Aris Mining Corporation
VIEW ALL NEWS
GCM Mining Corp. (formerly Gran Colombia Gold)
(TSX: GCM / OTCQX: TPRFF)
Accretive Acquisition to Drive Valuation Higher
BUY
https://www.researchfrc.com/wp-content/uploads/2022/08/GCM-Aug-2022-Update-1.pdf?vgo_ee=TMO54WfXmKl1gA%2FTEb1O1%2FlMy%2BOWWuyaZunZiCXh6gI%3D
Current Price: C$3.35
Fair Value: C$10.09
Risk: 3
GCM Mining Corp. PowerPoint Presentation
https://s28.q4cdn.com/389315916/files/doc_downloads/2022/07/Aris-Gold-GCM-presentation-25-July-2022.pdf
$2.5 Quadrillion Disaster Waiting to Happen – Egon von Greyerz
By Greg Hunter On November 1, 2022 In Market Analysis 98 Comments

https://usawatchdog.com/2-5-quadrillion-disaster-waiting-to-happen-egon-von-greyerz/
$In GOD We Trust - Real Money - AU Safety 6000yrs ![]() )
)

https://www.kitconet.com/images/quotes_7a.gif?1493417496003




https://www.kitco.com/images/live/silver.gif?0.8344882022363285
Gold & Silver is the only REAL Legal Tender -
by The Founding Fathers for your -
Rights, Liberty and Freedom -
http://www.biblebelievers.org.au/monie.htm
GOLD STANDARD; THE REAL LEGAL MONEY:
https://www.cs.mcgill.ca/~rwest/wikispeedia/wpcd/wp/g/Gold_standard.htm#:~:text=Advocates%20of%20a%20variety%20of,basis%20for%20a%20monetary%20system.
https://www.usdebtclock.org/
https://www.worldometers.info/coronavirus/country/us/
https://www.whatdoesitmean.com/index.htm
God Bless.America
MUST SEE WATCH LIVE: PRESIDENT DONALD J. TRUMP HOLDS SAVE AMERICA RALLY IN ROBSTOWN, TX 10/22/22
by RSBN
https://www.rsbnetwork.com/video/watch-live-president-donald-j-trump-holds-save-america-rally-in-robstown-tx-10-22-22/
Saturday, October 22, 2022: Join the RSBN broadcast team LIVE from Robstown, TX for all day coverage of President Donald J. Trump’s Save America rally.
President Donald J. Trump, 45th President of the United States of America, will hold a rally in Robstown, Texas on Saturday, October 22, 2022, at 7:00PM CDT.
Saturday, October 22, 2022, at 7:00PM CDT
President Donald J. Trump, 45th President of the United States of America, delivers remarks in support of his unprecedented effort to advance the MAGA agenda by energizing voters and highlighting the slate of 33-0 Trump Endorsed America First candidates in the Great State of Texas.
Additional Special Guest Speakers will be Announced.
Venue:
Richard M. Borchard Regional Fairgrounds
1213 Terry Shamsie Blvd.
Robstown, TX 78380
Timeline of Events:
8:00AM – Parking Lots, Vendor Row, and Registration Open
2:00PM – Doors Open
4:00PM – Special Guest Speakers Deliver Remarks
7:00PM – 45th President of the United States Donald J. Trump Delivers Remarks
Entertainment and Concessions will be available throughout the day!
JULIE GREEN PROPHETIC WORD [REVERSALS ARE COMING] CHANGE IS COMING - TRUMP NEWS
Trump News Channel Published October 15, 2022
The Lords Day

https://rumble.com/v1p43dv-the-lords-day.html

https://rumble.com/v1o4gov-julie-green-prophetc-word-reversals-are-coming-change-is-coming-trump-news.html
OVERDUE FROM THE HAGUE. INTERNATIONAL TRIALS DAY ONE - NWO CRIMES AGAINST HUMANITY
WATCH

https://www.bitchute.com/video/GjEgSnACO93V/
Gold Standard Bargain; Aris Mining Corp.
https://www.aris-mining.com/operations/operating-mines/segovia/overview/default.aspx
https://www.aris-mining.com/investors/events-and-presentations/default.aspx
https://www.aris-mining.com/
Ted Butler: Stand up against market manipulation and make a difference
By Ted Butler
SilverSeek.com
Friday, October 14, 2022
If you are tired of witnessing silver (and gold) continuing to be manipulated in price, here's a no-cost, no-risk, high-potential return action you can take that will only involve a few minutes of your time. Quite literally, there's absolutely nothing to lose and quite a lot of potential good to be had.
The Commodity Futures Trading Commission is the taxpayer-funded federal commodities regulator whose main mission is to prevent and root out manipulation and protect the public. Four of the five commissioners have been in office for little more than six months and it's not clear that they are even aware that silver has been manipulated in price on the Comex.
Here is your opportunity to ensure that this is an issue they should be concerned about. Please take the time to copy and paste the letter below and email it to addresses listed. If you would prefer using your own name and not mine, you have my permission to do so. ...
... For the remainder of the commentary:
https://silverseek.com/article/stand-and-make-difference
https://www.aris-mining.com/operations/operating-mines/segovia/overview/default.aspx
https://www.aris-mining.com/investors/events-and-presentations/default.aspx
https://www.aris-mining.com/
GOD'S Money Bargain "Gold Standard Restoration Act" Would Peg Dollar To Gold At Fixed Price
28,398 views Oct 11, 2022
The Mother Of All Debt Bubbles Is About To
























.jpg)










.jpg)
.jpg)





M_P Putin's Genocide on Holocaust Memorial Day
https://www.gatestoneinstitute.org/18474/putin-genocide-on-holocaust-memorial-day
Here's another shocker...
Political interference at Health and Human Services (NIH, CDC, FDA, ASPR)
https://rwmalonemd.substack.com/p/political-interference-at-health?s=r
What up here ???
EXCLUSIVE: Hundreds of CDC Employees Haven’t Received a COVID-19 Vaccine
Nearly 400 employees at the U.S. Centers for Disease Control and Prevention (CDC) have not received a COVID-19 ...
https://www.theepochtimes.com/exclusive-hundreds-of-cdc-employees-havent-had-a-covid-19-vaccine_4429636.html?utm_source=News&utm_campaign=breaking-2022-04-28-2&utm_medium=email&est=ZW2vW6aPmZAweXLTlDrBR%2F6O%2FEIc0odN9E3RhbcnoabcVEvV2lyYoO3M0nwYWGnT9Q%3D%3D
https://www.rt.com/shows/the-world-according-to-jesse/544353-pentagon-civilian-casualties-exposed/
The World According to Jesse
https://www.rt.com/shows/the-world-according-to-jesse/
https://www.rt.com/shows/the-world-according-to-jesse/547709-julian-assange-small-victory/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=168506668
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=168682323
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=168682697
INSIGHTEC RECEIVES FDA IDE APPROVAL AND “BREAKTHROUGH” DESIGNATION FOR NSCLC BRAIN METS STUDY AND IDE APPROVAL FOR LIQUID BIOPSY STUDY
https://insightec.com/insightec-receives-fda-ide-approval-and-breakthrough-designation-for-nsclc-brain-mets-study-and-ide-approval-for-liquid-biopsy-study/
Designation and approvals mark key milestones in advancing Insightec Blood Brain Barrier (BBB) clinical roadmap using Acoustic Therapy
HAIFA, Israel & MIAMI, Florida, March 9, 2022 — Insightec, a global healthcare company dedicated to using acoustic energy to transform patient care, today announced that it has received FDA approval of two pivotal investigational device exemptions (IDE) for its Exablate Neuro system – one for primary non-small cell lung cancer (NSCLC) that has metastasized to the brain in conjunction with standard-of-care Keytruda®, and one for enhanced efficacy of liquid biopsy for recurrence monitoring of patients with primary brain cancer. The FDA has also granted “Breakthrough Device” designation for the NSCLC treatment, helping to expedite its development and review.
Insightec plans to launch the LIMITLESS study (NSCLC) to assess the clinical benefit of using noninvasive, low-intensity focused ultrasound combined with systemic immunotherapy in the treatment of patients with brain metastases from lung cancer.
“The use of low-intensity focused ultrasound (LIFU) with microbubbles to open up the blood brain barrier (BBB) is an exciting advance in neuro-oncology that holds promise to become a paradigm shift in the treatment of patients with brain tumors,” said study Principal Investigator Dr. Manmeet Ahluwalia, MD, MBA, Chief of Solid Tumor Medical Oncology, Deputy Director and Chief Scientific Officer at Miami Cancer Institute, part of Baptist Health South Florida. “The unique synergy of LIFU BBB opening for improved drug delivery, neoantigen release and immune priming is a potential game-changer in our field that will help us build on current treatment options available for these patients.”
Liquid biopsy is a new approach for noninvasive cancer subtyping, treatment selection, monitoring residual disease, early identification of treatment responders versus non-responders, and assessment of tumor progression versus pseudoprogression.
“Despite remarkable progress in other cancers, liquid biopsy has had limited success in brain tumors due to the presence of the blood brain barrier, which limits signal detection in the bloodstream,” said Dr. Achal Singh Achrol, MD, FAANS, Chief Medical Officer of Insightec. “Preclinical and early human studies have demonstrated that low-intensity focused ultrasound (LIFU) can transiently increase the permeability of the blood brain barrier and allow passage of region-specific biomarkers to enable noninvasive liquid biopsy in brain tumors. This pivotal clinical trial will investigate for the first time the clinical benefit to this approach as a novel alternative to invasive neurosurgical biopsies.”
“Insightec continues to collaborate with leading researchers to advance the use of acoustic therapy in the brain,” said Maurice R. Ferré, MD, Insightec CEO and Chairman of the Board. “This work focuses on safely opening the blood brain barrier using low frequency acoustic energy and has the potential to transform current therapeutic and diagnostic approaches for conditions like brain cancer. Our ultimate goal is, as always, to positively impact patient lives.”
The Exablate Neuro device is already FDA approved for the treatment of medication-refractory Essential Tremor and Parkinson’s Disease. At the close of 2021 there were 42 medical centers in the United States using the Insightec Exablate Neuro device to treat patients with these conditions. The Insightec Exablate Prostate system has received FDA 510K clearance for ablating prostate tissue with high intensity Focused Ultrasound.
About Insightec
Insightec is a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy. The company’s Exablate Neuro platform focuses sound waves, safely guided by MRI, to provide tremor treatment to patients with medication-refractory Essential Tremor and Parkinson’s Disease. Research for future applications in the neuroscience space is underway in partnership with leading academic and medical institutions. Insightec is headquartered in Haifa, Israel, and Miami, with offices in Dallas, Shanghai, and Tokyo.
Follow us on Facebook, LinkedIn and Twitter or visit www.insightec.com for more information.
Exablate MR-guided focused ultrasound launched in US prostate cancer market
February 3, 2022
Urology Times staff
The first commercial patient in the United States has been treated for prostate disease with the Exablate Prostate system, according to Insightec, the developer of the technology.1
The patient received the treatment at Stanford Health Care, which had previously participated in the clinical trials examining the efficacy and safety of Exablate.
The FDA granted Exablate 510K clearance in 2021 for treating prostate tissue with high-intensity magnetic resonance (MR)–guided focused ultrasound (MRgFUS). The 510(k) clearance designation allows a company to market a medical device in the United States if the company can prove that its device is “substantially equivalent” to a device already being legally marketed.2
"We are deeply committed to continuing to transform patient care with minimally invasive, innovative surgical procedures. The Exablate Prostate system will help support a better quality of life for people diagnosed and living with prostate disease," Maurice R. Ferré MD, Insightec CEO and Chairman of the Board of Directors, stated in a news release.
Clinical support
Clinical findings from a phase 2b study (NCT01657942) supporting the use of Exablate MRgFUS in men with intermediate-risk prostate cancer were shared at the 2021 AUA Annual Meeting.3 Overall, the study included 101 men who were treated at 7 academic centers and 1 private practice.
The average patient age was 63 years and 86% of the cohort was White. The median PSA was 5.7 ng/mL. Eighty-four patients had at least clinical stage T1c disease. Seventy-eight percent of patients were in grade group 2 and 22% were in grade group 3.
At 24 months, 88% of men had no evidence of grade group 2 or 3 prostate cancer in the treatment area according to targeted prostate biopsy. Additionally, 60% of men had no evidence of grade group 2 or 3 disease anywhere in the prostate gland, including outside of the treatment area. Serum PSA levels decreased by almost 50% after treatment and stabilized after 6 months.
Erectile function scores were slightly worse at 24 months compared with baseline (HR, -3.5; 95% CI, -5.4 to -1.6). Ten (13%) reported no sexual activity and 81% of patients achieved erections adequate for intercourse. Lower urinary tract symptoms were similar at baseline and 24 months (mean score difference, 1.1; 95% CI, 0.33-1.8).
“Overall, approximately 75% of men were able to achieve functional reactions after treatment at 24 months without starting a new erectile function medication,” Behfar Ehdaie, MD, MS, a urologic surgeon at Memorial Sloan Kettering Cancer Center and Exablate trial PI, said at the time of the AUA presentation of the results. “No patients reported urinary incontinence and the estimated probability of urinary continence over time was excellent.”
References
1. First US commercial patient treated for prostate disease with Insightec incisionless focused ultrasound system. Published online February 3, 2022. Accessed February 3, 2022. https://prn.to/3AV92VY.
2. 510(k) Clearances. US Food and Drug Administration. Accessed August 27, 2021. https://www.fda.gov/medical-devices/device-approvals-denials-and-clearances/510k-clearances
3. Ehdaie B, Tempany C, Holland F, et al. MRI-guided focused ultrasound (MRgFUS) focal therapy for intermediate-risk prostate cancer: final results of a phase IIb multicenter study. Presented at: 2021 American Urological Association Annual Meeting; September 10-13, 2021; virtual. Abstract LBA02-12.
Related Content:
Prostate Cancer
Emerging Therapies for Metastatic Castration-Resistant Prostate Cancer
Sabizabulin for Metastatic Castration-Resistant Prostate Cancer
Hollings patient advocates for screenings after two battles with prostate cancer
Emerging Therapies for Metastatic Castration-Resistant Prostate Cancer
January 28, 2022
Robert Dreicer, MD
UroView | Second-Line Treatment Options in Advanced Prostate Cancer
Robert Dreicer, MD, describes the future of metastatic castration-resistant prostate cancer.
Play Video
Robert Dreicer, MD: I am often asked to sort of speculate where [I think] therapeutic management of mCRPC [metastatic castration-resistant prostate cancer] is going, and I think put simply it’s going to get complicated. It’s already complicated. It’s a very heterogeneous disease; we all understand that, because of the leftward earlier movement of the therapies we used for mCRPC, there are gaps. There are a number of drugs in the pipeline and there are a number of drugs that may see regulatory approval early in 2022, including lutetium177-PSMA-617. There are also PARP [poly adenosine diphosphate-ribose polymerase]/ARI [alpha reductase inhibitor] combinations for which level 1 evidence is forthcoming. We know that certain patients with DDR [DNA damage response and repair] mutations need to be targeted differently at least today, but the potential for “BRCA-ness,” or increasing synthetic lethality with a combination of a PARP and an ARI, may open up a very interesting time frame. There are other PSMA [prostate-specific membrane antigen] therapeutics coming, both in terms of radioactive materials including alpha particles, as well as a whole generation of PSMA-targeted immunomodulatory therapies—CAR [chimeric antigen receptor] Ts, biospecifics, etc. I think what we’re going to increasingly see is the segmentation hopefully based on some biomarker to begin to deliver different sequences to specific groups of patients going forward. We’re not there. We need better therapies, and again, at the end of the day, we’re not curing mCRPC, we need to move to a paradigm where that becomes an option for a subset of patients as these therapies come online.
Transcript edited for clarity.
Related Content:
Second-Line Treatment Options in Advanced Prostate CancerProstate Cancer
Hollings patient advocates for screenings after two battles with prostate cancer
New Hollings study reveals barriers, presents solutions to improve prostate cancer screening among Black men
Hollings' SC AMEN Program for prostate cancer screening launches with enthusiastic support
Sabizabulin for Metastatic Castration-Resistant Prostate Cancer
January 28, 2022
Robert Dreicer, MD
UroView | Second-Line Treatment Options in Advanced Prostate Cancer
A prostate cancer expert explains the potential advantages of sabizabulin, a new treatment for metastatic castration-resistant prostate cancer currently in a phase III clinical trial.
Play Video
Robert Dreicer, MD: Among the drugs that are in trials now, there is a drug that I have been asked about as I have a relationship with the ongoing phase 3 [trial] and that’s sabizabulin, which is an oral cytoskeletal disruptor. It probably disrupts the cytoskeleton in a number of different mechanisms. It’s not taxane; it’s not a taxane-like drug, although it has similar targets. This oral agent has been in phase 1/2 trials. We have data from a phase 1B study that was led by Dr Mark Markowski of Johns Hopkins Hospital [Baltimore, MD] and this data was presented at ASCO [American Society of Clinical Oncology annual meeting] last year and no doubt will likely be updated at coming meetings. What we know about this agent is it’s an oral agent and it’s relatively well tolerated. Its toxicities tend to be GI [gastrointestinal], nausea, and some diarrhea but at the dose being studied in phase 3, it is a relatively well-tolerated therapy. What we've seen so far in this phase 1B study, which was about 30 patients with mCRPC [metastatic castration-resistant prostate cancer] that had progressed on at least 1 ARI [alpha reductase inhibitor] is that there's some evidence of antitumor activity that’s manifested both in seeing some soft tissue responses, some PSA [prostate-specific antigen] declines, and a couple of these patients have been on a drug for a long period of time, more than a year, approaching 2 years. My perspective on this agent is that there’s activity, it strikes me as almost a static agent, again that’s me speaking, that’s not data that we have yet, but in terms of the ability to have a drug on-board and have the ability to alter the natural history. We’re not seeing a dramatic resolution of disease, but we are seeing people who are able to tolerate this agent for prolonged periods of time and have antitumor activity. It’s an interesting compound because of the data seen to date. There is an ongoing phase 3 trial to test this drug further.
Sabizabulin is in a phase 3 trial entitled VERACITY, and this is a relatively small phase 3 trial that is randomizing patients to receive sabizabulin vs the alternative ARI. Eligibility, mCRPC patients who’ve progressed on an ARI, only minimal taxane prior exposure was allowed, so standard therapy in the castrate-sensitive or metastatic CRPC setting is not allowed, so this is really an early study of ARI progressing patients. The primary end point in this trial; radiographic progression-free survival. The eligibility is otherwise relatively straightforward, encompassing the issue of prior treatment. This study is open at a number of sites both in the U.S. and in Europe, and hopefully, it’s going to accrue relatively rapidly and give us an understanding of the utility of sabizabulin in mCRPC.
If sabizabulin happens to demonstrate activity, meaning meeting its primary end point of radiographic progression-free survival, and would eventually receive regulatory approval, this is a drug that you could also perceive to be used relatively early on in the disease course. It’s being tested in patients who have not been particularly heavily treated, these are ARI progressors, and so these patients are not uncommon. It appears to be a relatively well-tolerated drug, given the fact that there are a number of patients in the early phase 1 studies that have tolerated the drug for long periods of time with disease control. You could see this agent being used exactly in the clinical setting in which it’s being tested, as a drug that may provide long-term disease control with a relatively good quality of life. Now, obviously, we’re not there yet. We need to see. We need to finish phase 3, get the data, and then assess the safety, but this is a drug that you could see being used relatively early in the disease course among a number of clinicians—medical oncologists, urologists— who manage advanced disease and that population of clinicians.
Transcript edited for clarity.
Related Content:
Second-Line Treatment Options in Advanced Prostate CancerProstate Cancer
New Hollings study reveals barriers, presents solutions to improve prostate cancer screening among Black men
Hollings' SC AMEN Program for prostate cancer screening launches with enthusiastic support
Second-Line Therapy Options in Metastatic Castration-Resistant Prostate Cancer
x
Insightec cuts SPAC merger valuation
4 Jan, 2022 15:28
Gali Weinreb
Elbit Medical, a shareholder in the Israeli medical device company, reports that if the merger goes ahead it will be at a lower valuation than previously reported.
Israeli medical device company Insightec is the latest company to fall 'victim' to the weakness on the SPAC merger market on Wall Street. The company reported in October that it was in talks for a SPAC merger at a company valuation of $2 billion. Today, Elbit Medical (TASE: EMTC), which has a 3.1% stake in Insightec, notified the Tel Aviv Stock Exchange (TASE) that due to the market conditions, if the merger goes ahead, it will be at a lower valuation than reported in October. No further details were disclosed. Insightec has developed a device for minimally invasive surgery based on ultrasound.
Only last week, Israeli trading platform eToro reported that it had agreed to cut the valuation of its planned SPAC merger by 15% to $8.4 billion, after money. Other Israeli companies waiting to complete their SPAC mergers are Gett (formerly Get Taxi) and fintech company Pagaya.
Insightec was founded in 1999 and has developed and markets a non-invasive system for using focused MRI-guided ultrasound waves to destroy tissue, for treatment of non-Parkinson tremors, and to clear "blood-brain blockage" and tumors.
Insightec is controlled by US company Koch Disruptive Technologies (KDT), which has invested an overall $200 million for a 40% stake. In 2020, KDT led a $136 million financing round at a $1.3 billion valuation. In May 2021, Insightec's second largest shareholder York Capital sold its 7% stake at a company valuation of $1 billion to Peregrine Ventures and Israeli institutional investors Leumi Partners and Mor Investments. York remains with a 13% stake. Elbit Medical, once a dominant investor in the company, has sold most of its shares and remains with a 3.1% stake.
Insightec has recently received two important FDA clearances, in treating Parkinson's disease and prostate growths, which paves the way for future marketing possibilities. The company tends to see its revenue grow from quarter to quarter but still reports large losses.
Published by Globes, Israel business news - en.globes.co.il - on January 4, 2022
Sol-Gel Technologies Announces Positive Update Related to FDA Review Status of EPSOLAY®
https://finance.yahoo.com/news/sol-gel-technologies-announces-positive-120000521.html
FDA informs Sol-Gel that it intends to conduct a pre-approval inspection of the production site for EPSOLAY during the week of February 14th, 2022
NESS ZIONA, Israel, Dec. 21, 2021 (GLOBE NEWSWIRE) -- Sol-Gel Technologies, Ltd. (NASDAQ: SLGL), a dermatology company focused on identifying, developing and commercializing branded and generic topical drug products for the treatment of skin diseases, announced today that the Food and Drug Administration (FDA) has informed the Company that it intends to conduct a pre-approval inspection of the production site for the Company’s drug product, EPSOLAY®, a proprietary topical formulation of benzoyl peroxide, 5%, during the week of February 14th, 2022. EPSOLAY is under review by the FDA for the treatment of the inflammatory lesions of rosacea in adults.
“We are pleased that the FDA has informed us of the timing of its pre-approval inspection of the production site for EPSOLAY, which, since April 26, 2021, has been delayed due to COVID-19 related travel restrictions,” said Alon Seri Levy, Ph.D., Chief Executive Officer of Sol-Gel. “We believe that EPSOLAY has the potential to change the treatment landscape for adult patients suffering from inflammatory lesions of rosacea, a skin disorder that affects millions of people in the U.S. and expect that this development will bring us closer to the anticipated EPSOLAY approval and launch in the U.S. by our partner Galderma.”
About EPSOLAY
EPSOLAY is a topical cream containing benzoyl peroxide, 5%, under FDA review for the treatment of inflammatory lesions of rosacea in adults. EPSOLAY utilizes a proprietary technology to encapsulate benzoyl peroxide within silica-based microcapsules to create a barrier between the medication and the skin. The silica-based shell is designed to slowly release benzoyl peroxide over time to provide a favorable efficacy and safety profile. EPSOLAY is protected by granted patents until 2040.
BiondVax signs definitive agreements for development and commercialization of innovative nanosized COVID-19 antibody therapy with Max Planck Institute of Biophysical Chemistry and University Medical Center Göttingen, Germany
https://finance.yahoo.com/news/biondvax-signs-definitive-agreements-development-120700203.html
- Max Planck Institute of Biophysical Chemistry, a world leading research institute, together with University Medical Center Göttingen have developed unique nanosized antibodies (NanoAbs*) for the treatment of COVID-19 that exhibit significant competitive advantages over existing therapies;
- BiondVax to lead development and commercialization with exclusive worldwide license; initial human clinical trials results expected in 2023;
- In-vitro data shows neutralization of all COVID-19 variants of concern including Delta, and based on in-silico studies, Omicron, at significantly lower dose levels than existing antibody treatments; These NanoAbs are also stable at very high temperatures and will be developed for convenient inhalation administration targeting the virus directly in the lungs and airways;
- Broader collaboration for the development of additional NanoAbs addressing diseases with large unmet medical needs and attractive commercial opportunities expected to be signed in January 2022.
JERUSALEM, Dec. 22, 2021 /PRNewswire/ -- BiondVax Pharmaceuticals Ltd. (Nasdaq: BVXV), a biopharmaceutical company focused on developing, manufacturing and commercializing innovative products for the prevention and treatment of infectious diseases and other illnesses, today announced the signing of definitive agreements with the Max Planck Society (MPG), the parent organization of the Max Planck Institute of Biophysical Chemistry, and the University Medical Center Göttingen (UMG), both in Germany, to enter into a strategic collaboration for the development and commercialization of innovative COVID-19 nanosized antibodies (NanoAbs*). These agreements are part of a broader collaboration, expected to be signed in January 2022, covering development and commercialization of NanoAbs for several other disease indications with large market sizes that leverage their unique binding affinity, stability at high temperatures, and potential for more effective and convenient routes of administration.
The lead NanoAb candidates for the COVID-19 program, developed by Professor Dirk Görlich, Director at the Max Planck Institute of Biophysical Chemistry, and Professor Matthias Dobbelstein, Professor of Molecular Oncology at the UMG, exhibit a strong competitive edge over existing COVID-19 therapies. For example, the NanoAbs exhibit superior affinity to the target, neutralizing the SARS-COV-2 virus, in-vitro, at drug concentrations about 100-times lower than those required by currently commercially available monoclonal antibodies. This may lead to increased efficacy, reduced side effects, and allow production at a lower cost. In addition, the NanoAbs' small size and hyper-thermostability at 95°C (200°F) potentially enables administration through inhalation and longer shelf life, and potentially avoids the need for enhanced cold chain storage and transport. Together, these attributes are designed to create significant competitive advantages.
The NanoAbs have demonstrated strong neutralization at very low concentrations of major Variants of Concern (VoCs) including Alpha, Beta, Gamma and Delta in in-vitro studies in Görlich's and Dobbelstein's laboratories and based on in-silico studies, are expected to neutralize Omicron. Given the potential of additional emerging VoCs, the BiondVax-MPG-UMG collaboration covers development of new NanoAbs as needed during the evolution of COVID-19. The methodologies employed by the Görlich and Dobbelstein labs enable generation of new NanoAb candidates within weeks of acquiring the sequence for any emerging variant, and thereby positions BiondVax to stay at the forefront of COVID-19 preparedness.
In addition to the COVID-19 NanoAb collaboration and as previously announced, BiondVax, MPG and UMG have signed a term-sheet for development of NanoAbs towards other indications with major opportunities in key therapeutic areas of psoriasis, asthma, macular degeneration, and psoriatic arthritis. These are all diseases with known and validated antibody drug targets (thereby shortening development timelines and lowering risk), and where the NanoAb advantages have the potential to capture significant market share. BiondVax anticipates finalizing the collaboration agreements for these additional NanoAbs in January 2022.
BiondVax is planning a rapid development path that leverages the company's expertise and capabilities in biological drug development and manufacturing. In particular, NanoAbs can be mass-produced through recombinant protein manufacturing in sites such as BiondVax's GMP biologics manufacturing facility in Jerusalem. BiondVax anticipates conducting COVID-19 NanoAb preclinical studies in 2022 with initial human clinical trials results in 2023.
Mr. Amir Reichman, BiondVax's CEO, commented, "As Omicron's recent and rapid emergence makes clear, improved therapeutics are needed to bring COVID-19 under control. The innovative COVID-19 NanoAb candidates offer the potential for a superior therapeutic platform. Overall, the NanoAb platform is an incredible opportunity for BiondVax. It will serve as a basis for an exciting new pipeline of commercially attractive products to address unmet therapeutic needs of diseases such as psoriasis, psoriatic arthritis, asthma, and macular degeneration. The technology is a great fit to our manufacturing site in Jerusalem and our experience and expertise in biological drug development. I wish to thank Professors Görlich and Dobbelstein for their enthusiasm for this project, and Max Planck Innovation's technology transfer team including Dr. Dieter Link for their assistance in developing this innovative collaboration. BiondVax is in the process of a transformative turnaround, and I am highly confident that these NanoAb therapies present compelling commercial potential and will address important healthcare needs."
Professor Dirk Görlich, Director at the Max Planck Institute for Biophysical Chemistry, commented, "We believe that the innovative platform developed at the Max Planck Institute for Biophysical Chemistry and the University Medical Center of Göttingen has great potential to provide patients with superior therapies. We are particularly pleased to team with BiondVax to use their drug development and manufacturing capabilities and expertise to advance the COVID-19 nanosized antibodies through additional development towards marketing approvals."
The collaboration has been facilitated by kENUP, a civic society organization promoting innovative industries in Europe.
FDA clears Insightec's MRI-guided sound wave technology to treat prostate cancer
by Andrea Park | Dec 3, 2021 11:20am
https://www.fiercebiotech.com/medtech/fda-clears-insightec-s-mri-guided-sound-wave-technology-to-treat-prostate-cancer
Quote:
The prostate cancer-focused technology’s FDA clearance comes just a month after Insightec racked up yet another regulatory approval for its Exablate Neuro system. The new indication allows the brain-centric version of the focused ultrasound tech to be used to treat patients with advanced Parkinson’s disease who are experiencing issues with mobility, rigidity and dyskinesia, a complication that causes involuntary muscle movements.
That FDA approval adds to Exablate Neuro’s other indications, which OK the system for use in treating tremors in patients with tremor-dominant Parkinson’s or medication-resistant essential tremor.
Though Exablate Neuro has become Insightec’s flagship product since its first FDA nod in 2016, Exablate Prostate marks a return to the Israeli company’s original focus outside the brain. Dating back to 2004, Insightec initially locked down the agency’s approval to apply its Exablate tech toward the treatment of uterine fibroids and metastatic bone cancer pains.
FDA clears Insightec Exablate Prostate device
https://www.massdevice.com/fda-clears-insightec-exablate-prostate-device/
DECEMBER 1, 2021 BY SEAN WHOOLEY
Insightec announced today that it received FDA 510(k) clearance for its Exablate Prostate system for treating prostate tissue.
Haifa, Israel-based Insightec designed its Exablate Prostate system to use high-intensity focused ultrasound to ablate targeted tissue in the prostate. Health providers perform the procedure under magnetic resonance imaging (MRI) guidance for high-resolution visualization of the patient’s anatomy for precise targeting and real-time temperature monitoring.
According to a news release, the treatment does not require incisions and is performed in a single session, allowing patients to quickly return to normal activity with minimal complications.
Clinical trial results demonstrated that the Exablate Prostate system resulted in minimal damage to adjacent structures with low rates of impact on potency and continence, Insightec said.
“Insightec remains focused on achieving strategic milestones and expanding into indications beyond neurosurgery such as approval for prostate tissue ablation,” Insightec CEO & Chairman Dr. Maurice R. Ferré said in the release. “Incisionless surgery is transforming patient care with a surgical option that is considerably less invasive.”
Insightec Announces FDA Approval of Exablate Neuro for the Treatment of Parkinson's Disease
The device is indicated for unilateral pallidotomy for medication-refractory patients with moderate to severe motor complications
https://www.prnewswire.com/news-releases/insightec-announces-fda-approval-of-exablate-neuro-for-the-treatment-of-parkinsons-disease-301414996.html
MIAMI, Nov. 3, 2021 /PRNewswire/ -- Insightec®, a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy, announced today that the U.S. Food and Drug Administration (FDA) has approved the Exablate® Neuro for treating advanced Parkinson's Disease patients suffering from mobility, rigidity, or dyskinesia symptoms.
The Exablate Neuro uses focused ultrasound waves to precisely target and ablate the globus pallidus (GPi) during a pallidotomy. The treatment is incisionless, does not require brain implants, and has less risk of infection than invasive surgery.
"Movement disorder neurologists now can offer their Parkinson's patients a less invasive surgical option as part of their treatment plan," Paul S. Fishman, MD, PhD, professor of neurology, pharmacology and neurobiology at the University of Maryland School of Medicine.
"This approval is significant in that it adds Focused Ultrasound as an incisionless surgical option to treat motor symptoms of Parkinson's disease," added Howard M. Eisenberg, MD, R.K. Thompson Professor, Department of Neurosurgery at the University of Maryland and Principal Investigator.
Parkinson's disease (PD), a neurodegenerative disorder, affects an estimated one million patients in the United States. Tremor, rigidity, slow movement (bradykinesia), and postural instability are the cardinal features of PD. When medication induces side effects or is not well tolerated, surgical treatments, such as Focused Ultrasound, may be considered.
"This expanded approval of clinical indications to treat Parkinson's Disease signifies the growing understanding and acceptance of Focused Ultrasound as an effective treatment modality," commented Maurice R. Ferré MD, Insightec CEO and Chairman of the Board of Directors. "More importantly, it drives our continued efforts to help transform the lives of people living with debilitating neurological and other conditions."
The Exablate Neuro device received approval by the FDA for the treatment of medication-refractory Essential Tremor in 2016 and for Tremor-dominant Parkinson's Disease in 2018. There are currently 37 medical centers in the United States using the Insightec Exablate Neuro device to treat patients with medication-refractory Essential Tremor and Tremor-dominant Parkinson's Disease.
Ivy Brain Tumor Center and SonALAsense Announce Positive Initial Results of the First-in-Human Phase 0/1 Clinical Trial of Non-Invasive Sonodynamic Therapy for Recurrent Glioblastoma
Ivy Brain Tumor Center
Mon, September 20, 2021, 7:00 PM GMT+3·3 min read
Trial provides proof of concept for this new metabolically targeted, rapidly acting technology platform for brain tumors and other lethal cancers.
https://finance.yahoo.com/news/ivy-brain-tumor-center-sonalasense-160000312.html
Ivy Brain Tumor Center
Mon, September 20, 2021, 7:00 PM GMT+3·3 min read
Trial provides proof of concept for this new metabolically targeted, rapidly acting technology platform for brain tumors and other lethal cancers
Nader Sanai, MD, director of the Ivy Brain Tumor Center and director of neurosurgical oncology at Barrow Neurological Institute
Ivy Center's clinical trial of sonodynamic therapy uses Insightec’s magnetic resonance-guided focused ultrasound (MRgFUS) technology in combination with SonALAsense’s proprietary intravenous formulation of 5-aminolevulinic acid (SONALA-001) to investigate the feasibility, safety, and biological effects of the treatment
Ivy Center's clinical trial of sonodynamic therapy uses Insightec’s magnetic resonance-guided focused ultrasound (MRgFUS) technology in combination with SonALAsense’s proprietary intravenous formulation of 5-aminolevulinic acid (SONALA-001) to investigate the feasibility, safety, and biological effects of the treatment
More
Ivy Center's clinical trial of sonodynamic therapy uses Insightec’s magnetic resonance-guided focused ultrasound (MRgFUS) technology in combination with SonALAsense’s proprietary intravenous formulation of 5-aminolevulinic acid (SONALA-001) to investigate the feasibility, safety, and biological effects of the treatment
PHOENIX, Ariz., Sept. 20, 2021 (GLOBE NEWSWIRE) -- The Ivy Brain Tumor Center at Barrow Neurological Institute, the largest early-phase drug development program for brain cancer in the world, and SonALAsense, a pioneer in developing non-invasive sonodynamic therapy (SDT) for managing fatal cancers so patients can become survivors, today announced positive initial results of the first-in-human Phase 0/1 clinical trial of SDT in recurrent glioblastoma patients. The initial results indicate SDT rapidly leads to targeted oxidative stress and cell death in human glioblastoma tissue. SDT was well-tolerated in all patients.
Video: How Sonodynamic Therapy Works
The Ivy Brain Tumor Center dosed its first patient in March 2021 and is currently evaluating SDT using Insightec’s magnetic resonance-guided focused ultrasound (MRgFUS) technology in combination with SonALAsense’s proprietary intravenous formulation of 5-aminolevulinic acid (SONALA-001) to investigate the feasibility, safety, and biological effects of the treatment.
Initial results of this study were presented today at the European Society for Medical Oncology Congress during a mini oral session on CNS tumors by Nader Sanai, MD, director of the Ivy Brain Tumor Center and director of neurosurgical oncology at Barrow Neurological Institute.
“Sonodynamic therapy has tremendous potential to become a new nonsurgical therapeutic modality to treat brain tumors,” said Nader Sanai, MD. “We found that MR-guided focused ultrasound combined with SONALA-001 demonstrated rapid and selective cell death in the patients’ tumors, an encouraging discovery of this new noninvasive treatment option.”
“The Ivy Center’s Phase 0/1 study has shown a promising translation of the preclinical results of SDT in patients with glioblastoma. The same biomarkers that were positive in preclinical studies, and contributed to a significant increase in survival in two animal glioma models, are being seen in the clinical trial patient participants,” said Stuart Marcus, MD, PhD, Founder, CEO and CMO of SonALAsense. “SONALA-001 targets only the glioblastoma tumor cells through selective uptake and conversion to a metabolite which is then activated by focused ultrasound, the SDT process, to kill just the tumor itself, not the surrounding tissue – a truly remarkable finding.”
This study is funded by the Ben and Catherine Ivy Foundation and the individual donors of the Barrow Neurological Foundation. Additional clinical trial information can be found at NCT04559685 or the Ivy Brain Tumor Center’s website.
###
About Ivy Brain Tumor Center
Ivy Brain Tumor Center at the Barrow Neurological Institute in Phoenix, AZ is a non-profit translational research program that employs a bold, early-phase clinical trials strategy to identify new treatments for aggressive brain tumors, including glioblastoma. The Ivy Center’s Phase 0 clinical trials program is the largest of its kind in the world and enables personalized care in a fraction of the time and cost associated with traditional drug development. Unlike conventional clinical trials focusing on single drugs, its accelerated trials program tests therapeutic combinations matched to individual patients. Learn more at IvyBrainTumorCenter.org. Follow the Ivy Brain Tumor Center on Facebook, Instagram, Twitter and LinkedIn.
About SonALAsense
SonALAsense is a clinical-stage company developing sonodynamic therapy (SDT) as a first-in-class, noninvasive drug-device combination for the treatment of recurrent glioblastoma multiforme, diffuse intrinsic pontine gliomas, and other deadly cancers. SDT utilizes MR-guided focused ultrasound (the device) in combination with aminolevulinic acid (a drug) to selectively target and kill tumor cells. For more information about SonALAsense, visit www.sonalasense.com. Follow SonALAsense on Twitter and LinkedIn.
India Ministry of Health and Family Welfare Approves Exablate Neuro Focused Ultrasound Platform
https://www.bakersfield.com/ap/news/india-ministry-of-health-and-family-welfare-approves-exablate-neuro-focused-ultrasound-platform/article_bdf9ad7d-8ba8-5514-9b11-891c555d6ae3.html
HAIFA, Israel and MIAMI, Fla., Sept. 20, 2021 /PRNewswire/ -- Insightec ®, a global healthcare company focused on transforming patient care, announced its incisionless neurosurgery platform, the Exablate® 4000, has received market approval by the Central Drugs Standard Control Organization (CDSCO), part of the Ministry of Health and Family Welfare of India.
The Exablate 4000 (Exablate Neuro) platform uses MR-guided focused ultrasound to precisely ablate a small target deep within the brain without incisions. The approval will enable treatment of patients in India living with Essential Tremor, Tremor-dominant Parkinson's Disease and Neuropathic Pain.
"This approval adds to the growing recognition of the value of Focused Ultrasound for global healthcare systems," commented Maurice R. Ferré MD, Insightec CEO and Chairman of the Board of Directors. "More importantly, expanded access to our technology is helping to transform the lives of people living with debilitating medical conditions."
Essential Tremor is a neurological condition that causes uncontrollable shaking in an estimated 41 million people worldwide. More than 80 medical centers around the globe are treating Essential Tremor patients with the Exablate Neuro on a regular basis. Performed in a single session in an MRI suite, many patients experience immediate tremor relief in the treated hand with minimal side effects.
"This technology offers appropriate patients immediate tremor control with a less invasive procedure that requires no incisions or anesthesia with minimal complications," said Prof. Paresh Doshi, Director of Neurosurgery at Jaslok Hospital and Research Centre and Group Advisor Functional Neurosurgery, Apollo Group of Hospitals, President of The Neuromodulation Society of India and Past President of the Indian Society for Stereotactic and Functional Neurosurgery. "Global clinical studies have demonstrated the procedure as approved is safe and helps patients to regain daily function."
About Insightec
Insightec is a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy. The company's Exablate Neuro platform focuses sound waves, safely guided by MRI, to provide tremor treatment to patients with medication-refractory Essential Tremor and Tremor-dominant Parkinson's Disease. Research for future applications in the neuroscience space is underway in partnership with leading academic and medical institutions. Insightec is headquartered in Haifa, Israel, and Miami, with offices in Dallas, Shanghai and Tokyo.
Jewish New Year fruit may hold seeds of hope for brain disease sufferers
Israeli neurologist, nanotech expert make pomegranate oil capsules that send antioxidants where they can have the most effect
https://www.timesofisrael.com/jewish-new-year-fruit-may-hold-key-to-graceful-ageing/
By SHOSHANNA SOLOMON
18 September 2017, 4:36 pm
As Jewish families across the world reach for the pomegranate that they customarily eat on Rosh Hashanah, they may not realize that the fruit, with its juicy red seeds and crown-like crest, could hold a key to graceful aging.
King Solomon is said to have designed his crown based on that of the pomegranate, and the image of the fruit often appears on ancient coins of Judea. The pomegranate is said to have 613 seeds, which correspond with the 613 Jewish precepts or commandments set out by the Torah regulating the Jewish way of life. For this reason, and because it represents fruitfulness, knowledge, learning and wisdom, and is seasonal in Israel, it customarily appears on Jewish New Year dinner tables.
The association of the pomegranate with knowledge, learning and wisdom may not be far from the truth. Pomegranate seed oil (PSO) contains high concentrations of punicic acid, or omega 5 as it is also called, which is believed to be one of the most powerful antioxidants in nature.
“Oxidation of proteins and lipids play an important role in aging and neuro degeneration in the brain in general,” said Prof. Ruth Gabizon, a researcher of degenerative brain diseases at the Neurology Department of Hadassah University Hospital in Jerusalem. “Brain cells die over time, from when we are teenagers, and they are not replaced.”
Common daily activities, such as digesting food and breathing, create free radicals that result in oxidation and damage to human cells, in particular to brain cells. Unlike blood or skin cells, brain cells do not get replaced by new ones. So free radicals are harmful to our health and end up impairing our thinking, memory, orientation and alertness, among others.
Degenerative brain disease and brain atrophy are typical of debilitating illnesses such as Alzheimer’s, Parkinson’s, and Creutzfeldt-Jakob disease in which brain cells are destroyed, followed by rapid functional and behavioral deterioration and eventual death.
The number of people worldwide living with dementia, for example, is expected to almost double every 20 years, reaching 75 million in 2010 and 131.5 million in 2050, according to Alzheimers’ Disease International.
Fighting fire with food
Aging and brain degeneration are a natural and unavoidable process, explained Gabizon, but they can be accelerated or slowed down depending on our lifestyles.
Antioxidants are known for their ability to protect against the destruction of brain and body cells. They can be found in foods such as cranberries, blueberries, beans, artichokes, pecans and foods containing Vitamin E.
GranaGard is a food supplement that contains pomegranate seed oil
“If we are able to control the levels of free radicals, maybe our cells will work better and live longer,” Gabizon said. “Our approach is that even if we cannot cure the severely affected patients with diseases such as Alzheimer’s, since they are diagnosed at a stage when large numbers of brain cells are already dead, perhaps we can delay the disease’s advance at early stages or even prevent disease outbreak in healthy people at risk of developing neurodegeneration, which is actually most of us.
“This, by extending the life span of brain cells, and improving their functioning even under dire conditions in which the body is filled with ‘biological garbage’ like the destructive oxidizing free radicals.”
Antioxidants, as they are present in many vegetables and fruits, may in principle protect against the destruction of brain and body cells. And this is the case for pomegranate seed oil.
Unfortunately, antioxidants that we consume through food and supplements do not always have the desired impact because they are consumed in too low of a concentration or broken down in the digestive system, and thus never make it to the brain or other cells.
The challenge, said Gabizon, is to make sure the pomegranate oil that we eat, which generally is filtered out by the liver, gets to the parts of our body which can benefit from it.
So Gabizon teamed up with Prof. Shlomo Magdassi — an expert in the field of nanotechnology from the Casali Institute for Applied Chemistry at the Hebrew University of Jerusalem — and together devised a way to break down the oil into tiny particles that can slip through the liver undetected and make their way to the brain.
The product they have developed, called GranaGard, has a high concentration of antioxidants that have a good chance of reaching the brain.
Prof. Shlomo Magdassi of the Institute of Chemistry at the Faculty of Science, February 02, 2012. (Nati Shohat/Flash90)
A study of GranaGard performed by Gabizon and Magdassi found that consumption by lab mice with multiple sclerosis delayed the spread of the disease and considerably reduced its intensity. An additional experiment with lab mice who suffered from Creutzfeldt-Jakob showed that the use of GranaGard “considerably delayed the spread of the disease and lowered the intensity of the accompanying degenerative-dementing processes,” Gabizon said.
The two studies were published in the international Journal of Nanomedicine in November 2015 and in the International Journal of Nanomedicine in 2014.
Gabizon and Magdassi patented the product and formed the firm Granalix Biotechnologies Ltd. They are now hoping to undertake clinical trials to test the effect of their formulation in humans with Creutzfeldt-Jakob, Alzheimer’s and multiple sclerosis, Gabizon said.
GranaGard is already on sale as a dietary supplement at www.granalix.com and is being used by patients with degenerative brain diseases, their family members and others, Gabizon said. The recommended dose is two capsules with breakfast.
Seeds of hope
M is a Jerusalem-based 50-year old medical professional who was diagnosed a year and a half ago with brain atrophy. He has been using GranaGard for a year and three months, after a friend recommended the supplement and after he met with Gabizon to find out more about it.
“One of the ways to deal with atrophy is via fighting free radicals,” M said, preferring not to make his identity known. Since his diagnosis, he has started an antioxidant diet, which includes taking GranaGard and his regular medications along with exercise. Since his diagnosis, he said, there has been no deterioration in his condition.
He would “absolutely” recommend taking the supplement, M said. “I cannot substantially say this (GranaGard) absolutely benefited me, but I have incorporated it as an important piece of my regime. The end result is that I am still functioning.”
Prof. Tamir Ben-Hur, the chairman of the Department of Neurology at the Hadassah Medical Center, knows the supplement and also knows Gabizon, but has no stake in the firm or the technology, he said.
“Oxidation is one of the important mechanisms of tissue injury in many diseases, including neurodegenerative diseases like Alzheimer’s,” he said in a phone interview. “To use antioxidants to stall the process has been around for a while but efforts have generally failed, because either the anti-oxidants were too weak or the active ingredient did not reach the brain.”
The concept behind GranaGard is not new, he said, but the way it is used makes it more powerful than other antioxidants available, such as Vitamin E.
Products that consumers find in health stores – either creams or products to be taken orally — are sometimes inconsistent in their dosages and don’t get effectively absorbed. “These capsules help the formulation surpass the liver, and this is the true advantage of this product versus other products in the stores.”
“There is no clinical proof that GranaGard can slow down Alzheimer’s, and I hope a clinical trial will prove that. But there is good biological research data that it is well absorbed by the gut, reaches the brain and has an antioxidant effect on the brain. We believe it may work with humans but we need to prove it,” he said.
Elderly men play backgammon at a country club in northern Tel Aviv, illustrative (Tomer Neuberg/Flash90)
“I would certainly recommend it to people who are aging and prone to degenerative diseases,” he added. “When you have a pre-Alzheimer’s condition there is nothing to prevent the disease from developing and nothing to stop it. There is no clinical proof of efficacy, but I suggest people take it, because there is good biological rationale, and good data in animals, so why not?”
Gabizon is cautious about raising hopes of patients who are suffering from these debilitating afflictions. At the moment, the only product her company has on the market is an over-the-counter food supplement. Most likely, to actually defeat these illnesses, a cocktail of various compounds will have to be developed, she said.
But each step forward is a victory, she added. Their component has shown a way antioxidants can be broken down to go undetected by the liver and has shown a “definite effect” on the pathology of mice.
“We have not yet proven the formulation’s effect on the human brain,” she said, “But the effect on mice has been proven and we hope to show the same effect with humans during the clinical trials. Even if we don’t find a cure for these diseases, if we are able to delay the degeneration process by a few years, we have done our job.”
$CEMI covid19 testing
Bioventus to exercise option to buy CartiHeal for $500m
Nir Altschuler
https://en.globes.co.il/en/article-bioventus-to-exercise-option-to-buy-cartiheal-for-500m-1001383334
30 Aug, 2021 15:49
Gali Weinreb
The Israeli company has developed cartilage implants made from coral.
US orthologics company Bioventus (Nasdaq: BVS) has announced today that it is exercising its option to acquire Israeli medical device company CartiHeal, which has developed cartilage implants made from coral. Bioventus will pay $360 million up front and milestone payments that could bring the deal up to $500 million.
Bioventus has elected to make a $50 million escrow payment pursuant to its Option and Equity Purchase Agreement with CartiHeal Ltd., signaling its intent to move forward with an acquisition of CartiHeal, as soon as the Israeli company's cartilage treatment receives FDA approval. CartiHeal recently successfully completed a major clinical trial and if there are no major surprises the company will receive FDA clearance in the coming year.
Bioventus SVP and CSO Alessandra Pavesion said, "The robust data generated from the pivotal clinical trial, a randomized controlled trial with Agili-C, demonstrated superiority over surgical standard of care, microfracture and debridement, in KOOS overall compared to baseline. We believe this product could be a strong alternative for the approximately 650,000 US patients annually receiving microfracture or debridement along with other cartilage treatment options. In combination with our HA products, Agili-C represents an exciting potential new offering for our portfolio designed to address the spectrum of osteoarthritis disease."
CartiHeal's technology was developed by founder and CEO Nir Altschuler. He said, "We found that the implant draws stem cells from the bone marrow. These cells gradually break down in the implant while rebuilding the damaged cartilage and bone."
Elron Electronic Industries Ltd. (OTC: ELRN; TASE:ELRN) said after the agreement was signed that it expects to receive $126-129 million if the option is exercised including $90-92 million at purchase and $36-37 million in milestone payments - if CartiHeal generates revenue of $100 million during a 12 month period.
Accelmed Fund, controlled by Uri Geiger, has a 15% stake in CartiHeal, and expects to receive $75 million, a return of seven times its investment. Peregrine Fund has a 5% stake in CartiHeal and expects to see returns of 50 times its $200,000 investment. Other shareholders include aMoon, Access Medical Fund, Partech, and Johnson & Johnson Innovations.
Israel becomes first country in world to ok third Covid shot
Israel’s health providers begin administering third dose of vaccine for adults with immune insufficiency.
https://www.israel21c.org/israel-becomes-first-country-in-world-to-ok-covid-booster-for-immunocompromised/?utm_source=The+Weekly&utm_campaign=weekly-2021-07-14&utm_medium=email
Huons to supply focused ultrasound MRI to SMC
https://www.koreabiomed.com/news/articleView.html?idxno=11548
Huons said Monday that it is speeding up installing ExAblate Neuro to Samsung Medical Center (SMC) in September to provide magnetic resonance-guided focused ultrasound surgery (MRgFUS) for those with brain diseases.
Huons said Monday that it would install a magnetic resonance-guided focused ultrasound device, ExAblate Neuro, at Samsung Medical Center to provide safer surgery for people with brain diseases.
Huons said Monday that it would install a magnetic resonance-guided focused ultrasound device, ExAblate Neuro, at Samsung Medical Center to provide safer surgery for people with brain diseases.
Huons acquired exclusive rights to supply ExAblate Neuro developed by the Israeli biotechnology company Insightec in 2019. The device uses a non-invasive surgical method to remove the specific tissue that causes abnormal brain function by using MRgFUS, unlike the existing therapy that requires cutting a bony opening in the skull.
The device has no risk of bleeding or infection, and the surgeons can monitor the surgical site in real-time through MRI images while performing an operation. Patients who receive surgery with ExAblate Neuro recover rapidly and return to their usual daily activities.
“If Huons installs the device at SMC, people with brain disorders will receive safer operations,” Huons said.
Regulatory agencies in 23 countries, including the U.S. Food and Drug Administration, have authorized ExAblate Neuro, and 82 hospitals and other medical institutions are using the device, it added.
“ExAblate Neuro is a new device that has shown excellent therapeutic effect for brain diseases, such as tremor,” Samsung Medical Center Neurosurgery Department Professor Lee Jung-il said. “We are glad to introduce a novel, cutting-edge treatment system that has drawn the attention of the neurosurgery communities worldwide.”
Ori Atar, the director of Insightec’s Asian Growth Markets, said, “We are pleased to supply ExAblate Neuro to Samsung Medical Center that provides excellent patient-centered medical services in Korea.”
Ori Atar added that the company expects ExAblate Neuro to become a new treatment option in Korea, stressing the effectiveness of treatment for brain diseases has been proven globally with studies of various illnesses.
Huons CEO Eom Gi-an also said. “Korean patients can receive therapies for brain disease using ExAbalte Neuro, as Samsung Medical Center decided to install the device. We will provide more treatment options for patients.”
Insightec Announces Registration Approval Of Exablate Neuro By Singapore Health Sciences Authority
Positive momentum continues to bring Focused Ultrasound to global patients living with Essential Tremor and Tremor-dominant Parkinson's Disease
http://m.koreaherald.com/amp/view.php?ud=20210705000139
HAIFA, Israel and MIAMI, July 5, 2021 /PRNewswire/ -- Insightec®, a global healthcare company focused on creating the next generation of patient care, announced approval of the Exablate® Neuro platform by the Health Sciences Authority (HSA) in Singapore.
The Exablate Neuro (Exablate 4000) platform uses Focused Ultrasound to ablate a target deep in the brain. The unilateral treatment is suitable for patients with Essential Tremor (age 22 or older) or Tremor-dominant Parkinson's Disease (age 30 or older) who have not responded to medications.
"The value of Focused Ultrasound for medical centers treating movement disorders is greatly improved patient care," said Maurice R. Ferré MD, Insightec CEO and Chairman of the Board of Directors. "Our technology enables patients to go from a life dominated by tremor to the potential for a calm, steady hand."
There are 81 leading medical centers globally that have established Focused Ultrasound programs with Exablate Neuro. Insightec is working with Transmedic Group, a leading distributor of advanced medical technology in Southeast Asia, to advance adoption of the Exablate Neuro in the Singapore market.
"We are excited to bring this transformational technology to medical centers in Singapore," commented Teo Kee Meng, Transmedic Managing Director. "More importantly, patients will now have the option to choose a treatment that is outpatient and incisionless."
Insightec Announces Samsung Medical Center Is Joining Focused Ultrasound Community
https://www.kpvi.com/news/national_news/insightec-announces-samsung-medical-center-is-joining-focused-ultrasound-community/article_3c35046e-deea-54c5-852a-471a2dfb7bc5.html
The program offers treatment for patients with Movement Disorders, pain, and behavioral disorders in South Korea
HAIFA, Israel and MIAMI, July 5, 2021 /PRNewswire/ -- Insightec®, a global healthcare company focused on creating the next generation of patient care, announced Samsung Medical Center is establishing a Focused Ultrasound program with the adoption of Exablate® Neuro. The Exablate Neuro (Exablate 4000) platform uses Focused Ultrasound to ablate targets deep in the brain for a therapeutic effect.
"Leading neuroscience departments at medical centers around the globe recognize the value of adopting Focused Ultrasound as a treatment option for patient care," commented Ori Atar, Director of Asia Growth Markets at Insightec. "We are excited by the growth of adoption in the Korean market."
"Focused Ultrasound enables performing precise, minimally invasive treatments," Prof. Jung Il Lee, Professor, Department of Neurosurgery, Samsung Medical Center explains. "The clinical evidence of the treatment demonstrates good tremor control and a favorable safety profile for patients whose hand tremor affects their daily functioning."
There are 81 leading medical centers globally that have established Focused Ultrasound programs with Exablate Neuro. Insightec is partnering with Huons, a manufacturer of pharmaceutical products and distributor of medical solutions, to expand Focused Ultrasound programs in the Korean market.
"Our mission is to deliver solutions that improve people's health," says Key-An Um, CEO of Huons. "We are eager to bring Focused Ultrasound to medical centers and help transform the lives of people living with movement disorders."
Israeli oral COVID-19 vaccine en route to clinical trials
Unlike the Pfizer or Moderna vaccines that use messenger RNA, this is an oral subunit vaccine, meaning the vaccine presents the antigen to the immune system without introducing pathogen particles.
By MAAYAN JAFFE-HOFFMAN JUNE 11, 2021 06:26
https://www.jpost.com/health-science/israeli-researchers-say-oral-covid-19-vaccine-works-as-booster-study-670679
More than one year after the MIGAL Galilee Research Institute committed to delivering the world’s first oral COVID-19 vaccine, its affiliate company MigVax has reported that its vaccine performed well on rats and it hopes to have the vaccine ready as a booster dose for people within the next year.
“Fifteen months into the pandemic, we now see that the struggle to keep COVID under control is nearly as challenging as getting it under control to begin with,” said Prof. Itamar Shalit, MigVax’s infectious disease expert. “Oral boosters such as our MigVax-101 will be key enablers that will help health organizations and the world transition from ‘panic mode’ to routine, due to their ability to reduce the cost and expand the reach of ongoing vaccination programs.”
The company is calling their vaccine MigVax-101. Unlike the Pfizer or Moderna vaccines that use messenger RNA, this is an oral subunit vaccine, meaning the vaccine presents the coronavirus antigen to the immune system without introducing pathogen particles, whole or otherwise.
In the preclinical trial, the company administered the vaccine in a boost format to the rats following a double injection of the coronavirus spike (S1) protein.
The results showed that using MigVax as a booster elicited markedly higher neutralizing antibody titers than rats receiving an oral placebo booster, a release explained. In addition, it elicited more effective neutralizing antibodies than a third S1 booster injection.
“They are looking to use this as a booster for those who took the Chinese vaccine, AstraZeneca or Moderna or Pfizer,” Dr. Morris Laster, who is responsible for medical technology investments for OurCrowd told The Jerusalem Post. OurCrowd led MigVax’s first investment round.
According to Laster, the vaccine has several advantages, including that because it is an oral vaccine it would not require administration by a medical professional. Also, it utilizes more than one kind of protein, which could make it more effective against variants. The vaccine does not require freezing conditions like the mRNA vaccines so it could be easier to store and distribute.
Finally, because it does not inject people with genetic or viral material, Laster believes that more people will be prone to taking it.
“People afraid to get injected should have an easier time swallowing protein,” he said. “It’s kind of like drinking yogurt.”
To launch Phase I and II clinical trials, the company is seeking $6 million to $7m., the company’s CEO, David Zigdon, told the Post. He said if the money comes through quickly, the vaccine could be on the market in as quickly as nine months.
He said the company has a road map and has already spoken with regulatory bodies to achieve clearance for its trials. However, he told the Post that the company does not yet have final Health Ministry approval to start the trial.
MIGAL was the first company worldwide to announce that it would put a vaccine on the market.
In February 2020, an announcement by MIGAL with a quote by then science, technology and space minister Ofir Akunis promised that “the vaccine could be ready within a few weeks and available in 90 days.” However, shortly thereafter the company stopped communicating and refused to comment despite multiple requests by the Post.
MIGAL had previously developed a vaccine against infectious bronchitis virus (IBV), which causes a bronchial disease affecting poultry. The effectiveness of the vaccine has been proven in preclinical trials carried out at the Veterinary Institute and researchers believed they could adapt the vaccine for human use.
Zigdon and Laster said MigVax ran into problems adapting the protein and producing it in efficient amounts to keep the price of the vaccine reasonable. Zigdon confirmed that the challenges have been overcome.
MigVax is not the only oral vaccine based on Israeli technology being evaluated for use against COVID-19.
In March 2021, Oramed Pharmaceuticals Inc., a clinical-stage pharmaceutical company based on technology developed by Hadassah-University Medical Center, announced a joint venture with Premas Biotech to develop a novel oral vaccine. Together they formed the company Oravax Medical Inc. The vaccine is based on Oramed’s “POD” oral delivery technology and Premas’s vaccine technology.
The company is preparing to launch its own clinical trial next quarter, Oramed CEO Nadav Kidron told the Post on Thursday.
He evaluated the release sent by MigVax and said: “There are 7 billion people who need and are waiting for vaccines, so I would welcome more initiatives for oral vaccines.”
Israel may have achieved herd immunity against Covid-19
With 56% of citizens vaccinated and another 15% recovered, Israel may be the first country where people who are immune protect those who aren’t.
https://www.israel21c.org/israel-may-have-achieved-herd-immunity-against-covid-19/?utm_source=The+Weekly&utm_campaign=weekly-2021-04-07&utm_medium=email
West Virginia University Addresses Addiction Crisis with Novel Ultrasound Treatment
https://www.itnonline.com/content/west-virginia-university-addresses-addiction-crisis-novel-ultrasound-treatment
WVU Rockefeller Neuroscience Institute staff prepare a patient for a first-in-the-world clinical trial to investigate the use of focused ultrasound technology to treat opioid use disorder. (WVU Photo/Brian Persinger)
March 17, 2021 — On the heels of the country’s deadliest year for drug overdoses, the West Virginia University Rockefeller Neuroscience Institute launched a first-in-the-world clinical trial to investigate the use of focused ultrasound technology to treat those with opioid use disorder. The procedure marks the potential for a new innovative treatment for addiction.
The goal of the study, conducted in collaboration with Insightec, is to evaluate the safety and potential of focused ultrasound in reducing substance cravings and addictive behaviors.
During the 30 minute procedure, RNI researchers applied focused ultrasound waves to the nucleus accumbens, a key structure in the brain involved in addiction and anxiety. The research team utilized visuals of drug use and drug paraphernalia to induce cravings prior to and during the procedure. While preliminary, RNI investigators were encouraged by the patient’s positive behavioral response to the focused ultrasound, and plan to further investigate the treatment by enrolling additional patients in the study.
The study’s first participant, a 39-year-old business owner from West Virginia, is currently enrolled in WVU’s Comprehensive Opioid Addiction Treatment (COAT) program. He has an over 15-year history of substance use disorder, specifically with prescription opioids, heroin and cannabis, and safely and successfully underwent the focused ultrasound procedure in February 2021.
“COVID-19 has tragically intensified our nation’s addiction crisis,” Ali Rezai, M.D., Rockefeller Neuroscience Institute executive chair and principal investigator of this study, said. “We need to explore technological innovations such as focused ultrasound to help patients and families impacted by addiction. At the RNI, we are routinely using focused ultrasound as an FDA-approved procedure to treat tremors and are conducting clinical trials to explore its potential for treating Alzheimer’s disease and brain tumors.”
Recently released data from the Centers for Disease Control and Prevention show the country’s addiction epidemic worsening during COVID-19. More than 81,000 Americans died from overdose in the 12 months leading up to May 2020, the highest one-year figure in recorded history.
In addition to focused ultrasound, the WVU team is exploring a range of new technologies to treat addiction that includes deploying the use of wearables and artificial intelligence, transcranial magnetic stimulation and deep brain stimulation. In 2019, the RNI launched a first-in-the-U.S. clinical trial exploring the use of deep brain stimulation to treat addiction, which has helped its first patient celebrate more than a year of sobriety after an 18-year battle with substance use.
“We were all impressed with the patient’s bravery to help explore new treatment options for substance use disorder. While the current treatment options for substance use disorder work for many, they don’t work for everyone for a variety of reasons,” James Mahoney, Ph.D., RNI addiction researcher, clinical neuropsychologist and assistant professor in the WVU Department of Behavioral Medicine and Psychiatry and Department of Neuroscience, said. “We need to continue exploring new treatments for those struggling with addiction, and I’m hopeful that focused ultrasound technology will provide another tool to assist those in need moving forward.”
“Clinical research and scientific discovery using the therapeutic power of acoustic energy are helping to transform lives,” Maurice R. Ferré, M.D., Chair and CEO at Insightec commented. “We are excited to continue supporting our partners to pioneer focused ultrasound treatments in the brain for critical diseases like addiction.”
Insightec’s Exablate Neuro uses focused ultrasound guided by magnetic resonance imaging (MRI) technology and is FDA approved to treat tremor from essential tremor and Parkinson’s disease. The company is also working with RNI investigators on first-in-the-U.S. trials of transient opening the blood-brain barrier to treat Alzheimer’s disease, which has shown a reduction in Alzheimer’s plaques in early results.
Aspirin users may have protection against Covid-19
People taking aspirin regularly to prevent cardiovascular disease could have reduced likelihood of Covid-19 infection and shorter duration of disease.
https://www.israel21c.org/aspirin-users-may-have-some-protection-against-covid-19/?utm_source=The+Weekly&utm_campaign=weekly-2021-03-10&utm_medium=email
By Abigail Klein Leichman MARCH 10, 2021, 1:42 PM
Healthy people taking aspirin regularly to prevent cardiovascular disease had a 29 percent lower likelihood of Covid-19 infection compared to aspirin non-users, according to an observational epidemiological study from Israel published in The FEBS Journal.
Researchers from Leumit Health Services, Bar-Ilan University and Barzilai Medical Center analyzed data of 10,477 members of the Leumit HMO who had been tested for Covid-19 from February 1, 2020 to June 30, 2020.
They knew that aspirin was widely used during the 1918 Spanish influenza pandemic several decades before its activity against RNA viruses was understood.
They were also aware of studies showing that aspirin, in addition to reducing inflammation, can help the immune system battle some viral infections.
They hypothesized that pre-infection treatment with low-dose aspirin (75mg) might have a l beneficial effect on Covid-19 susceptibility and disease duration.
“The present study sought to better understand the potential favorable effects of aspirin in aiding the human immune system battle Covid-19,” said Dr. Milana Frenkel-Morgenstern of the Azrieli Faculty of Medicine of Bar-Ilan University.
Aside from the lower likelihood of infection among aspirin users, the data also revealed that the conversion time of SARS-CoV-2 PCR test results from positive to negative among aspirin-using Covid-positive patients was significantly shorter, and the disease duration was two-three days shorter, depending upon the patients’ preexisting conditions.
“This observation of the possible beneficial effect of low doses of aspirin on Covid-19 infection is preliminary but seems very promising,” says Prof. Eli Magen from Barzilai Medical Center, who led the study.
Principal investigator Dr. Eugene Merzon, from Leumit Health Services, emphasizes the importance of repeating the study results using larger samples, and including patients from other hospitals and countries, to verify the results.
Blue-green Spirulina algae may prevent serious Covid-19
Study finds an extract of enhanced Spirulina reduces by 70% the release of an immune-system protein that causes dangerous cytokine storm in the lungs.
https://www.israel21c.org/blue-green-spirulina-algae-may-prevent-serious-covid-19/?utm_source=The+Weekly&utm_campaign=weekly-2021-03-03&utm_medium=email
By Abigail Klein Leichman FEBRUARY 25, 2021, 6:02 PM
Experts have discovered an extract of enhanced Spirulina can reduce the release of an immune-system protein that causes dangerous cytokine storm in the lungs.
An extract of Spirulina blue-green algae may help Covid-19 patients avoid getting seriously ill, according to a study by Israeli and Icelandic scientists published in the journal Marine Biotechnology.
“The potential health benefits of Spirulina are well documented,” the authors noted. “This blue-green algae contains C-phycocyanin (C-PC), a pigment-binding protein, which enhances antioxidation, anti-inflammation, and anti-tumor activities.”
The scientists found that an extract of photosynthetically enhanced Spirulina reduces by 70 percent the release of an immune-system protein that can cause a cytokine storm in the lungs leading to acute respiratory distress and organ damage.
It is believed that cytokine storms are responsible for critical cases of Covid-19.
The research was conducted at MIGAL Galilee Research Institute in northern Israel using algae grown at a lab in Iceland by Israeli company Vaxa, which received European Union funding to explore natural treatments for Covid-19.
“This indicates that the algae extract may be used to prevent cytokine storms if given to patients soon after diagnosis,” said co-lead author Asaf Tzachor, a biotechnology researcher at IDC Herzliya who is currently leading the Food Security and Global Catastrophic Risks Project at the Centre for the Study of Existential Risk at Cambridge University.
The other co-lead author is Or Rozen from MIGAL. Contributing authors include Soliman Khatib and Dorit Avi from MIGAL and Sophie Jensen from MATIS – Food and Biotech Research and Development, Reykjavík.
Clinical trials are planned next, with the goal of formulating oral spirulina drops.
Brazilian Health Regulatory Agency Approves MR-Guided Focused Ultrasound To Treat Essential Tremor And Tremor-Dominant Parkinson's Disease
https://www.prnewswire.com/il/news-releases/brazilian-health-regulatory-agency-approves-mr-guided-focused-ultrasound-to-treat-essential-tremor-and-tremor-dominant-parkinson-s-disease-893967506.html
Insightec Plans to Expand in Latin America through a partnership with Strattner.
HAIFA, Israel and MIAMI, Fl., Feb. 16, 2021 /PRNewswire/ -- Insightec®, a global healthcare company focused on creating the next generation of patient care, announced it received market approval by the Brazilian Health Regulatory Agency, ANVISA (Agência Nacional de Vigilância Sanitária), and is partnering with Strattner to bring Focused Ultrasound to the Brazilian market.
This approval of the Exablate® 4000 (Exablate Neuro) platform, which uses MR-guided focused ultrasound to precisely ablate tissue deep within the brain without incisions, will enable treatment for Essential Tremor and Tremor-dominant Parkinson's Disease that has not responded to medications and Neuropathic Pain.
Insightec is working with Strattner, a leading distributor of equipment for minimally invasive and robotic surgery, to reach leading medical centers and help advance adoption of Focused Ultrasound in the Brazilian market.
"Leading neuroscience departments at medical centers around the globe recognize the value of adopting Focused Ultrasound as a treatment option for patient care. We are excited by the opportunities of expansion into Latin America," commented Maurice R. Ferré MD, Insightec CEO and Chair of the Board of Directors.
Nearly 60 leading medical centers globally treat patients with debilitating hand tremor using Exablate Neuro. Performed in a single session in an MRI suite, many patients experience immediate tremor relief with minimal side effects.
"There are millions of Brazilians suffering from Essential Tremor or Tremor-dominant Parkinson's Disease and Exablate Neuro Focused Ultrasound has the potential to change their lives, through an incisionless procedure," said Felipe Strattner, Head of Strategy at Strattner. "We are eager to bring this transformational technology to medical centers across Brazil and help drive medicine forward in our country."
Exablate Neuro is the first and only focused ultrasound device approved by the U.S. Food and Drug Administration to treat medication-refractory Essential Tremor and Tremor-dominant Parkinson's Disease.
"Brazil represents a key strategic market in Latin America and this approval strongly positions Focused Ultrasound for future growth in the region," said J. Michael Quijano, Head of Strategic Partnerships & Business Development, Americas at Insightec.
Has Israel just found the cure for Covid?
Experimental Covid drug cures 30 out of 30 moderate to severe cases in Phase I clinical trial at Israeli hospital. Second new drug also shows promise.
https://www.israel21c.org/has-israel-just-found-the-cure-for-covid/?utm_source=The+Weekly&utm_campaign=weekly-2021-02-10&utm_medium=email
By Abigail Klein Leichman FEBRUARY 7, 2021, 2:10 PM
Even with Israel’s world-leading rollout of Covid-19 vaccinations, drugs to treat Covid patients are in desperate need across the world.
Two such drugs developed in Israel show great promise in clinical trials: EXO-CD24 and Allocetra.
EXO-CD24, an experimental inhaled medication developed at Tel Aviv Sourasky Medical Center, cured all 30 moderate-to-severe cases in a Phase I clinical trial.
Developed over the past six months at the hospital, EXOCD24 stops the “cytokine storm” – where the immune system goes out of control and starts attacking healthy cells – that occurs in the lungs of 5-7% of Covid-19 patients.
“To date, the preparation has been tried with great success on 30 severe patients, in 29 of whom the medical condition improved within two to three days and most of them were discharged home within three to five days. The 30th patient also recovered but after a longer time,” the hospital reports.
“The drug is based on exosomes, [vesicles] that are released from the cell membrane and used for intercellular communication. We enrich the exosomes with 24CD protein. This protein is expressed on the surface of the cell and has a known and important role in regulating the immune system,” explained Dr. Shiran Shapira, director of the laboratory of Prof. Nadir Arber, who has been researching the CD24 protein for over two decades.
“The preparation is given by inhalation, once a day, for only a few minutes, for five days,” Shapira said.
She said the experimental treatment has two unique characteristics. The first is that it inhibits the over-secretion of cytokines. The second is that it is delivered directly to the lungs and therefore has no systemic side effects that injected or oral drugs can cause.
“Even if the vaccines perform their function, and even if no new mutations are produced then still in one way or another the corona will remain with us,” said Arber, director of the medical center’s Integrated Cancer Prevention Center. “To this end, we have developed a unique drug, EXO-CD24.”
Arber added that this advanced preparation “can be produced quickly and efficiently and at a very low cost in every pharmaceutical facility in the country, and in a short time globally.”
Prof. Ronni Gamzu, CEO of the medical center, said, “Prof. Arber’s results for first-phase research were excellent and gave us all confidence in the method he has been researching [here] for many years. I personally assisted him in further obtaining the approvals from the Ministry of Health for further research.”
Allocetra
Meanwhile, Enlivex Therapeutics last week reported positive results from a multi-center Phase II clinical trial of its experimental Covid-19 immunotherapy drug Allocetra in severe and critical Covid-19 patients.
We reported in October that five Covid-19 intensive care patients were discharged from Hadassah University Medical Center in Jerusalem after treatment withAllocetra.
Nine severe and seven critical Covid-19 patients were treated with Allocetra in the Phase II clinical trial. Fourteen of them recovered and were discharged from the hospital after an average of 5.3 days.
The Phase II trial originally was expected to enroll 24 patients but was “completed early in support of anticipated accelerated regulatory filings of the trial’s positive safety and efficacy data,” Enlivex reported.
Altogether, 19 out of 21 Phase II and Phase Ib Allocetra trial patients recovered and were discharged from the hospital after an average of 5.6 days. Most of the patients in both studies had pre-existing risk factors such as male gender, obesity and hypertension.
“The results we have seen from the 12 Covid-19 patients treated to date with Allocetra are exciting,” said Prof. Vernon van Heerden, head of the General Intensive Care Unit at Hadassah and the lead investigator of both clinical trials.
“The Phase II patients who have been discharged from the hospital are currently healthy. We believe that these compelling results have demonstrated the safety and efficacy of Allocetra in these complicated patients, highlighting the potential of Enlivex’s product candidate to benefit severe and critical Covid-19 patients as well as others suffering from cytokine storms and organ dysfunctions across various clinical indications.”
Allocetra is based on the research of Enlivex chief scientific and medical officer Dr. Dror Mevorach, head of internal medicine and of one of Hadassah’s coronavirus wards. It works by restoring balance to the immune system.
Mevorach said Allocetra “may have utility as a safe and efficacious treatment … regardless of the specific coronavirus mutation that afflicted the patients, and across different life-threatening, high mortality clinical indications with high unmet medical needs.”
China National Medical Products Administration Approves MR-guided Focused Ultrasound To Treat Essential Tremor And Tremor-dominant Parkinson's Disease
https://www.prnewswire.com/il/news-releases/china-national-medical-products-administration-approves-mr-guided-focused-ultrasound-to-treat-essential-tremor-and-tremor-dominant-parkinson-s-disease-803587279.html
Insightec is strategically positioned for expansion in the China market
HAIFA, Israel and MIAMI, Feb. 10, 2021 /PRNewswire/ -- Insightec®, a global healthcare company focused on creating the next generation of patient care, announced it received market approval by The National Medical Products Administration (NMPA), the Chinese agency for regulating drugs and medical devices (formerly the China Food and Drug Administration, or CFDA).
The approval for the Exablate® 4000 (Exablate Neuro) platform, will enable unilateral Focused Ultrasound treatment for patients living with debilitating tremor from Essential Tremor and Tremor-dominant Parkinson's Disease that has not responded to medications.
"This achievement reflects positive momentum to bring Focused Ultrasound to movement disorder patients in China," commented Maurice R. Ferré MD, Insightec CEO and Chair of the Board of Directors. "We are committed to improving the quality of life of patients in China and worldwide with impactful therapeutic treatments."
MR-guided Focused Ultrasound (MRgFUS), also referred to as high-intensity Focused Ultrasound, delivers acoustic energy precisely to the Vim of the thalamus, considered to be responsible for tremor, to heat and ablate the tissue. The procedure does not require surgical incisions, implants or anesthesia.
Nearly 60 leading neurosurgeons in medical centers around the globe are treating patients with hand tremor from Essential Tremor or Tremor-dominant Parkinson's Disease on a regular basis with the Exablate Neuro. Performed in a single session in an MRI suite, many patients experience immediate tremor relief with minimal side effects, returning home the same day.
"There are millions of people in China living with diminished function due to hand tremors from Essential Tremor and Tremor-dominant Parkinson's Disease," said Peng Qiu, Insightec China Country Manager. "Our elderly communities will now have an option that uses highly advanced technology that is less invasive."
Exablate Neuro has received marketing approval for Essential Tremor from the U.S. Food and Drug Administration (FDA) and regulatory bodies in Argentina, Australia, Canada, Chile, Europe, Israel, Japan, Kazakhstan, Korea, Russia, Taiwan and Turkey.
Insightec's Exablate Body system is used in 20 hospitals across China to treat symptomatic uterine fibroids.
Fifteen Subjects Recruited to Bilateral Focused Ultrasound Study for Essential Tremor Pivotal Clinical Trial Sponsored by Insightec
MIAMI and HAIFA, Israel, Feb. 4, 2021 /PRNewswire/ -- Insightec, a global healthcare company focused on creating the next generation of patient care, today announced that it has received FDA IDE approval to conduct a pivotal study to evaluate staged, bilateral Focused Ultrasound treatment of patients with medication-refractory Essential Tremor. Fifteen subjects have been recruited to the study to date.
Focused Ultrasound guided by magnetic resonance imaging (MRI) allows treating a target deep in the brain with no incisions. Insightec's Exablate Neuro Focused Ultrasound device received U.S. Food and Drug Administration approval in 2016 to treat medication-refractory Essential Tremor in one hand, or unilaterally. The current clinical trial is evaluating the safety and efficacy of bilateral (both sides of the body) treatment in subjects 22 years or older with a diagnosis of medication-refractory Essential Tremor and who had unilateral Focused Ultrasound treatment at least nine months earlier.
"When Essential Tremor patients have significant tremor on both sides of their body, a staged, bilateral Focused Ultrasound procedure may offer clinical benefit. This is an important study to evaluate a second Focused Ultrasound treatment performed at least 9 months after the first arm was treated," commented Howard Eisenberg, MD, the Raymond K. Thompson, MD Chair in Neurosurgery, Professor, Department of Neurosurgery, University of Maryland School of Medicine, and neurosurgeon at the University of Maryland Medical Center.
Currently, the clinical trial (NCT04112381) is being conducted at the University of Maryland School of Medicine, Weill Cornell Medicine, University of Pennsylvania, and University of Virginia with additional centers planned.
"Incisionless surgery is helping people living with debilitating tremor return to a life of independence," said Maurice R. Ferré MD, Insightec CEO and Chair of the Board of Directors. "We will continue to collaborate with leading medical centers around the globe and support clinical research to expand treatment options for improved quality of life and quality of care."
Media Contact:
Lyndsay Isaksen
G&S Business Communications for Insightec
Tel.: +1 917 595 3032
E-mail: lisaksen@gscommunications.com
New Israeli drug cured 29 of 30 moderate/serious COVID cases in days — hospital
Medicine developed at Ichilov moderates immune response, helps prevent deadly cytokine storm, researchers say; 29 of 30 phase 1 trial patients left hospital within 3-5 days
Medicine developed at Ichilov moderates immune response, helps prevent deadly cytokine storm, researchers say; 29 of 30 phase 1 trial patients left hospital within 3-5 days
https://www.timesofisrael.com/new-israeli-drug-cured-moderate-to-serious-covid-cases-within-days-hospital/
Israel continues to shine as hub of med-tech innovation
https://www.bioworld.com/articles/502209-israel-continues-to-shine-as-hub-of-med-tech-innovation
January 8, 2021
By Meg Bryant
In recent years, Israel has proven to be a hot spot for med-tech innovation. The country boasts more than 1,500 companies in the health care and life sciences sectors, roughly 70% in medical devices and digital health. More than a hundred new companies focused on medical technology launch each year.
Israel, with a health care technology market valued at $6.2 billion, according to the U.S. International Trade Association, has combined its entrepreneurial spirit with technological curiosity and savvy to create life-altering and life-saving solutions. Contributing to its strength is its interdisciplinary capabilities, which bring together medicine, clinical expertise, materials science, electronics, software expertise and engineering skills.
“Israel is an incredible powerhouse for innovation and entrepreneurship,” Maurice Ferré, chairman and CEO of Insightec Ltd., told BioWorld. “I think this is largely rooted in the culture – a fairly young country that has survived many challenges. Using technology as a catalyst for transformation permeates throughout the country, from agriculture, to energy, to health care.”
The country’s world-renowned research universities and medical centers also serve as valuable partners, contributing to the development, testing and application of novel technologies.
Insightec
Tirat Carmel-based Insightec produces magnetic resonance (MR)-guided focused ultrasound equipment. The company’s Exablate Neuro incisionless neurosurgery platform is the first such device to gain U.S. FDA approval for two movement-related neurological indications – medication-refractory essential tremor in 2016 and medication-refractory tremor from Parkinson’s disease in 2018. The platform is CE-marked for both indications, as well as for neuropathic pain.
Last March, Insightec inked definitive agreements for a $150 million series F financing led by Koch Disruptive Technologies (KDT), a subsidiary of Koch Industries. The round had a post-money valuation of $1.3 billion.
Brett Chugg, managing director at KDT, saw potential in Insightec early on. “We recognized that Insightec was going to be very disruptive, with an order of magnitude better outcomes and order of magnitude lower costs for patients and health care providers,” he told BioWorld. “Insightec’s technology puts patients’ wellbeing first by disrupting the traditional approach to treating neurological diseases – like essential tremor, the most common movement disorder – that can have a significant impact on quality of life.”
In August, KDT added a second Israeli med-tech venture to its portfolio, leading a $71 million series D round in Sight Diagnostics Ltd. Based in Tel Aviv, the company makes the first direct-from fingerstick complete blood count analyzer approved by the FDA. The system’s artificial intelligence (AI) engine uses machine vision that assesses blood imagery to identify cell types and anomalies – essentially turning a sample into more than 1,000 images – at the point of care.
Silicon Wadi
Israel’s innovation prowess has earned it the nickname Silicon Wadi. “In a country that is fairly low in natural resources, we have had to pursue innovation in many industries to successfully compete in the global market,” Rafi Fisher, spokesman for Orcam Technologies Ltd., told BioWorld. “While much of Israeli innovation previously focused on ‘classical’ high tech – cybersecurity, telecommunications, software development, etc. – over the last decade or so, med tech has become a focal point of Israeli innovation.”
The industry also benefits from defense-based R&D. Many startups employ former military personnel from the renowned 8200 intelligence unit, and more and more of those are finding a home in med tech, Fisher said. Orcam itself boasts a team of nearly 200 experts focused solely on assistive technology development for people with various physical impairments.
Orcam harnesses highly advanced AI-driven computer vision and machine learning to provide increased independence to people who are blind or visually impaired, have reading challenges or hearing loss. The finger-size Orcam Myeye device affixes to eyeglasses and communicates elements of the visual world, such as printed or digital text and currency, to the user via near real-time audio. It also recognizes faces, products and colors.
To date, Orcam has raised $130.4 million. The company is planning for a pre-IPO funding round sometime in 2021, Fischer said.
The sector has also seen some big IPOs. That was evidenced by the $625.2 million raised by medical imaging startup Nano X Imaging Ltd. (Nanox) in its August public debut. The company’s low-cost Nanox.Arc is a “full-body, multipurpose, tomosynthesis X-ray system that we believe is capable, once approved by the applicable regulatory bodies, of delivering medical imaging for a wide range of clinical procedures, inside and outside of hospitals and larger imaging centers,” Ran Poliakine, Nanox’s founder and CEO, told BioWorld, noting the company has contracted deployments of 5,150 systems with nine service providers in 13 countries, contingent upon regulatory approvals. Additionally, Nanox has inked agreements aimed at deploying another 5,500 units in the U.S., South Korea and Vietnam, using its managed software as a service (MSaaS) business model.
The technology is derived from field emission display technology that was originally developed by Sony.
Nanox is awaiting a decision on its 510(k) submission to the FDA for a single-source version of Nanox.Arc and plans to file an application for a multi-source version by mid-2021. Efforts to obtain CE marking are also underway.
“Our goal is to globally deploy 15,000 Nanox.Arc units by the end of 2024,” Poliakine said.
More success stories
Meanwhile, numerous other ventures are solidifying Israel’s reputation as a med-tech hot spot, some of them leading to major collaborations.
Rehovot, Israel-based Todos Medical Ltd. is developing blood tests for the early detection of cancer and neurodegenerative diseases using Fourier-transform infrared spectroscopy. Last year, it announced plans to apply the technology in diagnosing infections, including COVID-19.
Another example is Haifa-based Bluewind Medical Ltd., which has built the Renova Istim implantable neuromodulation system for the treatment of overactive bladder, a problem that affects an estimated 20% of people worldwide. The company is enrolling patients in the U.S. and Europe in a pivotal trial.
In December, Zebra Medical Vision Ltd., of Shefayim, won FDA clearance for Healthpoint, an AI solution to extract bone measurements from X-ray scans. The tool is intended to aid in planning orthopedic surgical procedures.
Also last month, Emedgene Technologies Ltd., of Tel Aviv, unveiled a nonexclusive partnership with San Diego-based Illumina Inc. and the integration of automated interpretation into Illumina’s Trusight Software Suite for rare genetic diseases. The company’s Clinical Rare Disease application is one of a line of AI-powered applications for high-throughput genomic interpretation across clinical and research use cases.
And Envizion Medical Ltd., which has developed electromagnetic fields-guided feeding tube placement technology, reported in September that it was partnering with a major U.S. hospital chain to bring the FDA-cleared Envue navigation device to 150 hospitals nationwide to help meet demand during the coronavirus pandemic.
One of the technological sources for electromagnetic navigation is based on knowledge and devices sourced from the Israeli army, Envision CEO Doron Besser told BioWorld. “Add highly experienced investors, accumulated experience and a supportive government, you get the perfect recipe!” he wrote.
Supportive environment
All are examples of promising Israeli startups that could help to reduce the country’s dependence on imported medical devices. Helping to move med-tech innovation forward is an array of accelerators, incubators and government-funded programs.
Adi Jacobson, vice president of marketing at Zebra Medical Vision, said incubators and accelerators are a valuable source of services, tools and funding for startups. They play “an important role in ‘sipping’ out the best concepts from the sea of young Israeli startups and give them an early days edge,” he wrote in an email. “If you look closely at those startups who succeeded in growing to profitable companies, you’ll see that a strong visionary founding team and a product fit to a real market pain point is common to them all, which accelerators bet on in an early stage.”
“Israel is a great testbed for new and upcoming innovations: It has a population with high consumption of health services, it’s healthcare system is relatively concentrated (with 4 HMOs serving most of the country, so decisions are taken in a centralized fashion,” Einat Metzer, CEO and co-founder of Emedgene, told BioWorld. “The cultural mindset stresses the importance of being at the forefront of progress, and so decision-makers would always have an interest in getting the latest and greatest in any field of medicine. This means that health care entrepreneurs have relatively easy access to explore their concepts and perform early validation.”
Israel’s concentrated health care system also helps to support rapid innovation. “Most people stick with their HMO from cradle to grave, giving each HMO tremendous access to patient date,” Fisher said. “All of this data is recorded by each HMO’s completely interoperable electronic health record system.”
This rich trove of patient data contributes both to Israeli innovators’ ability to rapidly develop AI- and machine learning-based solutions and the ease with which they can be rolled out.
“We absolutely see other opportunities for investing in Israeli med-tech ventures,” said KDT’s Chugg. “If you look at what’s going on right now with the coronavirus pandemic, it has exposed many weaknesses to our health care system and shows how much opportunity there is to improve in terms of quality of care, reducing costs, or both.”
Philips & InSightec join forces to usher in the future of patient care
Insightec builds on 2020 momentum collaborating with one of the world's largest diagnostic imaging conglomerates...
Dor Schwartz / 20 Dec 2020 •
Philips & InSightec join forces to usher in the future of patient care
Israeli MedTech veteran InSightec, innovator of MR-guided Focused Ultrasound solution, and Royal Philips (NYSE: PHG), a multinational leader in health technology announced a collaboration to expand access to MR-guided Focused Ultrasound.
InSightec’s Exablate Neuro uses focused acoustic energy to precisely target and treat deep in the brain without incisions or implants. MR imaging provides high-resolution imaging and acts as a thermometer for real-time temperature monitoring. The incisionless treatment option is often performed in an outpatient setting. When used to treat Essential Tremor or Tremor-dominant Parkinson’s Disease, many patients demonstrated immediate tremor relief with minimal complications.
“InSightec is committed to expanding patient access for MR-guided focused ultrasound, which is rapidly being adopted as a standard of care worldwide for treating Essential Tremor, as well as continuing to develop a research pipeline for the treatment of many other disorders,” said Maurice R. Ferré MD, InSightec CEO and Chairman of the Board of Directors. “Partnering with Philips will help increase adoption of our innovative technology to transform patient care.”
In mid-2016, InSightec announced an agreement with Siemens Healthineers and then in 2018 received regulatory approvals for the Exablate Neuro platform compatible with several Siemens MRI scanners. Prior to that, the platform had been developed to be compatible with specific GE Medical scanners.
“As a leading innovator in MR imaging to support precise diagnosis and targeted therapies, we continue to expand the role of MR in providing precision guidance for minimally invasive therapy solutions into new treatment areas,” said Arjen Radder, General Manager for MR at Philips. “By partnering with InSightec we are expanding access to Philips’ advanced MR capabilities to a wider range of healthcare providers, including neurosurgery practices both in-hospital and in outpatient settings, as well as the global research community. Together, we aim to advance towards clear care pathways with predictable outcomes for every patient."
According to the agreement, InSightec and Philips expect market introduction for the compatible systems in 2023. InSightec closes out 2020 with yet another key milestone, following an up to $150 million KDT led Series F from March, in addition to securing national healthcare reimbursements in Japan and England.
https://www.geektime.com/philips-insightec-join-forces-to-usher-in-the-future-of-patient-care/
Philips and InSightec partner to expand access to MR-guided focused ultrasound for incisionless neurosurgery
December 17, 2020 04:00 ET | Source: Royal Philips
Companies to develop compatibility of Philips’ MR Ingenia 3.0T and 1.5T systems with the InSightec Exablate Neuro platform
Amsterdam, the Netherlands and Haifa, Israel – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology and InSightec, a global healthcare company focused on the therapeutic power of acoustic energy, today announced a collaboration to expand access to MR-guided focused ultrasound for incisionless neurosurgery. By developing compatibility between Philips’ advanced MR systems and the Exablate Neuro platform from InSightec, the two companies will support expanded access to MR-guided focused ultrasound for the treatment of Essential Tremor and other neurological disorders. The expansion of access to this innovative treatment option has the potential to significantly improve the patient and staff experience, enhance health outcomes and lower the cost of care.
Essential Tremor is a neurological condition that typically causes shaking of the hands, head and voice, and may also cause shaking of the legs and trunk. The tremor begins mildly and progresses over time, and in some patients, is severe enough to interfere with daily activities, such as personal care, eating or getting dressed. Essential Tremor is one of the most common movement disorders in the world [1]. MR-guided focused ultrasound uses high-intensity sound waves, guided by magnetic resonance imaging, to target and treat the area of the brain responsible for the tremor. The incisionless treatment option is often performed in an outpatient setting, with many patients showing immediate tremor relief with minimal complications.
“As a leading innovator in MR imaging to support precise diagnosis and targeted therapies, we continue to expand the role of MR in providing precision guidance for minimally invasive therapy solutions into new treatment areas,” said Arjen Radder, General Manager for MR at Philips. “By partnering with InSightec we are expanding access to Philips’ advanced MR capabilities to a wider range of healthcare providers, including neurosurgery practices both in-hospital and in outpatient settings, as well as the global research community. Together, we aim to advance towards clear care pathways with predictable outcomes for every patient.”
“InSightec is committed to expanding patient access for MR-guided focused ultrasound, which is rapidly being adopted as a standard of care worldwide for treating Essential Tremor, as well as continuing to develop a research pipeline for the treatment of many other disorders,” said Maurice R. Ferré MD, InSightec CEO and Chairman of the Board of Directors. “Partnering with Philips will help increase adoption of our innovative technology to transform patient care.”
The partnership will involve the development of compatibility between the InSightec Exablate Neuro platform and Philips’ MR Ingenia Elition 3.0T, Ingenia 3.0T and Ingenia 1.5T systems. This includes the Ambition 1.5T MR, the industry’s first and only commercially available fully sealed magnet for more productive, helium-free operations [2]. Philips’ MR therapy portfolio also includes its Ingenia MR-OR intraoperative MR for use during neurosurgical procedures, as well as its leading MR-only radiotherapy applications, which enable clinicians to quickly execute complete imaging protocols for prostate, female pelvis, brain, head and neck, and spine.
The parties expect market introduction for the compatible systems in 2023 with offerings available for both existing and new customers.
[1] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2938289/
[2] Compared to the Ingenia 1.5T ZBO magnet.
For further information, please contact:
Mark Groves
Philips Global Press Office
Tel.: +31 631 639 916
E-mail: mark.groves@philips.com
Lyndsay Isaksen
G&S Business Communications for InSightec
Tel.: +1 917 595 3032
E-mail: lisaksen@gscommunications.com
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people's health and well-being, and enabling better outcomes across the health continuum – from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Philips generated 2019 sales of EUR 19.5 billion and employs approximately 81,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.
About InSightec
InSightec is a global healthcare company creating the next generation of patient care by realizing the therapeutic power of acoustic energy. The company’s Exablate® Neuro platform focuses sound waves, guided by MRI, to provide tremor relief to patients with Essential Tremor and Tremor-dominant Parkinson’s Disease. Research for future applications in the neuroscience space is underway in partnership with leading academic and medical institutions. InSightec is headquartered in Haifa, Israel, and Miami, with offices in Dallas, Shanghai and Tokyo. For more information, please visit: www.insightec.com.
19-Nov-2020 INSIGHTEC® incisionless MRgFUS surgery to be funded by NHS England to treat Essential Tremor condition that causes shaking of the hands, head and voice, but it can also cause legs and trunk to shake. The tremor begins mildly and progresses over time to become more severe, and in some patients, severe enough to interfere with daily activities, such as eating and writing.
“This is extremely exciting news for those living with Essential Tremor,” said Prof Wady Gedroyc, MD, Consultant Radiologist at London’s St. Mary’s Hospital. “Essential Tremor can be incredibly debilitating and impacts virtually every aspect of a person’s life. There’s a significant unmet need in terms of treatment options so I am delighted that NHS England has recognised the value that this new technology can bring to patients. I am looking forward to seeing the first NHS patients from April next year.”
MRgFUS is an incisionless treatment option for Essential Tremor, often performed in an outpatient setting, enabling patients to return home the same day. High intensity ultrasound waves, guided by MRI, converge at a small target to create a permanent lesion in the specific area of the brain responsible for the tremor.
“Medicines only work in approximately 50% of people with Essential Tremor, and so to have another treatment option available that is incisionless is a major breakthrough for this patient group,” commented Prof Dipankar Nandi, MD, Consultant Neurosurgeon, Imperial College Healthcare NHS Trust. “Now that the NHS has agreed to make focused ultrasound available from next April, it is critical that we ensure equity of access across the country, and not just for those that can make it to London.”
“NHS funding is an important step in expanding patient access to Focused Ultrasound,” said Maurice R. Ferré MD, INSIGHTEC CEO and Chairman of the Board of Directors. "Most importantly, people living with tremor can now get back their independence and their lives.”
https://www.pharmiweb.com/press-release/2020-11-19/insightec-incisionless-mrgfus-surgery-to-be-funded-by-nhs-england-to-treat-essential-tremor
Insightec is a daughter company of EMITF
Lantheus Enters into Strategic Collaboration with Insightec for Use of Microbubbles in Combination with MR-Guided Focused Ultrasound for Treatment of Glioblastoma and Neurodegenerative Conditions
Published: Oct 29, 2020
NORTH BILLERICA, Mass.--(BUSINESS WIRE)-- Lantheus Holdings, Inc. (the “Company”) (NASDAQ: LNTH), parent company of Lantheus Medical Imaging, Inc. (“LMI”), a global leader in the development, manufacture and commercialization of innovative diagnostic imaging agents and products, today announced a strategic collaboration with Insightec Ltd., a global healthcare company focused on the therapeutic power of acoustic energy. Under the agreement, Lantheus’ microbubbles will be used in combination with Insightec’s investigational MR-guided Focused Ultrasound (MRgFUS) platform to evaluate treatment of glioblastoma and neurodegenerative conditions. The collaboration directly aligns with key Lantheus growth strategies of pursuing new applications for its microbubble franchise and expansion into oncology.
Under the terms of the agreement, Lantheus’ microbubbles will be used together with Insightec’s MRgFUS platform designed to deliver ultrasound energy deep in the brain with no incisions. The microbubbles, combined with the application of focused ultrasound, are being investigated in the temporary disruption of the blood brain barrier to potentially allow for more effective delivery of chemotherapeutic agents in the treatment of glioblastoma patients. Glioblastoma is the most aggressive malignant primary brain tumor affecting 3.19 Americans per one hundred thousand and has a less than 5% five-year survival rate.1
Lantheus will supply its microbubbles and activation devices while Insightec will be responsible for future regulatory submissions and commercialization. The deal terms include a transfer price and royalties.
“We are thrilled to partner with Insightec to further expand our microbubble franchise within oncology and potentially neurodegenerative conditions,” said Mary Anne Heino, President and Chief Executive Officer of Lantheus. “Our collaboration with Insightec could help change the paradigm in glioblastoma, an aggressive cancer for which patients and clinicians unfortunately have few options, and where the unmet medical need is truly significant.”
“Focused ultrasound has strong potential to transform the treatment of diseases that currently have poor outcomes and few options. Partnerships, like this one with Lantheus, will help Insightec’s future customers push the boundaries of what is possible in providing care to patients with neurooncologic as well as neurodegenerative diseases,” said Maurice R. Ferré MD, Chairman and CEO of Insightec.
About Glioblastoma
There are approximately 12,500 new cases of glioblastoma in the United States each year. Glioblastoma is the most commonly occurring primary malignant brain tumor, accounting for over 48% of all malignant tumors.2 Overall, median survival for glioblastoma patients is 12-15 months. Glioblastoma has a poor prognosis due to its high rate of tumor recurrence, which has been described clinically as inevitable.3 Worldwide, there are more than 240,000 estimated deaths from brain and central nervous system cancers each year, and glioblastoma represents one of the most common and lethal forms of these cancers.4
About Lantheus Holdings, Inc.
Lantheus Holdings, Inc. is the parent company of Lantheus Medical Imaging, Inc., Progenics Pharmaceuticals, Inc., and EXINI Diagnostics AB, and a global leader in the development, manufacture and commercialization of innovative diagnostic and therapeutic agents and products. Lantheus provides a broad portfolio of products, including the echocardiography agent DEFINITY® Vial for (Perflutren Lipid Microsphere) Injectable Suspension; TechneLite® (Technetium Tc99m Generator), a technetium-based generator that provides the essential medical isotope used in nuclear medicine procedures; AZEDRA® for the treatment of certain rare neuroendocrine tumors; and RELISTOR® for the treatment of opioid-induced constipation, which is partnered with Bausch Health Companies, Inc. The Company is headquartered in North Billerica, Massachusetts with offices in New York, New Jersey, Puerto Rico, Canada and Sweden. For more information, visit www.lantheus.com.
https://www.biospace.com/article/releases/lantheus-enters-into-strategic-collaboration-with-insightec-for-use-of-microbubbles-in-combination-with-mr-guided-focused-ultrasound-for-treatment-of-glioblastoma-and-neurodegenerative-conditions/
|
Followers
|
14
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1367
|
|
Created
|
09/23/05
|
Type
|
Free
|
| Moderators | |||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
