Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Sure is, find any real proof of either! ![]()
Naaw, sorry you lost on that one.
It's been what (?) 16 years that HALB has been a major loser, liar and fraud, (proven). And now its concussion-aide nasal spray that's going to prove a profit.
Nope, never. Take that to the bank.
They can't even provide a shred of official evidence there's even trial testing happening.
I'm not a hater, I provide honest trips. When I saw what was really going on, and how bashers here had the facts in hand. I got out, barely breaking even.
I come back once in a while, noting who's advice on this board to never take.
GLTY
Bit of a difference between Stage II clinical trail and phase II of a pre pilot study isn't there?😉
That's an internal release with no corroboration. Where are the published trials?
:) "Halberd Corp’s (OTC: HALB) Groundbreaking Traumatic Brain Injury (TBI) Mitigation Nasal Spray Shows Promising Phase II Initial Test Results" :)
Bet no one here can even find a Stage II Trials award document anywhere for HALB.
All ultimate fighting guys and gals can give themselves a squirt just before they get an elbow or foot kick to the head. :)
lol Just hit late Jan. price level. Maybe we can load some 30's again?
"just waiting for news" 😆
An item that all first responders should carry particularly paramedics and sports trainers however. All levels of sports all emergency crews everywhere!
" “We are optimistic that our successful Phase II testing will highlight the unique advantages of our patent-pending nasal spray...."
As far as encouragement goes, this part is all that matters at this time.
He did not say he was Confident and it is still Patent Pending. Should have just said, we need to sell some more shares, therefore, read this statement.
At like their previous endeavors, this will likely go nowhere IMO.
Let the Nay-Sayers chew on this for a while:
William A. Hartman, Halberd's Chairman, President, and CEO, added, “We are optimistic that our successful Phase II testing will highlight the unique advantages of our patent-pending nasal spray in preventing neurodegeneration following head trauma. Potential applications for Halberd’s nasal spray include military and civilian contact sports, law enforcement, hospitals, and emergency response units dealing with automobile accidents and falls. The market potential for this product is substantial if ongoing tests continue to support our initial optimism.”
These results are very encouraging to point of injury (POI) care. Administration of this novel TBI therapeutic translates to the potential of buddy care or self-administration via a developing auto-nasal injector under development at Halberd’s partner Athena GTX.
Set those shares with sell orders above a buck and keep the stocks from being played with by MM. :)
$HALB Halberd Corp’s (OTC: HALB) Groundbreaking Traumatic Brain Injury (TBI) Mitigation Nasal Spray Shows Promising Phase II Initial Test Results
https://www.globenewswire.com/en/news-release/2024/07/24/2917954/0/en/Halberd-Corp-s-OTC-HALB-Groundbreaking-Traumatic-Brain-Injury-TBI-Mitigation-Nasal-Spray-Shows-Promising-Phase-II-Initial-Test-Results.html
JACKSON CENTER, Pa., July 24, 2024 (GLOBE NEWSWIRE) -- Halberd Corporation (OTC-PINK: "HALB”) has announced encouraging preliminary outcomes from its Phase II testing at Mississippi State University (MSU) of its Tri-Ax™ nasal spray, aimed at mitigating the effects of traumatic brain injury (TBI) following head trauma.
Dr. Russell Carr, the project's Principal Investigator, commented: “In the early stages of our Phase II studies, we focused on assessing the efficacy of our innovative Tri-Ax™ nasal spray in producing positive outcomes when administered shortly after a TBI event. We included behavioral function as a key endpoint, which is significantly impacted by TBI.”
Dr. Carr elaborated, “Preliminary data reveal that higher impact forces lead to step-wise changes in motor activity levels. Notably, with one of the higher impact forces, TBI-induced motor activity changes were significantly improved when Tri-Ax™ was administered one hour post-impact. These findings suggest that the therapeutic benefits of Tri-Ax™ can be achieved when administered promptly after injury.”
William A. Hartman, Halberd's Chairman, President, and CEO, added, “We are optimistic that our successful Phase II testing will highlight the unique advantages of our patent-pending nasal spray in preventing neurodegeneration following head trauma. Potential applications for Halberd’s nasal spray include military and civilian contact sports, law enforcement, hospitals, and emergency response units dealing with automobile accidents and falls. The market potential for this product is substantial if ongoing tests continue to support our initial optimism.”
These results are very encouraging to point of injury (POI) care. Administration of this novel TBI therapeutic translates to the potential of buddy care or self-administration via a developing auto-nasal injector under development at Halberd’s partner Athena GTX.
That’s the registered agents address. That’s not their physical address
I would be expecting 100+ pennies when it's all ready for production. I can wait till then. :)
How about 100+ pennies.
Still waiting on the NEWS....DOD news will make this run into multiple pennies easily $$$
Just waiting for the news from the Halberd/AthenaGTX study. Athena is putting all the pieces of this puzzle together and looks to have their act together in dealing with the DOD.
I'm still locked and loaded. All of us need to set sell orders high and eliminate the MM from playing with our stocks. When we see acceptance we will be in buck land. :)
$HALB - "Halberd/AthenaGTX Data acquisition watches and sensors are up and running for the Watchdawg pilot study, ensuring comprehensive data collection. #HealthTech #Biotech"
$HALB Halberd/AthenaGTX Data acquisition watches and sensors are up and running for the Watchdawg pilot study, ensuring comprehensive data collection. #HealthTech #Biotech
— Halberd Corporation (@HalberdC) July 9, 2024
In yesterdays' tweet: IRB established.
The Institutional Review Board reviews and monitors biomedical research involving human subjects (Halberd is conducting clinical trials on Veterans and active duty personel). In accordance with FDA regulations, an IRB has the authority to approve, require modifications (to secure approval), or disapprove research.
$HALB - "Halberd/AthenaGTX The cloud-based platform for the Watchdawg study is up and functional, with psychology therapy in place and IRB established. #DigitalHealth #Biotech"
$HALB Halberd/AthenaGTX The cloud-based platform for the Watchdawg study is up and functional, with psychology therapy in place and IRB established. #DigitalHealth #Biotech
— Halberd Corporation (@HalberdC) July 8, 2024
"Halberd/AthenaGTX continue to recruit participants for the ongoing Watchdawg pilot study, now that compounded pharmaceuticals are manufactured and ready. #ClinicalTrials #Biotech #Watchdawg"
$HALB - "Halberd/AthenaGTX continue to recruit participants for the ongoing Watchdawg pilot study, now that compounded pharmaceuticals are manufactured and ready. #ClinicalTrials #Biotech #Watchdawg"
$HALB Halberd/AthenaGTX continue to recruit participants for the ongoing Watchdawg pilot study, now that compounded pharmaceuticals are manufactured and ready. #ClinicalTrials #Biotech #Watchdawg
— Halberd Corporation (@HalberdC) July 5, 2024
Apparently the Gouldings have slithered into another hole along with Tynan and Blum. Warning signs flashing IMO.
https://investorshub.advfn.com/boards/edit_msg.aspx?message_id=174702858
That means HALB will be getting something similar soon; either funding from Dept. of Defense, FDA approval, or even both.
Athena GTX was just granted FDA approval for their WVSM Pro Monitoring system.
Any news on the 2 gov. contracts?
Never mind, lol
Shall I put it into context for everyone? LOL
That's some funny shit, almost forgot about Premier Biomedical the first company Hartman ran into the ground. Thanks for sharing!
Know what's even funnier, 7 yrs. later and some fools are still buying into Hartman's miracle cures and technology.
[copy and paste from iHub at the stated time and date by Squid]
Squidster
Re: BOLD4GOLD post# 36303
Sunday, March 19, 2017 9:39:44 AM
Post# 36318
of 73675
Go
Mr. Hartman, I am heavily invested in your company and control more shares than any other individual on HUB. I want to commend you for keeping us informed of how your company and our investment is progressing. Looking forward to future updates.
Agreed. I had almost forgotten about his CBD venture.
Hartman should have stuck to peddling CBD patches 15 yrs ago, such a clown.
Never have had a basher operation going on at all. HALB has just been proven over and over, for close to 15 years now, to be a complete fraud. Fake videos, home office PR's, shooting their product line(s) all over the place, nothing of real substance.
How long have you been holding onto that bag?
They never give up Squid, will be drowning in Davey Jones' locker at the ocean's bottom by Thanksgiving.
This one scam operation has gone on for what, 15 years now?
Halberd's /Athena's treatment for PTSD is called WatchDawg. Athena coordinates all actions for the military, Halberd the non-military.
the program is being tested right now and is called WatchDawg
Athena also monitors the patient and gives feedback
Athena is adding AI and data collection
Patience grasshopper, patience
Jiminy Cricket once said " when you wish upon a star all your dreams will come true" Disney, seriously lmfao
Exactly what research have HALB/Athena GTX done? hmmmm
Not sure what qualifies as "industry behemoth" but I don't think estimated annual revenue between $5-10 million does it.
Not sure what qualifies as "massive sales contracts" but when you don't have product(s) to sell you don't have one.
When did working with a Yoga shop that promotes holistic healing for veterans become working with the U.S. Military?
How are those government contracts coming along?
Read the financials, disregard the fluff...
Hartman has yet to realize, after years of the same move, that no news is better than this sort of press release. We were trending up on the hope of good news coming soon. This sort of PR takes the wind out of the sails essentially saying we don’t have enough good information to anticipate releasing so we’ll try to buoy the price with something that we have on hand. Guarantee this is just a copy and paste of the one-sheeter they pass out to potential investors. Still long and strong HALB but Hartman can sure disappoint in the most naive ways.
$HALB Halberd/Athena GTX Research Promises a Much Brighter Future
https://www.accesswire.com/866120/halberdathena-gtx-research-promises-a-much-brighter-future
Walt Disney once said, "All our dreams can come true if we have the courage to pursue them".
JACKSON CENTER, PA / ACCESSWIRE / May 22, 2024 / Halberd Corporation (OTC PINK:HALB) in cooperation with Athena GTX, Inc is standing at the threshold of some major advancements to treat the issues facing not only our military but people around the world. In essence, the traumas of depression, anxiety, substance use disorders, and suicidal ideation can ruin our dreams, and this is the focus of our work. Is it easy? Absolutely not! But with patience and perseverance we are steadily progressing toward safe, easy to administer, efficacious solutions to these critically important issues.
Magnitude of the Suicide Problem:
The National Institute of Mental Health has stated that suicides in the Americas total 93,000 per year, or approximately 255 per day and over 10.6 per hour!!! Clearly, suicide is a huge problem in the general population as well as in the military, and is the basis for the military-focused House Bill sponsored by Congressman Zach Nunn of Iowa that passed the House by a unanimous vote. It has now gone to the Senate, where it is expected to pass overwhelmingly as well.
A recent article released by the Department of Defense concerns ongoing work in Alcohol and Substance Disorder Research. Here we focus on an exhaustiveexploration of the issue. Although this may not typify a normal Press Release, I thought it would be appropriate to relay some sobering statistics contained therein. The purpose of this is to allow our readers to truly grasp the magnitude of the problems that the Halberd and Athena technologies are addressing and the urgency of finding a viable solution to the crisis, not only in our own military, but globally. The sheer size of this 11-page DoD summary report and 39 noted references alone speaks to the magnitude of this crisis. However, this Press Release is intended to focus on the gaps in current treatment capability and how the Halberd/Athena methodology meets the needs of the PTSD & TBI problems.
We have known for quite a long time, for example, that 37% of those with a drinking problem also have mental health conditions (National Institute on Alcohol Abuse and Alcoholism (NIAAA), 2009). We also know that heavy alcohol use has historically been part of the culture of the military (Ames and Cunradi, 2004; Ames et. al. 2009; Bryant, 1979; and Ingraham, et.al. 2016). We know also that Post Traumatic Stress Disorder (PTSD) is a chronic debilitating anxiety disorder that may develop in both acute and chronic exposure to traumatic events; yet we are seeing almost twice the incident rates from post Iraqi War veterans now. In one study (Hoge, et. al. 2008) of Army infantry soldiers produced rates of PTSD up to nearly 44%, a ten-fold rate higher than the community incidence rates. Through our project maturation activities with our partners at Athena GTX, Inc. it is clear we are on the right track. Halberd's patented technologies and IP are aligned to these gaps.
Let's explore these three main gaps in current treatment identified by the Department of Defense in cooperation with the Veterans Administration and the National Institute of Health. First, we will paraphrase the identified gap and then describe Halberd's work in addressing those gaps.
Effective medications for substance abuse and co-morbidities- specifically, effective medications for the various core substances linked to abuse with and without the co-morbidities of TBI and PTSD.
LDN+: This novel combination of low dose Naltrexone and Cyclobenzaprine, patented by Halberd, has a half-century of medical usage behind it. While there are thousands of articles attesting to the analgesic effects, such as https://link.springer.com/article/10.1007/s10067-014-2517-2, all relate to the endorphin release as a contributing factor. The most prevalent hypothesis, advanced by Dr. Ian Zagon and colleagues, states that inducing a small and transient opioid blockade will prompt the body to compensate by upregulating both endogenous opioids and opioid receptors https://www.sciencedirect.com/science/article/abs/pii/S0306987708005070?via%3Dihub and anti-inflammatory capabilities at the neural level. While the use for chronic pain has been well described, we believe that acute pain can be modulated especially in combination with low dose Cyclobenzaprine. Cyclobenzaprine, in micro-dose has been ill-studied as well, but the effect is not dissimilar to low-dose Naltrexone, yet in combination, we have seen what we feel to be marked improvement over Naltrexone alone. In micro-dose, the side effects of Cyclobenzaprine are virtually non-existent. https://www.ncbi.nlm.nih.gov/books/NBK279656/ Note: While this product has been patented and used effectively and safely in patients, to be available on a commercial basis would involve a simple safety study for final submission to the FDA for approval, though currently can be compounded in physicians' offices for use for their patients. The key to our work now is to complete the studies as no commercial form of Low dose Naltrexone or low dose Cyclobenzaprine is available.
Effective psychotherapies for substance abuse and co-morbidities, specifically examining integrated and sequential approaches for addressing SUD and PTSD in military veteran populations.
There is no clearer call for solutions than this and our current pilot study directly aligns to this need. Our pharmacological interventions, when added to 24/7 physiological and psychological monitoring and help access, tied to providing state-of-the-art therapies including yoga and meditation is a game changer. The program is called Watchdawg™ and includes state-of-the-art assessment and therapies available to those suffering the most. We also know that the current thrusts are for troubled veterans to seek out-of-country "magic mushroom" therapies. We should and must do better with lower risk, lower cost alternatives. Watchdawg will do just that.
Effective complementary and integrated approaches to treating pain and co-morbidities in military/veteran populations (e.g., mindfulness, hypnosis/meditation, etc.), specifically testing a variety of non-pharmacological approaches to treating pain and co-morbidities in these populations.
We understand the issues and the individualism in patients and the general acceptance and resistance to COTs (Chronic Opioid Therapies) pharmaceuticals and adjunct therapies such as yoga, CBD, etc. Fact of the matter, we see the physiology and psychology of the individuals differ and yet often are related. Our Watchdawg program sits at the crossroads of therapies that work and the patient dynamics. Interestingly, LDN+ could also be therapeutic in the following conditions not outside of issues with active-duty military personnel and veterans:
Pain- an analgesic (pain-relieving) effect;
Neurological Conditions- beneficial for multiple sclerosis (MS) and Parkinson's by reducing inflammation and promoting neuro-protective effects improving the quality of life;
Mood and Mental Health- benefit for mood disorders such as depression, anxiety and PTSD, often a trigger for drug and alcohol abuse; Cancer - an adjunctive multi-layer program which may help modulate the immune system while providing an overall relaxation;
Modulation of the Immune System- has shown key immunomodulatory effects in rheumatoid arthritis, multiple sclerosis, and Crohn's disease; and sleep studies have reported improvements in sleep quality as lack of therapeutic sleep is common in military veterans.
These areas are a critical and timely area of need. We know this is a growing crisis now globally. According to DODReleases the Annual Report on Suicide in the Military: Calendar Year 2021, 20 Oct 2022, 519 Service members died by suicide with young, enlisted male Service members at greatest risk. This report shows that the suicide rates for Active Service Members have gradually increased since 2011. Additionally, in CY 2020, the report found that 202 dependents died by suicide, including 133 spouses and 69 other dependents.
Halberd's patented LDN+ is being tested as a potential multi-layer program for military-related PTSD/Suicidal Ideation. Halberd's patent-pending nasal spray, which produced outstanding results in limited animal studies at Mississippi State University is another potential winner in that it has shown potential to stop the cascade of neuro-degenerative events resulting from Traumatic Brain Injury and potential PTSD/Suicidal Ideation following head trauma by a reduction of Glial Fibrillary Acidic Protein (GFAP) of over 50% in Phase #1 tests. GFAP is the "Gold Standard" measure of TBI. It should be noted that elevated GFAP levels are also found in Multiple Sclerosis (MS), Parkinson's Disease, and Alzheimer's Disease patients. We believe the nasal spray could be a huge commercial success with applications to every war-fighter in the field, military MASH units and hospitals, all contact sports teams, civilian hospitals, ER units, and rest homes, where senior falls are another huge problem.
The future looks much brighter for PTSD & TBI patients because of Halberd/Athena GTX research.
Good news! I contacted Senator Hassen and received back an email acknowledging the Senate received H.R.3722 and it is now S.132. Senator Hassen said she will urge all her colleagues to vote for it in the full Senate.🤗🤗🤗
Right Said, Fred...I couldn't have expressed it any better or more succinctly myself....We appear to have a health industry that doesn't give a rip about food and a food industry that doesn't give a rip about health...To boot, lobbyists or other influence peddlers from both groups see to it that Congress does next to nothing about correcting this lurid state of affairs even though unnumbered millions suffer and die needlessly as a direct result of corporate indifference--and avarice...
Nefarious government agencies such as the Federal Death Agency (FDA) and the Covid Death Center (CDC), have colluded with Big Pharma to push garbage that does no one any good (clot shots). It's pay to play with these crooked agencies.
On the other hand - Halberd offers real solutions to real medical problems. They should rightly get an EUA.
yes thanks for reminding me,i am going to buy more
(responsible mod: Bill Moynihan/EHRjunkie)
Halberd Corp. (OTC PINK:HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements.
Halberd holds the exclusive rights to 4 granted U.S. Patents, no. 9,216,386 no. 8,758,287 and no. 11,186,629, no. 8,865,733
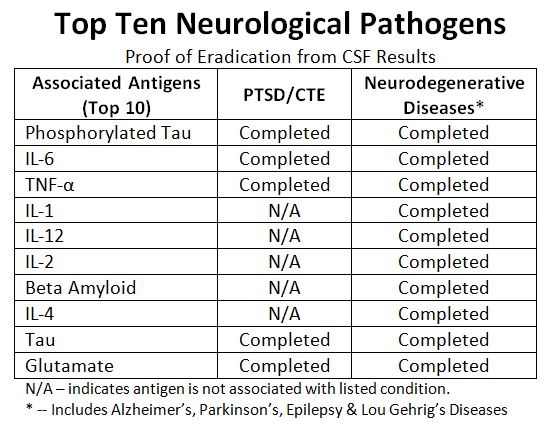
Halberd Corporation is a biotechnology company and with its partners (Dr. Qiang Shawn Chen and his team at Arizona State University; Dr. William Sturrus and his team at Youngstown State University; Dr. Richard Carr and his team at Mississippi State University), is developing an extra-corporeal (out of the body) treatment against PTSD, CTE, Alzheimer's Disease and other virusses and diseases. With the use of antibodies the disease antigens is targeted. Using radiofrequency / laser technology the "antibody-metallic moiety-antigens complex" is destroyed/eradicated. All based on 4 granted patents and 22 pending provisional/non-provisional (some PCT) patent applications.
2024: In close cooperation with partner (revenue sharing agreement in place) AthenaGTX, Halberd Corp is working on contracts from the military, to develop treatments to tackle Traumatic Brain Injury/TBI (patent pending nasal spray) and Post Traumatic Stress Syndrome/PTSD (patented Low-Dose-Naltrexone product).
JACKSON CENTER, PA / ACCESSWIRE / March 14, 2023 / Halberd Corporation (OTC PINK:HALB) is scheduled to meet with Center of Disease Control (CDC) representatives to discuss Halberd's incredible success in eradicating multiple strains of antibiotic resistant (AR) E. coli bacteria and Candida auris samples previously provided by the CDC.
JACKSON CENTER, PA / ACCESSWIRE / September 6, 2023 / Halberd Corporation's (OTC PINK:HALB) research at Mississippi State University (MSU) has shown significant reduction in two key markers of traumatic brain injury (TBI) in early testing of Halberd's patent-pending nasal spray to mitigate the effects of TBI. Anecdotal data in preliminary testing reveals a 20% reduction in Neuron Specific Enolase and a 50% reduction in the levels of Glial Fibrillary Acid Protein compared to the control group.
JACKSON CENTER, PA / ACCESSWIRE / September 20, 2023 / Halberd Corporation (OTC PINK:HALB) and Athena GTX, Inc, of Johnston, Iowa commenced collaboration on critical development of submissions for government contracts seeking proposals for the detection and treatment of traumatic brain injury (TBI). The initial focus will be on accelerating and fine-tuning Halberd's proprietary nasal spray for suppressing the effects of traumatic brain injury (TBI).
JACKSON CENTER, PA / ACCESSWIRE / December 13, 2023 / Halberd Corporation (OTC PINK:HALB) and CrodenHealth.com of Canada agree to produce, market, sell and distribute Halberd's patented low dose naltrexone combo product(s) worldwide. The agreement also allows CrodenHealth.com right of first refusal regarding similar rights to future products in Halberd's development pipeline.
JACKSON CENTER, PA / ACCESSWIRE / January 10, 2024 / Halberd Corporation (OTC-PINK:HALB) is thrilled to declare the formalization of a profitable revenue-sharing accord with Athena GTX, Inc., of Johnston, Iowa, (www.athenagtx.com). Athena GTX is a private enterprise specializing in providing medical care solutions tailored for first responders in both civilian and government sectors.
Halberd's / Athena's treatment for PTSD is called WatchDawg
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |