Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
I'm in MNKD after doing some DD. ![]()
Thank you!
We each have own way to trade and I've noticed a "pattern" in my own way: I spotted several companies, it's like I felt "attracted" to them, that I bought shares of and sold a few weeks or a few months later whereas they went much higher only 2-3 years later (PPS multiplied by 3 to 20) so yes, I forced myself to have some patience with IDIX. ![]()
I bought IDIX around $12 but a long time ago when BMY bought out Inhibitex and some people said IDIX could be bought out for around $25 per share with the same type of valuation. I didn't sell when it was clear it wouldn't happen right away...
Please do your DD and take a closer look at ELTP....
Thanks!
Indeed, the Yahoo message boards are full of spammers and nonsense so I find them hard to read also. There were people that seemed to do some good DD on the IDIX message board though.
For other companies, I don't know, I would have to do some DD which I don't have a lot of time for so my activity in the stock market is likely to be limited.
There will the obvious potential buyout candidates like ACHN and others that will be on everybody's radars but I'm going to wait to see if the prices come down in the case a buyout doesn't happen right away.
I'm going to check the company you mentioned too and I'm going to research and follow FNMA (and/or FMCC) closer because it could be delisted by the US governement and become private (I'm going to check that again) but I need to know what its valuation would be in that case and/or in the case the housing market keeps improving.
Then, I'm going to follow the VIX index because if the bull market begins to end at some point, it's likely to go up.
The last shares I'm holding of any company just now are from IHAI, a penny stock, but I don't expect anything from this company. It's still a remnant of mistakes of the past but it wouldn't make a lot of difference if I sold so I'm going to hold. Maybe IHAI is different from the other scams...
Thanks!
To be honest, I bought IDIX around $12 but a long time ago when BMY bought out Inhibitex and some people said IDIX could be bought out for around $25 per share with the same type of valuation. I didn't sell when it was clear it wouldn't happen right away and also didn't buy more shares when the PPS was around $3 to average down. Still, that's twice what I paid for the shares.
A few months ago, I found strange that IDIX went up to more than $9 on not a lot news so I didn't sell and then, Seth Klarman, the billionaire hedge fund manager, bought a lot of shares at $6.20 which was quite a high price considering IDIX went down to $3 several times and I found it "suspicious", like he knew something, so I held. ![]()
For the rest, I'm not trading very much anymore as I'm busy with other things and can't do much DD but, in any case, I'm a bit wary of this very long bull market.
Good for you champ ![]() .. Not easy to make money in this toppy market - congrats. Yest. I saw on CNBC that Merck bought IDIX for $24 & they flashed the stock quickly - was abt $9-10. Haha.. Didnt know stock so I had a quick look & nxt time I checked price it was $24!.. End of story..
.. Not easy to make money in this toppy market - congrats. Yest. I saw on CNBC that Merck bought IDIX for $24 & they flashed the stock quickly - was abt $9-10. Haha.. Didnt know stock so I had a quick look & nxt time I checked price it was $24!.. End of story.. ![]()
Good for you Flowing. I missed this one. They are very hard to spot. The Yahoo message boards are garbage as you have to sift threw all the crappy messages that are off topic. Picking Winners is a tough thing. I have one right now and it does not come around very often. Check the board out here and you can see how others think about MannKind MNKD. It's close to FDA approval. So perhaps I will get my payday.
What other stocks do you like at the present time? Any other Bio companies.
It wasn't a sleeper at all but the message board that was active was on Yahoo. I had 130 shares, not many, that I held through today. I made many "mistakes" in the stock market but not the one of selling IDIX when it went down again around $5 in the last months. ![]()
CNBC article explains the rationale for MRK-IDIX deal: #msg-103075542.
Does the Idenix deal suggest that Achillion is the next takeover target?
http://lastfinancier.com/impact-on-hcv-players-after-merck-acquired-idenix/
Congrats to all! Very nice profits$$$
Congrats Longs!!! Totally Super Sweet!
Wow! did anybody see this one coming. Since this board is lightly posted evidently they didn't. I wish that I onwned a position in this company. I have been following another company getting ready for FDA approval and can only hope that it reacts like this one did being bought. That stock is
MNKD Mannkind. It has an active board here on Investor Hub. Have to look for another sleeper like IDIX.
Congrats to all the shareholders.
May continued success continue.
TRUTH
Big!!!Ouch to the Weekend ShortTailMen$.
What an awesome move! Wish I had some!
unbelievable. Wish I had this one. Congratulations to those of you who do. This is the kind of thing us owners of biotechs dream of.
NVS’ equity stake in IDIX was 22% at 12/31/13: #msg-97352801.
IDIX @ Stockcharts
Solid steady movement!
Phase 2 news:http://finance.yahoo.com/news/idenix-pharmaceuticals-announces-initiation-enrollment-120000177.html
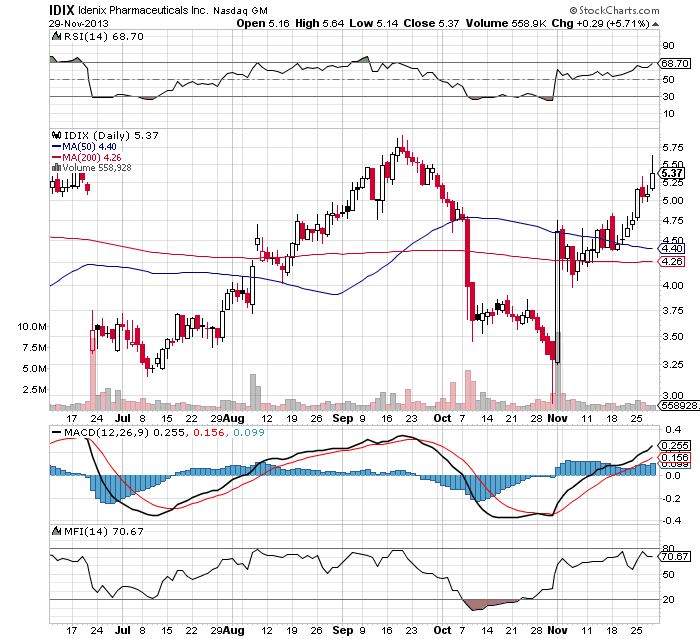
3.45 holding, 3.52 lod, early yet but we'll see
I like it. Support seems key here.
What was the 'Huge News'?
Huge news coming this week! A birdy told me!
~ $IDIX ~ Daily Par Sar Buy Signal ~ Criteria alert triggered during a recent trading session!

$IDIX has just triggered the "Parabolic SAR Buy Signals" scan criteria at Stockcharts.com
~ http://tinyurl.com/SAR-BUY ~



For a more in Depth study and DD profile, similar to the one contained in this link: ~ http://tinyurl.com/DDexample ~
Click the following link and type ticker or brief message asking me about the DD: ~ http://tinyurl.com/GET-THE-DD ~
What does the scan "Parabolic SAR Buy Signals" mean? Below is an image example and study link.
~ http://stockcharts.com/school/doku.php?id=chart_school:technical_indicators:parabolic_sar ~

To find other similar posts of "IDIX" utilize the links that follow.
Search MACDgyver's "Parabolic SAR Buy Signals" posts: ~ http://investorshub.advfn.com/boards/msgsearchbymember.aspx?searchID=251916&srchyr=2013&SearchStr=ParSarBuyScan ~
Search MACDgyver's posts for symbol "IDIX": ~ http://investorshub.advfn.com/boards/msgsearchbymember.aspx?searchID=251916&srchyr=2013&SearchStr=IDIX ~
Search Ihub for "IDIX" posts: ~ http://investorshub.advfn.com/boards/msgsearch.aspx?SearchStr=IDIX ~
For more in depth training and information visit Chartschool on the Stockcharts page.
~ http://stockcharts.com/school/doku.php?id=chart_school ~
Also don't forget the Ihub Edu Channel.
~ http://investorshub.advfn.com/boards/education.aspx ~


c
IDIX is recuperating and moving through a resistance point. Good sign. In my books.
$IDIX - Idenix Pharmaceuticals: Potential Multi-Bagger Means Little Risk/High Reward
http://seekingalpha.com/article/1581612-idenix-pharmaceuticals-potential-multi-bagger-means-little-risk-high-reward?source=email_investing_ideas&ifp=0
Editor's notes: IDIX is a biopharma play with substantial buyout opportunities and a strong research and product pipeline with as much as 146% upside.
Alpha-Rich articles are our best money-making long and short investment ideas.
They are released exclusively to Seeking Alpha Pro users 24 hours before publication.
Learn more about Seeking Alpha Pro.
Disclosure: I am long IDIX. (More...)
One of my favorite books that I have learned a great deal from in my investing journey is Seth Klarman's book "Margin of Safety: Risk Averse Value Investing Strategies For Thoughtful Investors." As the title indicates, the book drills in the idea of investing in companies with a margin of safety because doing so will give the lowest risk in losing capital. Seth doesn't jump on investment fad bandwagons but waits with a large cash position for prices to come down to what he is comfortable paying and is aware that "many of the forces that cause securities prices to depart from underlying value are temporary."
With Seth Klarman's consistent absolute returns year in and year out hovering near 20% for decades, even with large cash positions, I often find it instructive to look through his hedge fund Baupost Group for some investing ideas. One company Baupost has been investing in for years, and in large quantities recently, is Idenix Pharmaceuticals (IDIX). Baupost owns 18.48% of the company. At first, I questioned why a deep value investor with one of the greatest amounts of risk aversion would be investing in a company that has zero cash flow, funds itself through stock issuance and potentially might not make a successful product to justify any valuation. However, the more I read about Idenix, the more I understood the risk/reward potential and that the margin of safety is large considering the non-tangible assets the company possesses.
I will first describe what the situation is with Hepatitis C, its current treatment limitations and the opportunity that nuceloside/tide analogs and other direct acting treatments have in the treatment of HCV. I'll then take a closer look at Idenix's management, pipeline and underappreciated intellectual property, followed by a valuation of the company through take-out values of similar companies in 2011 and 2012 and a valuation as a going concern. This analysis shall show how the current negative perception of pharmaceutical companies focused on HCV next-generation treatment is well overdone and only temporary. A low end estimation of a buyout value would be a multi-bagger at 14.65 times the current price, which means a 10% chance of a buyout would equal 146% over the next year. A conservative estimate of the company on a stand-alone-basis, if a pan-genotypic drug is developed, could be worth similarly by 2017 leading to a 140% CAGR with many catalysts.
HCV Overview
Hepatitis C is a virus that mainly affects the liver and can go many years without being detected. The World Health Organization estimates that approximately 150 million people worldwide are chronically infected with HCV and three to four million people are infected each year. More than 350,000 people die every year from hepatitis C related liver diseases. I should note that the figures of Hepatitis C infection are extremely conservative because many people are unaware that they are infected with HCV. The result is that the true number of infected people, especially in developing countries, could be greatly larger than currently reported.
There are six different strands, or genotypes, of Hepatitis C that can be found in different populations throughout the world. The current treatment is highly ineffective and there are huge market opportunities for a drug that has higher effectiveness, lower substantial side effects, high barriers of resistance and can be used to treat a wider portion of the HCV-infected population.
Current HCV Treatment Limitations
Conventional treatment of HCV, an injection of a combination of pegylated interferon plus ribavirin, is quite unsuccessful in suppressing the HCV virus for extended periods of time, also called SVR or sustained virologic response. The rates of success for genotype 1, which accounts for 80% of HCV patients in the US and Europe, is extraordinarily low at less than 50%. To make the low success and inconvenience of administering PEG treatment even worse, conventional treatment leads to numerous substantial side effects such as fatigue, bone marrow suppression, anemia and neuropsychiatric effects.
In 2011 Victrelis and Incivek were approved for addition to the conventional treatment of genotype 1 and did increase SVR rates for patients who can tolerate the triple combination of the therapy. These two treatments are also not without side-effects. Side-effects even with these new drugs continue to be severe causing allergic reactions, fatigue and blood problems that could sometimes even be life-threatening.
Why Nucleoside/tide Analogs and Other Direct Acting Treatments Are Better
Conventional treatment with PEG is limited because they don't directly target the virus. Companies in development of next generation HCV therapies are focused on the development of protease inhibitors, polymerase inhibitors and NS5a inhibitors which inhibit the enzymatic activity of protease (NS3), polymerase (NS5a) and NS5a stopping the replication of the virus. Nucleoside/tide analog polymerase inhibitors show the most promise by blocking the synthesis of HCV RNA and stopping the replication of the virus.
Idenix Pharmaceuticals' goal is "…to develop all oral combinations of direct-acting antiviral, or DAA, drug candidates that will eliminate the need for interferon and/or ribavirin as currently used in the treatment for HCV."
Why Are Idenix Shares Depressed?
The market has recently been ungrateful to HCV's most promising treatment, nucleotide/side inhibitors, after numerous FDA holds and the death and hospitalization of test subjects in Bristol-Myers Squibb's (BMY) nucleotide inhibitor research. Idenix's lead nucleotide inhibitor IDX184, although blocked a few times by the FDA previously, was blocked in connection with Bristol-Myers Squibb's death and abandoned by Idenix's management because the drugs were from the same 2'- methyl guanosine nucleotide inhibitor family. More recently on Thursday evening, Vertex Pharmaceuticals (VRTX) reported that the FDA issued a partial hold on its nucleotide inhibitor VX-135 due to high liver toxicity in some patients. The current market view is that these nucleotide/side inhibitors are too toxic for the FDA to approve.
The market's current negativity towards nucleotide/side inhibitors has only been a recent affair. In 2011 and 2012, large pharmaceutical companies were on a buying spree picking up many of the smaller drug development companies with a nucleotide/side inhibitor in their pipeline. These large pharmaceuticals bought these small companies for a very large premium and for good reason - the development of an all-oral, direct acting, non-interferon HCV treatment will be a blockbuster treatment.
Pipeline and Intellectual Property
Now that Idenix has suspended its lead nucleotide inhibitor, all is lost, right? I think that is too quick of a conclusion. Idenix has a pipeline, research department and intellectual property that is, I believe, well above the companies that were acquired in 2011 and 2012.
Idenix's IDX-719 NS5a Inhibitor is the drug that is currently furthest along in IDIX's pipeline, currently in phase two studies with Janssen's non-nucleoside inhibitor TMC647055 and eventually simeprevir. IDX719 is special because it is the first NS5a by itself to show pan-genotypic activity while competitors NS5a's are focused towards genotype 1.
(click to enlarge)
A detailed comparison between IDX179 and BMS-790052 further shows the pan-genotypic effectiveness of IDX179 compared to other competitors' NS5a drugs. Below, the y-axis indicates the suppression of HCV replication and the x-axis indicates the different genotypes. The main goal of HCV treatment is to suppress virus replication, so lower replication values are better. We see that IDX719 shows significantly greater suppression of HCV replication across all genotypes when compared to BMS-790052.
Source: Idenix Presentation
NS5a inhibitors will play a part in next generation HCV treatment as a combination with nucleotide/side inhibitors with their low-potential for drug-drug interactions; however, the main focus goes back to nucleotide/side inhibitors with their unmatched high barriers to resistance and potency. Before I get into Idenix's new nucleotide, I think it would be helpful to talk about some chemistry. ATP (adenosine triphosphate) is necessary in the transport of genotoxic substances in the liver, however; the HCV virus uses ATP to replicate thereby lowering ATP. In pre-clinical work, one is able to determine the potency of a nucleotide/side through ATP (adenosine triphosphate) levels that are found in the liver, and Idenix's uridine nucleotide candidate shows great promise when compared to Gilead's GS-7977 in mice and monkeys. Below we see that liver triphosphate levels of Idenix's uridine nuc candidate in monkeys and mice are 25.36 times and 51 times greater than GS-7977, respectively.
Source: Idenix Presentation
Potency this high would allow Idenix to keep dosage rates low and Ron Renaud, CEO of Idenix, believes that the new uridine nucleotide inhibitor could be a once daily dose under 100mg and potentially under 50mg. This dosage can be compared with Vertex Pharmaceuticals' VX-135 which showed elevated liver toxicity with dosages at 400 and 200mg. Idenix also has two other non-2'methyl guanosine nucleotide/side inhibitors that are in pre-clinical stages which combined with the uridine nucleotide, provide numerous catalysts to share price.
Hidden Value
The biggest asset that I feel the market overlooks is Idenix's vast intellectual property that has been gathered in the search for the most promising nucleotide. Researchers at Idenix have had to synthesize thousands of nucleotides and figure out the best for treatment of HCV. That data that hasn't produced beneficial HCV treatment data, however, isn't useless. There are a number of other non-HCV ailments that could potentially benefit from nucleotide/side inhibitors. Previously Idenix wasn't able to look further into the development of nucleotide/sides for non-HCV treatments because of the Novartis (NVS) agreement that allowed Novartis access to all of Idenix's pipeline. After successfully renegotiating, Idenix is free from Novartis to pursue all avenues in drug development and keep their drugs. The Novartis agreement could have also been a deterrent in another company not acquiring Idenix in 2012. The CEO stated that since the renegotiation, third parties have been extremely interested in screening Idenix's library. I feel that large third party interest could mean that Idenix could sell some of its development library, make them that much more of an interest to be bought out or used in the development of drugs in the future. I won't say that I have the expertise to value or have the amount of information to attempt to value the intellectual property, but we can look at recent buyouts to come up with a value for Idenix.
Take-Out Value
There have been three buyouts of similar companies since 2011 that we will compare to Idenix. As shown below, Pharmasset was purchased by Gilead for $11 billion, Anadys was purchased by Roche for $230 million and Inhibitex was purchased by Bristol-Myers Squib for $2.5 billion.
Source: Various 10Ks Before buy-outs, IDIX most recent Q
I feel there are only a few ways of looking at the data, the amount of money spent on R&D and multiple paid on shareholders' equity. We see that Idenix is most similar to Pharmasset in regards to both of those metrics. To be conservative in my estimations, I took all of the multiples that were paid to shareholders' equity and averaged them, 43 is what I got. So, 43x shareholders equity of $219 million gives a value of $9.5 billion. To get an even more conservative estimation, I took the $9.5 billion and discounted it by 20% to account for potential hotness in the M&A activity at the time. So, I would estimate that in the case of buyout, the low-end valuation of Idenix would be roughly $7.5 billion, the mid valuation at $9.5 billion and the upper valuation above $11 billion. The current market cap of Idenix is $479 million, so my most conservative estimate of $7.5 billion would be 14.65x higher than today's price or shares would be worth $52 instead of $3.58. Even though I don't think that a buyout is highly probable in the next year or so, a 10% chance of a buyout at 14.65x our current price would still equal 146% upside.
Valuation In the Event of Business-as-is
Since I believe there is a greater chance that Idenix does not get bought out in the next year, I feel it is also necessary to conservatively estimate the value of the company running on a stand-alone basis. According to Renub Research, the Hepatitis C market is expected to grow more than 3 times by 2018. With the current market around $20 billion that would mean that the market would swell to $60 billion by 2018. It is pretty safe to say that other drug developers will be first to market their nucleotide/side inhibitor, however, Idenix is positioned to be one step ahead, as described above, with its next-generation all-oral pan-genotypic HCV treatment. A pan-genotypic drug would mean that Idenix could sell the same drug to more people worldwide, where half the world is not genotype 1. If by 2017 Idenix has only 15% of the HCV market with $9 billion in revenues and $1.5 billion in EBIT growing at 15% for 5 years followed by stable growth at 2% and these other inputs:
Source: My Rough Calculations
(click to enlarge)Source: My Rough Calculations
The above input approximations are based on Amgen in 2003 when they had $8 billion and a few drugs on the market, though my inputs are more conservative. These conservative approximations would lead to a valuation of $48 without taking into context the effect of Net Operating losses that Idenix has been generating which would be slightly offset by stock options and dilution to shareholders, which would be hard to approximate. A $48 value is slightly lower than my take-out value and a time frame of 3 years would mean this company on a stand-alone basis could generate a Compounded Annual Growth Rate of north of 140%.
Source: My Rough Calculations
Cash Position = Downside Protection
Idenix has a strong balance sheet of $205 million and zero debt. With that amount of money, it would be able to last through the middle of next year, complete IDX719, and get its uridine and other nucleotide inhibitors further down the pipeline. The company could issue more stock and with its potential upside, I think shareholders would be willing to take some dilution to keep the company running as they have done in the past. At a market cap of nearly $480 million, Idenix is trading for 2.34x cash, so I see very limited downside, especially when Seth Klarman and his large cash hoard is on the same side.
Management
First, management has their interests aligned with shareholders because they have stock options with exercise prices 200% higher than today's price. Also, I think it is very interesting to point out that the CEO Ron Renaud Jr. has an interesting background that might be conducive to shareholder friendliness. Ron spent two years from 2004 to 2006 as a senior analyst in the biopharama sector at JPMorgan Securities and was in the investor relations division in Amgen.
Risks
There are a number of risks but mainly the risk of the FDA putting holds on any of Idenix's pipeline will send the stock lower. Also, any hold instated on similar Idenix drugs developed by other companies could lead to share price declines. Stock dilution also is a risk. Until the successful development of a drug, this stock will be extremely volatile.
Conclusion
As Seth Klarman once said, "many of the forces that cause securities prices to depart from underlying value are temporary." He, as well as I, believe the temporary negativity surrounding Idenix Pharmaceuticals will pass, launching shares to the underlying value of the business. Any drug that moves forward in Idenix pipeline would send shares higher, and the potential for a multi-bagger makes this a company with good risk/reward characteristics.
Additional disclosure: This article is meant for instructional purposes and not meant as a recommendation to buy or sell. The only kind of intelligent investing is through your own due diligence.
I see IDIX going lower in the short term but I would wait for strong consolidation in a tight trading range to confirm a bottom. Looking at the weekly chart this is an absolute paradise for flippers where the chart looks like a wild roller coaster ride. An entry point here might be worth 25 percent of a position but it still seems a bit premature. This company is too cash rich to be trading at this low of a price and I believe there will be heady profits to take in a month or two. I am still sitting on the sidelines waiting to strike as I am not buying yet. I see red coming tomorrow and next week unless there is good news. I like the risk/reward ratio here with a cash rich company trading at 52 week lows because of the peace of mind of avoiding bankruptcy and when IDIX straightens out the setback with the FDA there will be fireworks. GLTU and happy trading.
What do you think IDIX can do next, I Know BS? The chart looks like it is trying to consolidate, but they really need news to attract speculators to take a toss on this.
As of 3/31/2013 they had 205 million cash on hand.
Biotechs seem to be LOTTO tickets. When the Feds are involved, anything can happen. Bet they have a PR out Monday morning. What is their cash on hand?
My take on the latest setback, FWIW: #msg-89254221.
Idenix says FDA delays hepatitis C drug trials
Idenix says FDA wants more safety data on experimental hepatitis C drug, delaying human trials
NEW YORK (AP) -- Idenix Pharmaceuticals Inc. said Thursday the Food and Drug Administration wants more safety data on its experimental hepatitis C drug IDX20963 before it will allow the company to begin human testing.
The company's shares plunged $1.39, or 27 percent, to $3.75 in aftermarket trading following the announcement.
Idenix said clinical trials are on hold until it addresses the FDA's concerns. The company did not say how long it believes clinical testing for IDX20963 might be delayed by the request for more preclinical data.
The Cambridge, Mass., company does not have any approved products. Idenix is running mid-stage clinical trials of another hepatitis C drug, IDX719, and is running preclinical trials of other potential drugs.
Shares of Idenix fell 3 percent to $5.13 Thursday as the broader markets slumped during the regular trading session.
8:01AM Idenix Pharma reports favorable resistance profile for IDX719, a potent, pan-genotypic HCV NS5A inhibitor, at EASL Meeting (IDIX) 3.95 : Co reported detailed resistance data from in vitro studies and from a three-day monotherapy clinical trial of IDX719, the co's once-daily, potent, pan-genotypic NS5A inhibitor for the treatment of hepatitis C virus (HCV) infection. These data are being presented on Saturday, April 27, in a poster session at the 48th Annual Meeting of the European Association for the Study of the Liver (EASL), which is being held April 24 - 28, 2013 in Amsterdam, The Netherlands. Data from the three-day proof-of-concept study demonstrated that IDX719 was well-tolerated at daily doses up to 100 mg and showed potent antiviral activity across HCV genotypes 1-4, with mean maximal viral load reductions up to approximately 4.0 log10 IU/mL. These data were supported by earlier in vitro findings. Clinical plasma samples at baseline, at end of treatment and at one week post-treatment were sequenced for mutations in NS5A at known IDX719 resistance-associated locations.
Anyone have any more recent news? I can't find anything.. the recording of the latest conference call is here http://ir.idenix.com/events.cfm
April 8, 2013 Idenix Announces Data Presentation at the 48th Annual Meeting of the European Association for the Study of the Liver (EASL)
CAMBRIDGE, Mass., April 8, 2013 (GLOBE NEWSWIRE) -- Idenix Pharmaceuticals, Inc. (Nasdaq:IDIX), a biopharmaceutical company engaged in the discovery and development of drugs for the treatment of human viral diseases, today announced that it will present data on IDX719, the Company's pan-genotypic HCV NS5A inhibitor, at the 48th Annual Meeting of the European Association for the Study of the Liver (EASL) which is being held April 24 — 28, 2013 in Amsterdam, the Netherlands. Full abstracts can now be viewed at the EASL website at www.easl.eu.
McCarville, et al, "Treatment-Emergent Variants Following 3 Days of Monotherapy with IDX719, a Potent, Pan-Genotypic NS5A Inhibitor, in Subjects Infected with HCV Genotypes 1-4", will be presented in a poster session on Saturday, April 27.
About Idenix
Idenix Pharmaceuticals, Inc., headquartered in Cambridge, Massachusetts, is a biopharmaceutical company engaged in the discovery and development of drugs for the treatment of human viral diseases. Idenix's current focus is on the treatment of infections caused by hepatitis C virus. For further information about Idenix, please refer to www.idenix.com.
7:03AM Idenix Pharma announces collaboration with Janssen, unit of Johnson and Johnson (JNJ) to initiate Phase II All-oral combination studies including IDX719, Simeprevir (IDIX) 4.73 : Co announced a non-exclusive collaboration with Janssen Pharmaceuticals for the clinical development of all-oral direct-acting antiviral HCV combination therapies. The collaboration will evaluate combinations including IDX719, Idenix's once-daily pan-genotypic NS5A inhibitor, simeprevir, a once-daily protease inhibitor jointly developed by Janssen and Medivir AB, and TMC647055, a once-daily non-nucleoside polymerase inhibitor, boosted with low dose ritonavir, being developed by Janssen (JNJ). Clinical development plans include an initial drug-drug interaction study to begin in the first quarter of 2013, followed by phase II studies as agreed between the companies, and pending approval from regulatory authorities.
Idenix Pharmaceuticals, Inc. (Nasdaq:IDIX), a biopharmaceutical company engaged in the discovery and development of drugs for the treatment of human viral diseases, today announced a non-exclusive collaboration with Janssen Pharmaceuticals, Inc. for the clinical development of all-oral direct-acting antiviral (DAA) HCV combination therapies.
What happened today? This thing is up ~17% today.
IDIX has effectively thrown in the towel on IDX184: #msg-83247601.
$IDIX 11/08/12 Alert at 4.06. Manic Stock Alerts been on this long since the super short she was.
2nd alert went out 2 weeks ago looking at the 5 call @.30 which is well over 100%
ManicTrader Member Level
Thursday, November 08, 2012 10:30:16 PM
Re: ManicTrader post# 10091
Post # of 11430
$IDIX 4.06, On Watch here, looking interesting
1:15AM Idenix Pharma reports clinical data for HCV Drug Candidates - IDX719 and IDX184; demonstrated that IDX719 was well-tolerated at daily doses up to 100 mg and showed potent antiviral activity across HCV genotypes 1-4 (IDIX) 4.17 : Co announces the presentation of safety and antiviral activity results for the Company's clinical-stage hepatitis C virus drug candidates - IDX719, a next-generation pan-genotypic NS5A inhibitor, and IDX184, a nucleotide polymerase inhibitor. Idenix presented updated clinical findings from a three-day proof-of-concept study, which demonstrated that IDX719 was well-tolerated at daily doses up to 100 mg and showed potent antiviral activity across HCV genotypes 1-4, with mean maximal viral load reductions up to approximately 4.0 log10 IU/mL. The Company also presented updated cardiovascular safety and antiviral activity data from the phase IIb study of IDX184 in combination with pegylated interferon and ribavirin. There were no safety-related discontinuations or serious adverse events. IDX719 was safe and well tolerated at daily doses up to 100 mg for three days. Idenix intends to initiate a phase II clinical trial evaluating IDX719 in combination therapy in 2013. Additionally, No patient experienced virologic breakthrough during the 12-week IDX184/Peg-IFN/RBV treatment period. The majority of patients are in the ongoing PegIFN/RBV extension treatment phase, and complete sustained virologic response results will be available in 2013.
|
Followers
|
41
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
3757
|
|
Created
|
10/19/05
|
Type
|
Free
|
| Moderators | |||
http://finance.yahoo.com/q/ks?s=IDIX+Key+Statistics
Idenix Pharmaceuticals, Inc., a biopharmaceutical company, engages in the discovery and development of drugs for the treatment of human viral and other infectious diseases in the United States and Europe. The company primarily focuses its research and development on the treatment for hepatitis C virus. The company develops nucleoside/nucleotide polymerase inhibitors, including IDX184, a liver-targeted nucleotide prodrug candidate, which is in phase IIa clinical trial; and IDX102 that completed late-stage preclinical development. It also develops IDX375, a novel palm-binding polymerase inhibitor that is in phase I program; IDX320, a protease inhibitor candidate, which is in phase I healthy volunteer clinical study; and NS5A inhibitors. In addition, the company licensed its telbivudine (Tyzeka/Sebivo), a drug for the treatment of hepatitis B virus, to Novartis Pharma AG; and GSK2248761, a drug candidate from the class of compounds known as non-nucleoside reverse transcriptase inhibitors (NNRTIs), for the treatment of human immunodeficiency virus type 1 and acquired immune deficiency syndrome to GlaxoSmithKline. The company was founded in 1998 and is headquartered in Cambridge, Massachusetts.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
