Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
WTF!!!!!!!!!!!!!!!!???????????????????
Both market caps are EXACTLY $33 million.
12 month comparison:
-----------------------------------------------------------------------------------------------------
Acura Pharmaceuticals (ACUR) - OTC
Revenue: 2,245,000
Operating Income: -1,108,000
Net Income: -1,151,000
Working Capital: -135,000
Total Assets: 1,406,000
Total Capitalization: 401,000
Cash minus Debt: -235,000
Operating Cash Flow: -396,000
Market Cap - $33 million
-----------------------------------------------------------------------------------------------------
Elite Pharmaceuticals (ELTP) - OTC
Revenue: 26,057,000 ........................................... (1,161% over Acura)
Operating Income: 2,321,000 ............................... (310% over Acura)
Net Income: 5,716,000 ......................................... (597% over Acura)
Working Capital: 9,579,000 .................................. (7,196% over Acura)
Total Assets: 30,624,000 ...................................... (2,078% over Acura)
Total Capitalization: 21,975,000 ........................... (5,380% over Acura)
Cash minus Debt: 1,358,000 ............................... (678% over Acura)
Operating Cash Flow: 2,355,000 ......................... (695% over Acura)
Market Cap - $33 million
-----------------------------------------------------------------------------------------------------
Corporate officers buying up lots of shares here. Something big in the making?
ACUR $$$
News Acura Pharmaceuticals Provides an Update on the Development of LTX-03
Press Release | 01/02/2020
PALATINE, Ill., Jan. 02, 2020 (GLOBE NEWSWIRE) -- Acura Pharmaceuticals, Inc. (OTC Pink: ACUR), today provided an update on its development of LTX-03 (Hydrocodone Bitartrate and Acetaminophen) Tablets using Acura’s LIMITx™ technology intended to mitigate risks associated with overdose of the hydrocodone. In conjunction with a third party formulation developer, the formulation and manufacturing process for LTX-03 has been optimized for commercial scale. Acura’s partnered commercial manufacturer has received hydrocodone quota from the Drug Enforcement Administration and is in the process of acquiring specific auxiliary equipment for the identified manufacturing process. Acura has also completed a non-clinical small animal study to identify the benefits of a reduction in peak plasma drug concentrations (Cmax) on opioid induced respiratory depression (OIRD).
LIMITxTM Technology is designed to retard the release of active drug ingredients when too many tablets are accidentally or purposefully ingested by neutralizing stomach acid with buffer ingredients, but to also deliver efficacious amounts of drug when taken as a single tablet with a nominal buffer dose. Acura’s clinical testing to date indicates a reduction in opioid Cmax of up to 65% may be achieved in overdose situations. However, little is known about the influence of Cmax on OIRD particularly at overdose levels.
In study APT-RDR-300 all doses above 100 mg/kg demonstrated with statistical significance (p<.05) SpO2 measured OIRD at all time points post-dosing. The 100 mg/kg dose was not statistically significant for OIRD at any time point post-dosing. The mortality rate was correlated with higher doses. In all animals exhibiting OIRD, OIRD was acutely evident within 30 minutes of dosing which was consistent with the Cmax of the hydrocodone dose. Increased Cmax was generally associated with an increased prevalence of acute OIRD (SpO2 ≤70%). Approximately 50% of animals reaching this acute OIRD level resulted in death Due to a high variability in the pharmacokinetics and pharmacodynamics observed in the study, no further associations were possible. Acura believes the results of this study generally support the development of opioid products with a reduction in Cmax in overdose situations. The Company is assessing the results to determine if additional clinical or non-clinical OIRD studies should be performed.
Five groups of 11 Sprague-Dawley rats were orally administered doses of hydrocodone ranging from 100mg of drug per kg of body weight (mg/kg) up to 300 mg/kg. 8 subjects in each group were measured for respiratory depression assessing peripheral oxygen saturation (SpO2) of the blood over a 4 hour observation period. 36 subjects were analyzed as successfully completing the dosing. The additional 3 subjects in each group provided blood samples analyzed for hydrocodone at .5, 1, 2 and 4 hours post-dosing.
About Acura Pharmaceuticals
Acura Pharmaceuticals is a specialty pharmaceutical company engaged in the research, development and commercialization of technologies and product candidates intended to mitigate the risk of outcomes associated with product misuse. The Company has three proprietary technologies: LIMITx™ Technology, AVERSION® Technology and IMPEDE® Technology.
LIMITxTM Technology utilizes acid neutralizing ingredients to precisely control gastric acidity, which limits the release of drug from tablets and its subsequent systemic absorption when multiple tablets are ingested. LIMITxTM Technology is useful with products whose side effect risks can be mitigated by limiting exposure to a drug in overdose situations.
AVERSION® Technology, used in the FDA approved drug OXAYDO® (oxycodone HCl) marketed by Zyla Life Sciences, utilizes polymers designed to limit the abuse of the product by nasal snorting and injection. AVERSION® Technology is also licensed to Kempharm for use in certain of their products.
IMPEDE® Technology, used in NEXAFED® (pseudoephedrine HCl) and NEXAFED® Sinus (pseudoephedrine HCl/acetaminophen) marketed by MainPointe Pharmaceuticals, utilizes polymers and other ingredients to disrupt the extraction and processing of pseudoephedrine from the tablets into methamphetamine.
Forward-Looking Statements
Certain statements in this press release constitute “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements to be materially different from any future results, performance, or achievements expressed or implied by such forward-looking statements. Forward-looking statements may include, but are not limited to:
our ability to fund or obtain funding for our continuing operations, including the development of our products utilizing our LIMITxTM and IMPEDE® Technologies;
whether our licensees will terminate the license prior to commercialization;
the expected results of clinical studies relating to LTX-03, IMPEDE® or any successor product candidate, the date by which such studies will complete and the results will be available and whether any product candidate will ultimately receive FDA approval;
the ability of LTX-03 single tablets to achieve bioequivalence or to demonstrate efficacy in a clinical study;
whether our licensing partners will exercise their options to additional products;
whether LIMITxTM Technology will retard the release of opioid active ingredients as dose levels increase;
whether the extent to which products formulated with the LIMITxTM Technology mitigate respiratory depression risk will be determined sufficient by the FDA;
our and our licensee’s ability to successfully launch and commercialize our products and technologies;
our and our licensee’s ability to obtain necessary regulatory approvals and commercialize products utilizing our technologies;
the market acceptance of, timing of commercial launch and competitive environment for any of our products;
our ability to develop and enter into additional license agreements for our product candidates using our technologies;
the ability to avoid infringement of patents, trademarks and other proprietary rights of third parties;
the ability of our patents to protect our products from generic competition and our ability to protect and enforce our patent rights in any paragraph IV patent infringement litigation;
the adequacy of the development program for our product candidates, including whether additional clinical studies will be required to support an NDA and FDA approval of our product candidates;
changes in regulatory requirements;
adverse safety findings relating to our commercialized products or product candidates in development;
whether or when we are able to obtain FDA approval of labeling for our product candidates for the proposed indications and whether we will be able to promote the features of our technologies; and
whether our product candidates will ultimately perform as intended in commercial settings.
In some cases, you can identify forward-looking statements by terms such as "may," “will”, "should," "could," "would," "expects," "plans," "anticipates," "believes," "estimates," “indicates”, "projects," “predicts," "potential" and similar expressions intended to identify forward-looking statements. These statements reflect our current views with respect to future events and are based on assumptions and subject to risks and uncertainties. Given these uncertainties, you should not place undue reliance on these forward-looking statements. We discuss many of these risks in greater detail in our filings with the Securities and Exchange Commission.
Contact:
for Acura Investor Relations
investors@acurapharm.com
847-705-7709
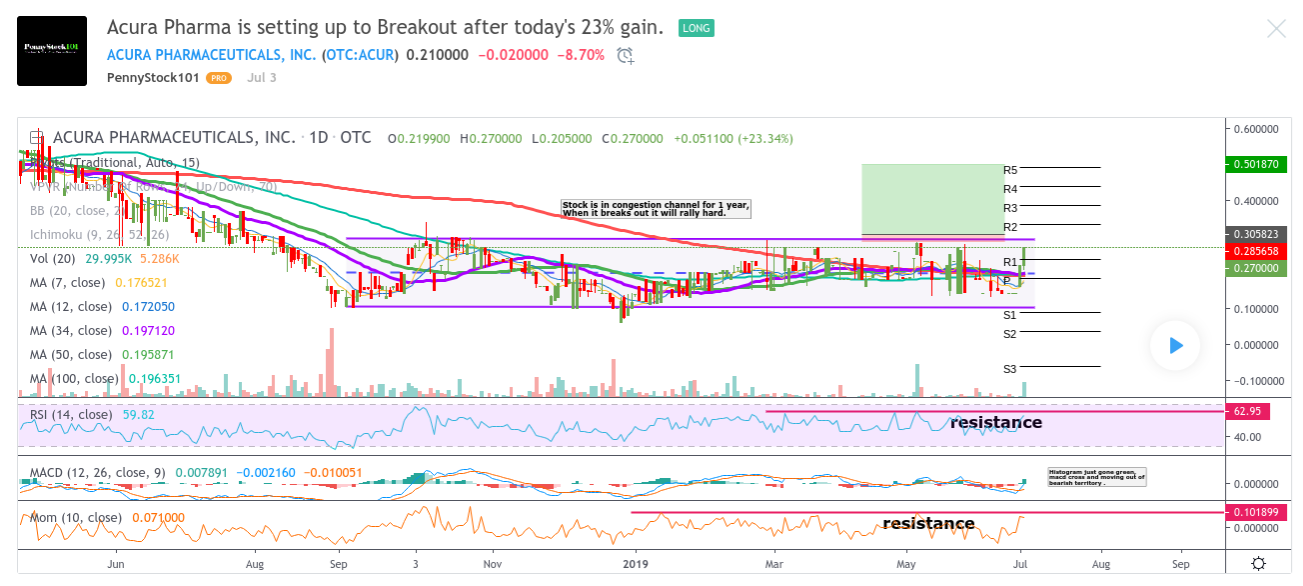
Acura Pharma is setting up to Breakout after today's 23% gain.
ACURA PHARMACEUTICALS IN. (OTC ACUR ) What a lovely trade this is setting up to be, everything is pointing to a breakout from a 1 year old channel of congestion, RSI , MON, MACD and Histogram are becoming bullish
and just like the price, entering a area of resistance. Because the stock has been so range bound for such a long time, any breakout will be amazing and fast, buys will be triggered and off it will go.
NOTEWORTHY ANNOUNCEMENT
https://www.benzinga.com/users/shanthi-r...
Acura Pharmaceuticals , Inc. (OTC: ACUR ) announced a licensing, development and commercialization agreement with Abuse Deterrent Pharma – a special purpose company representing a consortium of investors, which will finance the company's operations and completion of the development of LTX-03 immediate-release tablets using Acura's patented LIMITx technology.
The investigational drug addresses the consequences of excess oral administration of opioid tablets, the most prevalent route of opioid overdose and abuse.
The transaction is valued up to $21.3 million, not including royalties. The agreement also provides for Acura receiving royalties and potential sales-related milestones and AD Pharma retaining commercialization rights.
COMPANY PROFILE
Acura Pharmaceuticals Inc is a specialty pharmaceutical company based in the US. It is engaged in the research, development, and commercialization of technologies and products intended to address medication abuse and misuse. The company has developed Aversion and Limitx technologies are intended to address methods of abuse associated with opioid analgesics whereas Impede technology is directed at minimizing the extraction and conversion of pseudoephedrine (PSE), into methamphetamine. Its product line includes OXAYDO which is a Schedule II narcotic indicated for the management of acute and chronic moderate to severe pain; and other pipeline products.
It has been a nice rebound for those who took the risk at .10-.13 in September.
Bill passed. ACUR could make comeback company of the year.
Senate passes opioid crisis bill https://seekingalpha.com/news/3390995?source=ansh $ENDP, $MNK, $INVVY, $RBGLY, $JNJ, $INSY, $ELTP, $OPNT, $AGN, $TEVA, $ABBV, $ALKS, $AMPH, $COLL, $PTIE, $ACUR, $PTX, $ACRX, $IPCI, $KMPH, $TRVN, $CXW, $BDSI, $MYL, $PRGO, $ASRT, $NKTR
ACUR mentioned in article about abuse deterrent opioid industry.
https://seekingalpha.com/article/4206069-abuse-deterrent-opioids-business-answer-opioid-abuse-crisis
The government is cracking down on opiate abuse. Acur has the drugs to help them do that. You figure it out!
OOOOOOOOH!!! ! I think $ACUR about to BLOWUP$$$$$$$$$$$$$$$$$
FDA updates on multifaceted plan to combat opioid crisis https://seekingalpha.com/news/3386549?source=ansh $ENDP, $MNK, $INVVY, $RBGLY, $JNJ, $INSY, $ELTP, $OPNT, $AGN, $TEVA, $ABBV, $ALKS, $AMPH, $COLL, $PTIE, $ACUR, $PTX, $ACRX, $IPCI, $KMPH, $TRVN, $CXW, $BDSI, $MYL, $PRGO, $ASRT, $NKTR
Buy recommendation upgrade
https://www.fairfieldcurrent.com/2018/08/21/acura-pharmaceuticals-acur-upgraded-to-buy-at-valuengine.html
8k out. Got more money and Will continue to get money until licensing and dad.imo
ACUR locked and loaded this little bull with 1.23 target!!
ACUR has been painful.
Pun intended
Lvl 2 super thin on ask. Tic toc tic
ACUR delisted from the Nasdaq to the OTC, effective 08:41 A.M.:
http://otce.finra.org/DLAdditions
Kerplow! AMAZING pop & gonna keep running up from here!
Looking great today - Love this stock!
Nope if it can hit back to 1 by the end of day then back strong
Thats a Mythos ;) Volume Coming in right now
Bids lining Up 1,10... Maybe PM bottom
ACUR 1.2 PREMARKET. Buy dips to 1.12 - 1.2, stop 1.10. Looking for red green action , ramp to 1.50+
Clearly something is up!
almost 50% Institutionally owned= http://finviz.com/quote.ashx?t=acur
correct the article was just released today @ 4:11pm
I think I will take evening creative writing CLA$$E$ .. LOL.
Ha, I wish! It would probably make trading easier with a group of followers hinging on my every word.
Are you a SA author?...Lol.
For the record, the company filed the S-1 on 2/3/17 aka last Friday. I assume the SA author has a short position now so he wants everyone to know about it lol...SA is so terribly manipulative.
S1 was file on 2/3 not new news?
|
Followers
|
28
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
824
|
|
Created
|
08/05/06
|
Type
|
Free
|
| Moderators | |||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
