Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$EMED Preliminary revenue for the twelve months ended Dec 31 was approximately $1.15 million, which represents a total increase in sales of 26.7% on a year-over-year basis.
$EMED (OTC-QB: EMED) ("Electromedical" or the "Company"), a pioneer in the development and production of bioelectronic devices designed to relieve chronic, intractable, and acute pain through frequency and electro-modulation, is pleased to announce preliminary unaudited financial performance data for the three months and twelve months ended December 31, 2022.
Preliminary revenue for the twelve months ended Dec 31 was approximately $1.15 million, which represents a total increase in sales of 26.7% on a year-over-year basis.
During the three months ended Dec 31, preliminary revenue increased 79% on a quarterly year-over-year basis and 51% on a sequential quarterly basis.
Matthew Wolfson, Founder and CEO commented, "Q4 was a breakout quarter. We saw topline sales jump in response to further expansion in our brand awareness and sales & marketing performance. Overall, it was our best quarter since we went public and provides strong market feedback that our next-generation R&D and market positioning represents the start of an exciting new chapter for Electromedical. And the path is only going to get more exciting as we get ready to launch our revolutionary Infinity device."
As the Company moves closer to production of the new next-generation flagship device, management plans to offer a special upgrade and exchange program to existing clients, which will be bundled with a lifetime warranty.
The WellnessPro Infinity™ sets a new standard in the bioelectronics and electrotherapy marketplace featuring support for all modalities:
Designed to support*: TENS, Microcurrent, IF, EMS, PEMF, CES, VNS, PNS, COLD Laser, POD Synchronization, IDNA™, Deep Pulse™, cloud access and much more.
Unprecedented accuracy.
Easiest to use electrotherapy device on the market.
New Doctor-Patient Portal for updates and personalized treatments.
Wolfson concluded, "Last year was an ambitious year focused on key R&D objectives that we have met and now we are laying a strong foundation for handling business at a far larger scale. We accomplished everything we set out to accomplish and managed to log impressive sales growth along the way. We are also in the process of expanding consumer resources for purchasing Electromedical products. We have added FSA/HSA purchasing optionality already, and we have some more news coming that could sharply increase customer affordability without impacting our margins, and we look forward to discussing that in further detail very soon."
Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
The Company has approximately 10,000 past clients. We expect that we will be successful in encouraging a significant part of our past clients to exchange their old units for the improved Wellness Pro Infinity unit with lifetime warranty. Our development process is progressing and we look forward to providing more updates very soon. We will be in a position to start accepting advance deposits in Q2 for these units, which will greatly enhance the Company's cash position.
About Electromedical Technologies
Headquartered in Scottsdale, Arizona, Electromedical Technologies, Inc. is a commercial stage, FDA cleared, bioelectronic medical device manufacturing company initially focused on the treatment of various chronic, acute, intractable, and post-operative pain conditions. Through University collaboration agreements, the Company is working to develop a comprehensive research program in defining the effects of electro-modulation on the human body. By studying the impacts of electrical fields in cell signaling and effects on virus assembly and immune responses, the Company's goal is to reduce pain and improve overall human wellbeing. The Company's current FDA cleared product indications are for chronic acute post traumatic and post-operative, intractable pain relief.
For more information, please visit www.electromedtech.com
$EMED Electrotherapy can be used to treat a wide range of of acute and chronic pain.
It also supports healing and recovering and can be used for post-surgical discomfort and to boost your body’s natural recovery properties.
$EMED Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
$EMED March 14, 2023 /PRNewswire/ -- Electromedical Technologies, Inc. (OTC-QB: EMED) ("Electromedical" or the "Company"), a pioneer in the development and production of bioelectronic devices designed to relieve chronic, intractable, and acute pain through frequency and electro-modulation, is pleased to announce preliminary unaudited financial performance data for the three months and twelve months ended December 31, 2022.
Preliminary revenue for the twelve months ended Dec 31 was approximately $1.15 million, which represents a total increase in sales of 26.7% on a year-over-year basis.
During the three months ended Dec 31, preliminary revenue increased 79% on a quarterly year-over-year basis and 51% on a sequential quarterly basis.
Matthew Wolfson, Founder and CEO commented, "Q4 was a breakout quarter. We saw topline sales jump in response to further expansion in our brand awareness and sales & marketing performance. Overall, it was our best quarter since we went public and provides strong market feedback that our next-generation R&D and market positioning represents the start of an exciting new chapter for Electromedical. And the path is only going to get more exciting as we get ready to launch our revolutionary Infinity device."
As the Company moves closer to production of the new next-generation flagship device, management plans to offer a special upgrade and exchange program to existing clients, which will be bundled with a lifetime warranty.
The WellnessPro Infinity™ sets a new standard in the bioelectronics and electrotherapy marketplace featuring support for all modalities:
Designed to support*: TENS, Microcurrent, IF, EMS, PEMF, CES, VNS, PNS, COLD Laser, POD Synchronization, IDNA™, Deep Pulse™, cloud access and much more.
Unprecedented accuracy.
Easiest to use electrotherapy device on the market.
New Doctor-Patient Portal for updates and personalized treatments.
Wolfson concluded, "Last year was an ambitious year focused on key R&D objectives that we have met and now we are laying a strong foundation for handling business at a far larger scale. We accomplished everything we set out to accomplish and managed to log impressive sales growth along the way. We are also in the process of expanding consumer resources for purchasing Electromedical products. We have added FSA/HSA purchasing optionality already, and we have some more news coming that could sharply increase customer affordability without impacting our margins, and we look forward to discussing that in further detail very soon."
Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
The Company has approximately 10,000 past clients. We expect that we will be successful in encouraging a significant part of our past clients to exchange their old units for the improved Wellness Pro Infinity unit with lifetime warranty. Our development process is progressing and we look forward to providing more updates very soon. We will be in a position to start accepting advance deposits in Q2 for these units, which will greatly enhance the Company's cash position.
About Electromedical Technologies
Headquartered in Scottsdale, Arizona, Electromedical Technologies, Inc. is a commercial stage, FDA cleared, bioelectronic medical device manufacturing company initially focused on the treatment of various chronic, acute, intractable, and post-operative pain conditions. Through University collaboration agreements, the Company is working to develop a comprehensive research program in defining the effects of electro-modulation on the human body. By studying the impacts of electrical fields in cell signaling and effects on virus assembly and immune responses, the Company's goal is to reduce pain and improve overall human wellbeing. The Company's current FDA cleared product indications are for chronic acute post traumatic and post-operative, intractable pain relief.
For more information, please visit www.electromedtech.com
$EMED 0038 on alert! When it comes to electricity created in the human body, the energy source is chemical and has to do with the composition of the atoms and molecules present. All the elements we take into our bodies, such as oxygen, sodium, potassium, calcium, magnesium etc., have a specific electrical charge. How those molecules are bound together and how they react to other molecules near them is how chemicals create energy. Some of these electrical impulses found naturally in our bodies activate functions needed for healing.
$EMED
The WellnessPro Plus®
triggers our body’s natural processes. Electrical impulses travel from nerves around the injured area to our brain, delivering the unpleasant sensation of pain. WellnessPro Plus® operates by disrupting these pain signals received by our synapses, mimicking our natural electric signals to relieve patients from pain.
Electromedical Technologies brought together some of the leading biomedical researchers from around the world to develop next generation TENS machines. Through product innovation and a forward-thinking approach, we succeeded to develop specific algorithms in electrotherapy that work to alleviate pain.
$EMED University research centers, doctors, and physicians around the world have overseen treatments using electrotherapy with outstanding results in the reduction of pain for a wide range of acute and chronic conditions. The WellnessPro Plus is an effective complementary treatment modality for pain relief.
$EMED Electromedical Technologies, Inc. (OTC-QB: EMED) ("Electromedical" or the "Company"), a pioneer in the development and production of bioelectronic devices designed to relieve chronic, intractable, and acute pain through frequency and electro-modulation, is pleased to announce preliminary unaudited financial performance data for the three months and twelve months ended December 31, 2022.
Preliminary revenue for the twelve months ended Dec 31 was approximately $1.15 million, which represents a total increase in sales of 26.7% on a year-over-year basis.
During the three months ended Dec 31, preliminary revenue increased 79% on a quarterly year-over-year basis and 51% on a sequential quarterly basis.
Matthew Wolfson, Founder and CEO commented, "Q4 was a breakout quarter. We saw topline sales jump in response to further expansion in our brand awareness and sales & marketing performance. Overall, it was our best quarter since we went public and provides strong market feedback that our next-generation R&D and market positioning represents the start of an exciting new chapter for Electromedical. And the path is only going to get more exciting as we get ready to launch our revolutionary Infinity device."
As the Company moves closer to production of the new next-generation flagship device, management plans to offer a special upgrade and exchange program to existing clients, which will be bundled with a lifetime warranty.
The WellnessPro Infinity™ sets a new standard in the bioelectronics and electrotherapy marketplace featuring support for all modalities:
Designed to support*: TENS, Microcurrent, IF, EMS, PEMF, CES, VNS, PNS, COLD Laser, POD Synchronization, IDNA™, Deep Pulse™, cloud access and much more.
Unprecedented accuracy.
Easiest to use electrotherapy device on the market.
New Doctor-Patient Portal for updates and personalized treatments.
Wolfson concluded, "Last year was an ambitious year focused on key R&D objectives that we have met and now we are laying a strong foundation for handling business at a far larger scale. We accomplished everything we set out to accomplish and managed to log impressive sales growth along the way. We are also in the process of expanding consumer resources for purchasing Electromedical products. We have added FSA/HSA purchasing optionality already, and we have some more news coming that could sharply increase customer affordability without impacting our margins, and we look forward to discussing that in further detail very soon."
Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
The Company has approximately 10,000 past clients. We expect that we will be successful in encouraging a significant part of our past clients to exchange their old units for the improved Wellness Pro Infinity unit with lifetime warranty. Our development process is progressing and we look forward to providing more updates very soon. We will be in a position to start accepting advance deposits in Q2 for these units, which will greatly enhance the Company's cash position.
About Electromedical Technologies
Headquartered in Scottsdale, Arizona, Electromedical Technologies, Inc. is a commercial stage, FDA cleared, bioelectronic medical device manufacturing company initially focused on the treatment of various chronic, acute, intractable, and post-operative pain conditions. Through University collaboration agreements, the Company is working to develop a comprehensive research program in defining the effects of electro-modulation on the human body. By studying the impacts of electrical fields in cell signaling and effects on virus assembly and immune responses, the Company's goal is to reduce pain and improve overall human wellbeing. The Company's current FDA cleared product indications are for chronic acute post traumatic and post-operative, intractable pain relief.
For more information, please visit www.electromedtech.com
$EMED Despite the benefits of a diverse consumer drug market for pain managment, the non-medical use of pharmaceutical opioids has triggered new health threats & an escalating drug abuse epidemic.
$EMED Electrotherapy can be used to treat a wide range of of acute and chronic pain.
It also supports healing and recovering and can be used for post-surgical discomfort and to boost your body’s natural recovery properties.
$EMED Despite the benefits of a diverse consumer drug market for pain managment, the non-medical use of pharmaceutical opioids has triggered new health threats & an escalating drug abuse epidemic.
$EMED The WellnessPro Infinity™ sets a new standard in the bioelectronics and electrotherapy marketplace featuring support for all modalities:
Designed to support*: TENS, Microcurrent, IF, EMS, PEMF, CES, VNS, PNS, COLD Laser, POD Synchronization, IDNA™, Deep Pulse™, cloud access and much more.
Unprecedented accuracy.
Easiest to use electrotherapy device on the market.
New Doctor-Patient Portal for updates and personalized treatments.
Wolfson concluded, "Last year was an ambitious year focused on key R&D objectives that we have met and now we are laying a strong foundation for handling business at a far larger scale. We accomplished everything we set out to accomplish and managed to log impressive sales growth along the way. We are also in the process of expanding consumer resources for purchasing Electromedical products. We have added FSA/HSA purchasing optionality already, and we have some more news coming that could sharply increase customer affordability without impacting our margins, and we look forward to discussing that in further detail very soon."
Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
The Company has approximately 10,000 past clients. We expect that we will be successful in encouraging a significant part of our past clients to exchange their old units for the improved Wellness Pro Infinity unit with lifetime warranty. Our development process is progressing and we look forward to providing more updates very soon. We will be in a position to start accepting advance deposits in Q2 for these units, which will greatly enhance the Company's cash position.
About Electromedical Technologies
Headquartered in Scottsdale, Arizona, Electromedical Technologies, Inc. is a commercial stage, FDA cleared, bioelectronic medical device manufacturing company initially focused on the treatment of various chronic, acute, intractable, and post-operative pain conditions. Through University collaboration agreements, the Company is working to develop a comprehensive research program in defining the effects of electro-modulation on the human body. By studying the impacts of electrical fields in cell signaling and effects on virus assembly and immune responses, the Company's goal is to reduce pain and improve overall human wellbeing. The Company's current FDA cleared product indications are for chronic acute post traumatic and post-operative, intractable pain relief.
For more information, please visit www.electromedtech.com
$EMED (OTC-QB: EMED) ("Electromedical" or the "Company"), a pioneer in the development and production of bioelectronic devices designed to relieve chronic, intractable, and acute pain through frequency and electro-modulation, is pleased to announce preliminary unaudited financial performance data for the three months and twelve months ended December 31, 2022.
Preliminary revenue for the twelve months ended Dec 31 was approximately $1.15 million, which represents a total increase in sales of 26.7% on a year-over-year basis.
During the three months ended Dec 31, preliminary revenue increased 79% on a quarterly year-over-year basis and 51% on a sequential quarterly basis.
Matthew Wolfson, Founder and CEO commented, "Q4 was a breakout quarter. We saw topline sales jump in response to further expansion in our brand awareness and sales & marketing performance. Overall, it was our best quarter since we went public and provides strong market feedback that our next-generation R&D and market positioning represents the start of an exciting new chapter for Electromedical. And the path is only going to get more exciting as we get ready to launch our revolutionary Infinity device."
As the Company moves closer to production of the new next-generation flagship device, management plans to offer a special upgrade and exchange program to existing clients, which will be bundled with a lifetime warranty.
The WellnessPro Infinity™ sets a new standard in the bioelectronics and electrotherapy marketplace featuring support for all modalities:
Designed to support*: TENS, Microcurrent, IF, EMS, PEMF, CES, VNS, PNS, COLD Laser, POD Synchronization, IDNA™, Deep Pulse™, cloud access and much more.
Unprecedented accuracy.
Easiest to use electrotherapy device on the market.
New Doctor-Patient Portal for updates and personalized treatments.
Wolfson concluded, "Last year was an ambitious year focused on key R&D objectives that we have met and now we are laying a strong foundation for handling business at a far larger scale. We accomplished everything we set out to accomplish and managed to log impressive sales growth along the way. We are also in the process of expanding consumer resources for purchasing Electromedical products. We have added FSA/HSA purchasing optionality already, and we have some more news coming that could sharply increase customer affordability without impacting our margins, and we look forward to discussing that in further detail very soon."
Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
The Company has approximately 10,000 past clients. We expect that we will be successful in encouraging a significant part of our past clients to exchange their old units for the improved Wellness Pro Infinity unit with lifetime warranty. Our development process is progressing and we look forward to providing more updates very soon. We will be in a position to start accepting advance deposits in Q2 for these units, which will greatly enhance the Company's cash position.
About Electromedical Technologies
Headquartered in Scottsdale, Arizona, Electromedical Technologies, Inc. is a commercial stage, FDA cleared, bioelectronic medical device manufacturing company initially focused on the treatment of various chronic, acute, intractable, and post-operative pain conditions. Through University collaboration agreements, the Company is working to develop a comprehensive research program in defining the effects of electro-modulation on the human body. By studying the impacts of electrical fields in cell signaling and effects on virus assembly and immune responses, the Company's goal is to reduce pain and improve overall human wellbeing. The Company's current FDA cleared product indications are for chronic acute post traumatic and post-operative, intractable pain relief.
For more information, please visit www.electromedtech.com
$EMED During the three months ended Dec 31, preliminary revenue increased 79% on a quarterly year-over-year basis and 51% on a sequential quarterly basis.
$EMED University research centers, doctors, and physicians around the world have overseen treatments using electrotherapy with outstanding results in the reduction of pain for a wide range of acute and chronic conditions. The WellnessPro Plus is an effective complementary treatment modality for pain relief.
$CLNV
“Clean Vision Enters into Definitive Agreements for $20M Michigan Pyrolysis Joint Venture Project” $CLNV is pleased to bring you todays Clean-Seas Nu Way Go Press! #CleanVision #fuelcell #cleanenergy #greenEnergy #savetheplanet #CleanSeasMovement https://t.co/hvJ2X43v9o pic.twitter.com/KTIY5V1QzK
— Clean Vision Corporation (@CleanVisionCorp) April 11, 2023
$EMED The WellnessPro Infinity™ sets a new standard in the bioelectronics and electrotherapy marketplace featuring support for all modalities:
Designed to support*: TENS, Microcurrent, IF, EMS, PEMF, CES, VNS, PNS, COLD Laser, POD Synchronization, IDNA™, Deep Pulse™, cloud access and much more.
Unprecedented accuracy.
Easiest to use electrotherapy device on the market.
New Doctor-Patient Portal for updates and personalized treatments.
Wolfson concluded, "Last year was an ambitious year focused on key R&D objectives that we have met and now we are laying a strong foundation for handling business at a far larger scale. We accomplished everything we set out to accomplish and managed to log impressive sales growth along the way. We are also in the process of expanding consumer resources for purchasing Electromedical products. We have added FSA/HSA purchasing optionality already, and we have some more news coming that could sharply increase customer affordability without impacting our margins, and we look forward to discussing that in further detail very soon."
Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
The Company has approximately 10,000 past clients. We expect that we will be successful in encouraging a significant part of our past clients to exchange their old units for the improved Wellness Pro Infinity unit with lifetime warranty. Our development process is progressing and we look forward to providing more updates very soon. We will be in a position to start accepting advance deposits in Q2 for these units, which will greatly enhance the Company's cash position.
About Electromedical Technologies
Headquartered in Scottsdale, Arizona, Electromedical Technologies, Inc. is a commercial stage, FDA cleared, bioelectronic medical device manufacturing company initially focused on the treatment of various chronic, acute, intractable, and post-operative pain conditions. Through University collaboration agreements, the Company is working to develop a comprehensive research program in defining the effects of electro-modulation on the human body. By studying the impacts of electrical fields in cell signaling and effects on virus assembly and immune responses, the Company's goal is to reduce pain and improve overall human wellbeing. The Company's current FDA cleared product indications are for chronic acute post traumatic and post-operative, intractable pain relief.
For more information, please visit www.electromedtech.com
$EMED Electrotherapy can be used to treat a wide range of of acute and chronic pain.
It also supports healing and recovering and can be used for post-surgical discomfort and to boost your body’s natural recovery properties.
$EMED PRNewswire/ -- Electromedical Technologies, Inc. (OTC-QB: EMED) ("Electromedical" or the "Company"), a pioneer in the development and production of bioelectronic devices designed to relieve chronic, intractable, and acute pain through frequency and electro-modulation, is pleased to announce preliminary unaudited financial performance data for the three months and twelve months ended December 31, 2022.
Preliminary revenue for the twelve months ended Dec 31 was approximately $1.15 million, which represents a total increase in sales of 26.7% on a year-over-year basis.
During the three months ended Dec 31, preliminary revenue increased 79% on a quarterly year-over-year basis and 51% on a sequential quarterly basis.
Matthew Wolfson, Founder and CEO commented, "Q4 was a breakout quarter. We saw topline sales jump in response to further expansion in our brand awareness and sales & marketing performance. Overall, it was our best quarter since we went public and provides strong market feedback that our next-generation R&D and market positioning represents the start of an exciting new chapter for Electromedical. And the path is only going to get more exciting as we get ready to launch our revolutionary Infinity device."
As the Company moves closer to production of the new next-generation flagship device, management plans to offer a special upgrade and exchange program to existing clients, which will be bundled with a lifetime warranty.
The WellnessPro Infinity™ sets a new standard in the bioelectronics and electrotherapy marketplace featuring support for all modalities:
Designed to support*: TENS, Microcurrent, IF, EMS, PEMF, CES, VNS, PNS, COLD Laser, POD Synchronization, IDNA™, Deep Pulse™, cloud access and much more.
Unprecedented accuracy.
Easiest to use electrotherapy device on the market.
New Doctor-Patient Portal for updates and personalized treatments.
Wolfson concluded, "Last year was an ambitious year focused on key R&D objectives that we have met and now we are laying a strong foundation for handling business at a far larger scale. We accomplished everything we set out to accomplish and managed to log impressive sales growth along the way. We are also in the process of expanding consumer resources for purchasing Electromedical products. We have added FSA/HSA purchasing optionality already, and we have some more news coming that could sharply increase customer affordability without impacting our margins, and we look forward to discussing that in further detail very soon."
Management notes that 2023 will see the launch of the WellnessPro Infinity™, continued expansion in its sales team, a strong focus on value-add strategic partnerships, research partnerships and further customer affordability strategies as the Company monetizes its market-leading next-generation drug-free pain therapy technology.
The Company has approximately 10,000 past clients. We expect that we will be successful in encouraging a significant part of our past clients to exchange their old units for the improved Wellness Pro Infinity unit with lifetime warranty. Our development process is progressing and we look forward to providing more updates very soon. We will be in a position to start accepting advance deposits in Q2 for these units, which will greatly enhance the Company's cash position.
About Electromedical Technologies
Headquartered in Scottsdale, Arizona, Electromedical Technologies, Inc. is a commercial stage, FDA cleared, bioelectronic medical device manufacturing company initially focused on the treatment of various chronic, acute, intractable, and post-operative pain conditions. Through University collaboration agreements, the Company is working to develop a comprehensive research program in defining the effects of electro-modulation on the human body. By studying the impacts of electrical fields in cell signaling and effects on virus assembly and immune responses, the Company's goal is to reduce pain and improve overall human wellbeing. The Company's current FDA cleared product indications are for chronic acute post traumatic and post-operative, intractable pain relief.
For more information, please visit www.electromedtech.com
$EMED Electrotherapy can be used to treat a wide range of of acute and chronic pain.
It also supports healing and recovering and can be used for post-surgical discomfort and to boost your body’s natural recovery properties.
$EMED Electromedical’s commitment to create long-lasting drug-free solutions to pain management is honored by the development of The WellnessPro Plus®, a technologically advanced TENS (transcutaneous electrical nerve stimulation) therapy device focusing on pain relief.
$EMED's preliminary revenue for the twelve months ended Dec 31 was approximately $1.15 million, which represents a total increase in sales of 26.7% on a year-over-year basis.
$EMED University research centers, doctors, and physicians around the world have overseen treatments using electrotherapy with outstanding results in the reduction of pain for a wide range of acute and chronic conditions. The WellnessPro Plus is an effective complementary treatment modality for pain relief.
$CURR Nicole Kidman Endorsed Wellness and Beauty Products; On-Track for Accelerated Sales Growth with Company Rebranded as Avenir Wellness Solutions, Inc : CURR https://apnews.com/press-release/ein-presswire-newsmatics/medical-devices-ein-presswire-newsmatics-amazoncom-inc-nicole-kidman-business-029cfbbec3e20399b4ebf17a7275e454
$EMED Electrotherapy can be used to treat a wide range of of acute and chronic pain.
It also supports healing and recovering and can be used for post-surgical discomfort and to boost your body’s natural recovery properties.
$EMED In 2018, 67,367 drug overdose deaths occurred in the United States, 70% of them involved opioids.
https://electromedtech.com/investors-relations/
$TLIF Development Review Committee hearing happened yesterday! >>
$TLIF Development Review Committee hearing in Apopka happening right now for Be Climbing gym. #progress pic.twitter.com/JPUs7AXFSz
— Tocca Life (@Tocca_Life) March 15, 2023
$CDSG EXTENDS LARGE SOUTH LITHIUM ZONE 2.5 KMS WESTWARD WITH HIGH GRADE RESULTS INCLUDING 4.82% LI2O ON TITAN 1
Las Vegas, NV., March 13, 2023 (GLOBE NEWSWIRE) -- Titan Lithium Inc., (OTC Markets: CDSG) (the "Company" or "CDSG") is pleased to announce strong Lithium geochemical sampling results from the continuation of the surface sampling program at the company's huge Titan 1 lithium property in Tanzania.
As part of our follow up program designed to outline the boundaries of the expansive Titan 1 Lithium anomaly, extensions of the original survey lines were made to the north and south and new roughly parallel soil geochemical survey lines were added, such that the surveyed area now covers a 10-mile x 3.3-mile (16.3 x 5.3 km) area.
Samples were all removed from the B soil horizon, from a depth of 8 to 11 inches with the line orientation running approximately North-South.
The majority of results from the western-most line have returned. The addition of this line is beginning to show that there appear to be three distinct high-grade areas emerging from the survey, a north, mid and south 'zone'.
This west line encountered nul to high grade values, with the high-grade values lining up with the locations of the three separate high-grade zones. Several new samples from the line returned values which averaged 2.64% Li2O (12,266 ppm Li) over 1.25 miles (2kms). The highest grade encountered on the western line area returned 4.82% Li2O (22,400 ppm Li).
The company has previously described the north central zone which remains open to the north, however, with the extension of the Southern zone it has become the largest, measuring slightly over 5 x 2.5 miles in extent, containing Lithium values that average 1.76% Li2O (8,177 ppm Li). This zone currently remains open to the east and west.
Craig Alford, MSc, PGeo, and CEO of the Company, a Qualified Person under NI 43-101, noted "We are excited to see the sample results as they come in. Each time we think we've closed off one of the three high-grade Lithium zones, it remains open and further sample lines are being planned."
Rigorous Quality control of the samples was maintained and all samples were prepared and analyzed at the independent and ISO 9001 certified African Minerals and Geosciences Centre (AMGC) in Dar es Salaam.
About the Company: China Dongsheng International Inc. (OTC Markets: CDSG), through its wholly owned subsidiary Titan Lithium, Inc., is a lithium explorer and developer with operations in Nevada, USA and The United Republic of Tanzania.
Contact:
Harp Sangha-Chairman
harp@titanlithiuminc.com
+1-702-595-2247
http://titanlithiuminc.com
Forward-Looking Statements:
This press release may contain "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995, such as statements relating to financial results and plans for future development activities and are thus prospective. Forward-looking statements include all statements that are not statements of historical fact regarding intent, belief or current expectations of the Company, its directors or its officers. Investors are cautioned that any such forward-looking statements are not guarantees of future performance and involve risks and uncertainties, many of which are beyond the Company's ability to control. Actual results may differ materially from those projected in the forward-looking statements. Among the factors that could cause actual results to differ materially from those indicated in the forward-looking statements are risks and uncertainties associated with the Company's business and finances in general, including the ability to continue and manage its growth, competition, global economic conditions and other factors discussed in detail in the Company's periodic filings with the Securities and Exchange Commission.
$TLIF Tweet Out regarding Shell Status Removal! >>
$TLIF issued a Supplemental this evening following the procedures as required by OTCM regarding shell status changes. Our request is to remove shell status. OTCM must now review and process the request for change of shell status.https://t.co/xLmLoAw510
— Tocca Life (@Tocca_Life) March 7, 2023
$ACGX's Quarterly Revenues Increased $300,445 compared to 2021 for a 11.1% increase!
$PVSP News and Headlines https://www.barchart.com/stocks/quotes/PVSP/news
$PVSP SEATTLE, Jan. 17, 2023 (GLOBE NEWSWIRE) -- Pervasip Corp. (OTCPK: PVSP) (“Pervasip” and the “Company”) today announced that it will spin-off 100% of its wholly-owned subsidiary, Artizen Corporation (“Artizen”), as a separate public company, with an anticipated record date between July 1, 2023, and September 30, 2023. As a result of the spin-off, all Pervasip shareholders of record as of the designated record date shall receive shares in the newly public Artizen in proportion to their ownership in Pervasip. Artizen conducts 100% of Pervasip’s cannabis business segment operations through its Zen Asset Management LLC subsidiary ("Zen”).
https://www.nasdaq.com/press-release/pervasip-announces-artizen-spin-off-2023-01-17
$GBHL
$AITX Their Subsidiary Robotic Assistance Devices Healthcare Market Expansion with RAD Light My Way https://finance.yahoo.com/news/aitxs-subsidiary-robotic-assistance-devices-134000709.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$PVSP's identification of a series of exciting expansion opportunities, including potential acquisitions involving licensed wholesale and retail operations that would offer Artizen the potential to establish itself as a vertically integrated multi-state operator (“MSO”).
$PVSP (OTCPK: PVSP) (“Pervasip” and the “Company”) today announced that it will spin-off 100% of its wholly-owned subsidiary, Artizen Corporation (“Artizen”), as a separate public company, with an anticipated record date between July 1, 2023, and September 30, 2023. As a result of the spin-off, all Pervasip shareholders of record as of the designated record date shall receive shares in the newly public Artizen in proportion to their ownership in Pervasip. Artizen conducts 100% of Pervasip’s cannabis business segment operations through its Zen Asset Management LLC subsidiary ("Zen”).
Positioning for Expansion
“Spinning out Artizen will allow us to properly capitalize the business to take advantage of multiple expansion opportunities to build on our existing foundation,” said German Burtscher, Chairman and Chief Executive Officer of Pervasip and Artizen.
Pervasip previously announced its identification of a series of exciting expansion opportunities, including potential acquisitions involving licensed wholesale and retail operations that would offer Artizen the potential to establish itself as a vertically integrated multi-state operator (“MSO”).
While the local Washington market presents compelling opportunities for the acquisition and roll-up of wholesale flower, concentrate, and other related production assets by Zen’s independent cultivators, the regulatory environment in Washington prevents vertical integration into retail assets. That limitation is a significant constraint on growth since Zen’s independent cultivators cannot acquire dispensary assets to internalize retail revenues that are typically about three times wholesale receipts. Stated differently, Artizen could be generating about three times its current revenues, or about $46 to $52 million, if Zen’s existing footprint was in another state. Thus, diversification has been and remains a key strategic focus – both within Washington by expanding Zen’s offerings and exploiting compliant acquisition opportunities, and in valuable emerging cannabis markets by replicating Artizen’s proven formula for success in new U.S. and Canadian geographies.
Burtscher added, “Timing is important. The valuations and fates of MSOs and other participants in the cannabis industry have vacillated in several waves since state legalization commenced. We believe that increased legalization is inevitable in time, both at the federal level and in the form of improved regulatory environments in Washington. Preparing ourselves for that day is an important aspect of our long-term plans, including by expanding our brands and building on our footprint in valuable new geographies, and accessing long term equity capital to do so with shareholder friendly structures. Critically, the Artizen capital structure after the spin-out is completed has been specifically designed to allow us to raise equity financing on terms that vastly exceed anything that we can accomplish with Pervasip’s current capital structure.”
Improved Capital Structure
Pervasip currently has about 5,000,000,000 shares of common stock issued and outstanding, corresponding to about 15% of Pervasip’s fully diluted issued and outstanding common stock. Pervasip additionally has 850,000 shares of Series K convertible preferred stock issued and outstanding, corresponding to 85% of Pervasip’s fully diluted issued and outstanding common stock, as well as various other convertible securities.
Holders of Pervasip’s issued and outstanding common shares as of the record date will receive an aggregate of 15,000,000 shares of Artizen common stock, corresponding to 15% of Artizen’s fully diluted issued and outstanding common shares upon completion of the transaction (in addition to retaining their shares in Pervasip). Likewise, holders of Pervasip’s Series K and other convertible securities will receive an aggregate of 85,000,000 shares of Artizen common stock, the vast majority of which will be subject to lock-up restrictions prohibiting sales prior to Artizen’s realization of material growth and other performance objectives. Artizen will have 100,000,000 common shares outstanding on a fully diluted basis upon completion of the transaction, with no convertible debt or other securities.
The Company is working with its auditors to complete the required financial audits, including one for Pervasip on a consolidated basis and another for Artizen on a consolidated basis (without Pervasip). Once complete, a Form 10 Registration Statement will be filed with the SEC for Artizen to initiate the spin-off process. Additional information regarding the status and timing of the transaction and the various required regulatory and other approvals will be provided as it becomes available. Pervasip has commenced evaluation of potential targets for acquisition upon completion of the Artizen spin-off.
Pervasip Corporation
Pervasip Corp., a developer of companies and technologies in high value emerging markets, owns Artizen Corporation and its subsidiary, Zen Asset Management LLC, a diversified asset management company founded to acquire, develop, and support companies and technologies in the cannabis industry. ZAM’s existing clients operate four licensed cannabis cultivation and one processing facility in Washington. Most of the biomass produced by these independent cultivators has been sold historically under the Artizen™ brand, including all-time top selling products in flower in Washington state. Additional information on Artizen-branded products is available online at www.artizencannabis.com. Pervasip additionally owns 5% of KRTL Biotech, Inc., a developer of biotechnologies with a focus on pharmaceutical applications of cannabinol and psilocybin. Additional information on KRTL is available online at www.krtlbiotech.com.
$PVSP Company Announces Artizen Spin Off https://finance.yahoo.com/news/pervasip-announces-artizen-spin-off-141500515.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
budfoxrun Thanks - Good Evening My Friends - it would be very interesting to hear
Your opinions about a few gold producers I am invested in;
Monument Mining - they also have added a sulphide mill and welcome -
here is a -
Gold Mines Producer undervalued and IMO will do very well in
the short & long time future -
Monument in International news…Production late January

https://im-mining.com/2022/10/13/monument-minings-selinsing-flotation-plant-almost-complete-ready-for-pre-commissioning/
Corporate Presentation December 2022
https://monumentmining.com/site/assets/files/4320/mmy-corporate-presentation-december-2022.pdf
Ex.gratia - Risk Averse Approach on How to become Wealthy Very Quickly
Very simply, buy MMY about 10 cents.
Risk averse because...
1.....currently trading below cash and liquid investments
2...owns highly valued strategically located Australian Gold deposits proxy valued on its 20%
ownership of ODY ( Odyssey Gold in ASX ...market cap $29 m ) at over $60 million
3....strong growth profile from resumption of gold production of 35000+ ounces from its
Selinsing new Floatation plant......( Fair value estimated at $0.43 per share by recent
analyst report
4...Recurring revenue from 1.25% Royalty from Mengapher poly metallic mines
5...Substantial position has been accumulating in the open market since 2019
6.Insider ownership in excess of 30% so management fully aligned with ordinary
shareholders
7..Hard book value of $0.48 per share
8...Very low cash costs due to location and very low G&A operating costs
9...Substantial exploration upside already tangibly demonstrated from its exploration
tenements surrounding its Selinsing mine
10...Showing all signs that it will be taken private soon , with fair value multiples of current
share price.
$20,000 invested now by buying over 200,000 shares below $0.10 should get you at least
$50,000 by exit 2023 on either its growing low cost gold production fundamentals and/or it
being taken over or maybe go private at $0.25 or above?
•••
Comment by romara on Feb 13, 2023 1:08pm
This the main reason we are here.....It's all in para 7 7..Hard book value of $0.48 per share ..... Richard
Company Profile & Information (TSXV:MMY)
Contact Information
Address
1100 Melville Street, Suite 1580, Vancouver, BC, CA, V6E 4A6
Telephone
+1 604 638-1661
Website
http://www.monumentmining.com
Fax
+1 604 638-1663
Email
info@monumentmining.com
Details
CEO
Cathy Zhai
----
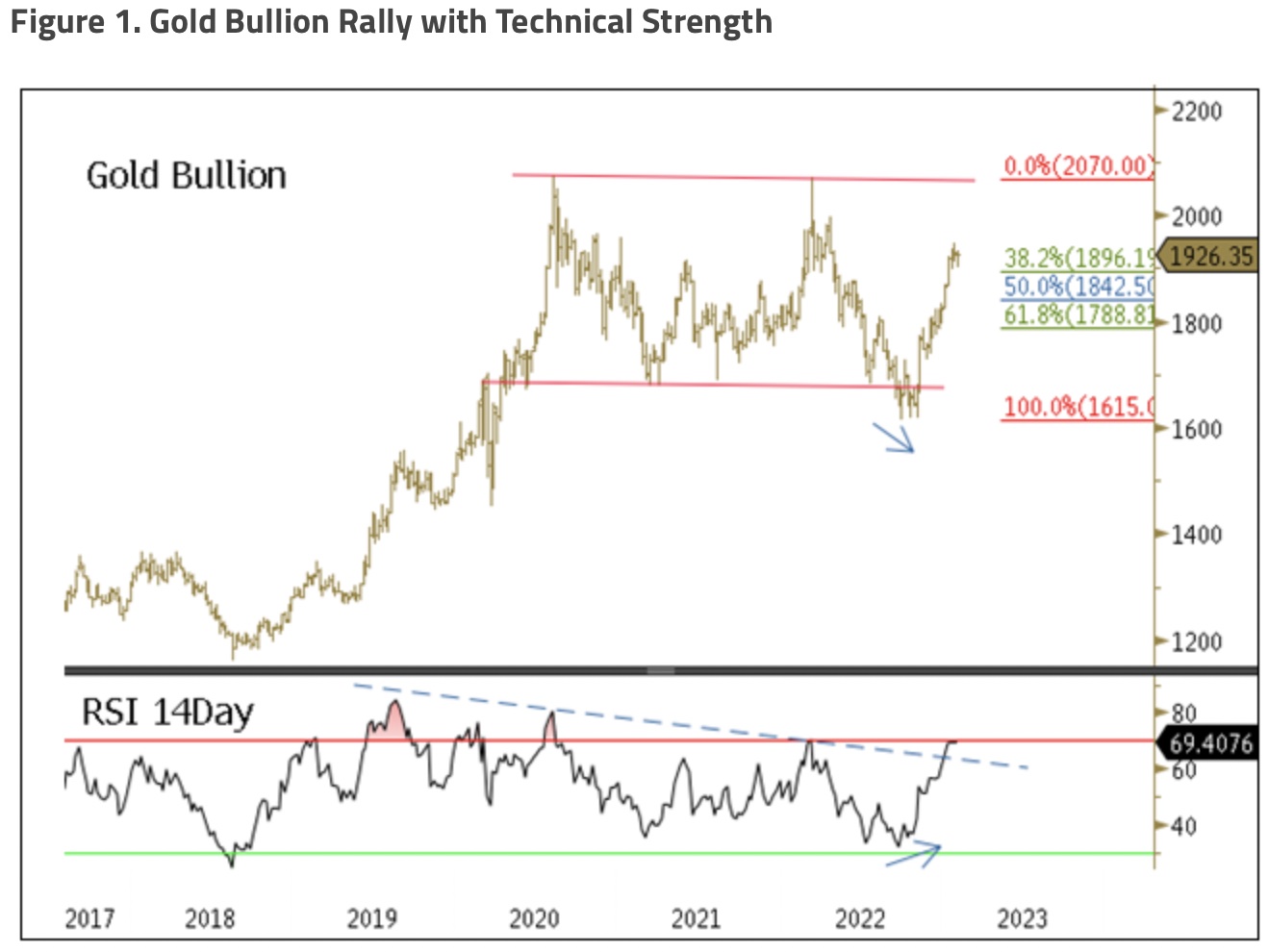
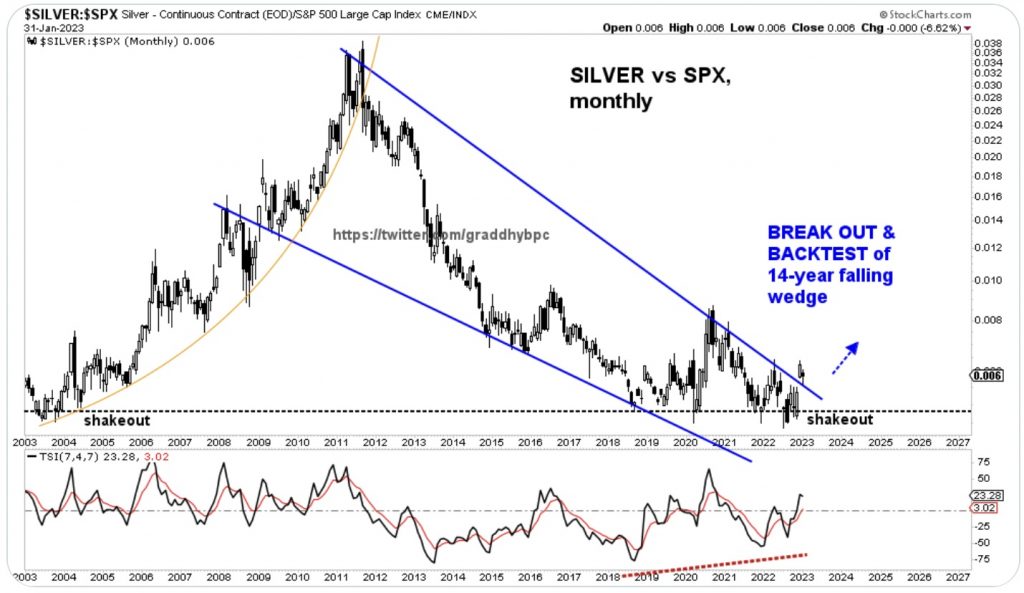
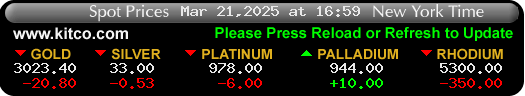
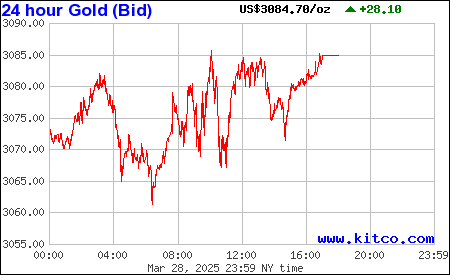
$Gold Forecast: Temporary Correction Followed by a Breakout Above $2000 -
$Aris Mining Red Cloud On-Site Interview at VRIC
Here is the link to Tyron Bretonbach providing an interview to Red Cloud at VRIC 2023
Mining Conference.





$PVSP PERVASIP is in the process of looking for other opportunities to create value.
$PVSP “Spinning out Artizen will allow us to properly capitalize the business to take advantage of multiple expansion opportunities to build on our existing foundation,” said German Burtscher, Chairman and Chief Executive Officer of Pervasip and Artizen.
Pervasip previously announced its identification of a series of exciting expansion opportunities, including potential acquisitions involving licensed wholesale and retail operations that would offer Artizen the potential to establish itself as a vertically integrated multi-state operator (“MSO”).
While the local Washington market presents compelling opportunities for the acquisition and roll-up of wholesale flower, concentrate, and other related production assets by Zen’s independent cultivators, the regulatory environment in Washington prevents vertical integration into retail assets. That limitation is a significant constraint on growth since Zen’s independent cultivators cannot acquire dispensary assets to internalize retail revenues that are typically about three times wholesale receipts. Stated differently, Artizen could be generating about three times its current revenues, or about $46 to $52 million, if Zen’s existing footprint was in another state. Thus, diversification has been and remains a key strategic focus – both within Washington by expanding Zen’s offerings and exploiting compliant acquisition opportunities, and in valuable emerging cannabis markets by replicating Artizen’s proven formula for success in new U.S. and Canadian geographies.
Burtscher added, “Timing is important. The valuations and fates of MSOs and other participants in the cannabis industry have vacillated in several waves since state legalization commenced. We believe that increased legalization is inevitable in time, both at the federal level and in the form of improved regulatory environments in Washington. Preparing ourselves for that day is an important aspect of our long-term plans, including by expanding our brands and building on our footprint in valuable new geographies, and accessing long term equity capital to do so with shareholder friendly structures. Critically, the Artizen capital structure after the spin-out is completed has been specifically designed to allow us to raise equity financing on terms that vastly exceed anything that we can accomplish with Pervasip’s current capital structure.”
Improved Capital Structure
Pervasip currently has about 5,000,000,000 shares of common stock issued and outstanding, corresponding to about 15% of Pervasip’s fully diluted issued and outstanding common stock. Pervasip additionally has 850,000 shares of Series K convertible preferred stock issued and outstanding, corresponding to 85% of Pervasip’s fully diluted issued and outstanding common stock, as well as various other convertible securities.
Holders of Pervasip’s issued and outstanding common shares as of the record date will receive an aggregate of 15,000,000 shares of Artizen common stock, corresponding to 15% of Artizen’s fully diluted issued and outstanding common shares upon completion of the transaction (in addition to retaining their shares in Pervasip). Likewise, holders of Pervasip’s Series K and other convertible securities will receive an aggregate of 85,000,000 shares of Artizen common stock, the vast majority of which will be subject to lock-up restrictions prohibiting sales prior to Artizen’s realization of material growth and other performance objectives. Artizen will have 100,000,000 common shares outstanding on a fully diluted basis upon completion of the transaction, with no convertible debt or other securities.
The Company is working with its auditors to complete the required financial audits, including one for Pervasip on a consolidated basis and another for Artizen on a consolidated basis (without Pervasip). Once complete, a Form 10 Registration Statement will be filed with the SEC for Artizen to initiate the spin-off process. Additional information regarding the status and timing of the transaction and the various required regulatory and other approvals will be provided as it becomes available. Pervasip has commenced evaluation of potential targets for acquisition upon completion of the Artizen spin-off.
Pervasip Corporation
Pervasip Corp., a developer of companies and technologies in high value emerging markets, owns Artizen Corporation and its subsidiary, Zen Asset Management LLC, a diversified asset management company founded to acquire, develop, and support companies and technologies in the cannabis industry. ZAM’s existing clients operate four licensed cannabis cultivation and one processing facility in Washington. Most of the biomass produced by these independent cultivators has been sold historically under the Artizen™ brand, including all-time top selling products in flower in Washington state. Additional information on Artizen-branded products is available online at www.artizencannabis.com. Pervasip additionally owns 5% of KRTL Biotech, Inc., a developer of biotechnologies with a focus on pharmaceutical applications of cannabinol and psilocybin. Additional information on KRTL is available online at www.krtlbiotech.com.
$GBHL
$PVSP SEATTLE, Jan. 17, 2023 (GLOBE NEWSWIRE) -- Pervasip Corp. (OTCPK: PVSP) (“Pervasip” and the “Company”) today announced that it will spin-off 100% of its wholly-owned subsidiary, Artizen Corporation (“Artizen”), as a separate public company, with an anticipated record date between July 1, 2023, and September 30, 2023. As a result of the spin-off, all Pervasip shareholders of record as of the designated record date shall receive shares in the newly public Artizen in proportion to their ownership in Pervasip. Artizen conducts 100% of Pervasip’s cannabis business segment operations through its Zen Asset Management LLC subsidiary ("Zen”).
Positioning for Expansion
“Spinning out Artizen will allow us to properly capitalize the business to take advantage of multiple expansion opportunities to build on our existing foundation,” said German Burtscher, Chairman and Chief Executive Officer of Pervasip and Artizen.
Pervasip previously announced its identification of a series of exciting expansion opportunities, including potential acquisitions involving licensed wholesale and retail operations that would offer Artizen the potential to establish itself as a vertically integrated multi-state operator (“MSO”).
While the local Washington market presents compelling opportunities for the acquisition and roll-up of wholesale flower, concentrate, and other related production assets by Zen’s independent cultivators, the regulatory environment in Washington prevents vertical integration into retail assets. That limitation is a significant constraint on growth since Zen’s independent cultivators cannot acquire dispensary assets to internalize retail revenues that are typically about three times wholesale receipts. Stated differently, Artizen could be generating about three times its current revenues, or about $46 to $52 million, if Zen’s existing footprint was in another state. Thus, diversification has been and remains a key strategic focus – both within Washington by expanding Zen’s offerings and exploiting compliant acquisition opportunities, and in valuable emerging cannabis markets by replicating Artizen’s proven formula for success in new U.S. and Canadian geographies.
Burtscher added, “Timing is important. The valuations and fates of MSOs and other participants in the cannabis industry have vacillated in several waves since state legalization commenced. We believe that increased legalization is inevitable in time, both at the federal level and in the form of improved regulatory environments in Washington. Preparing ourselves for that day is an important aspect of our long-term plans, including by expanding our brands and building on our footprint in valuable new geographies, and accessing long term equity capital to do so with shareholder friendly structures. Critically, the Artizen capital structure after the spin-out is completed has been specifically designed to allow us to raise equity financing on terms that vastly exceed anything that we can accomplish with Pervasip’s current capital structure.”
Improved Capital Structure
Pervasip currently has about 5,000,000,000 shares of common stock issued and outstanding, corresponding to about 15% of Pervasip’s fully diluted issued and outstanding common stock. Pervasip additionally has 850,000 shares of Series K convertible preferred stock issued and outstanding, corresponding to 85% of Pervasip’s fully diluted issued and outstanding common stock, as well as various other convertible securities.
Holders of Pervasip’s issued and outstanding common shares as of the record date will receive an aggregate of 15,000,000 shares of Artizen common stock, corresponding to 15% of Artizen’s fully diluted issued and outstanding common shares upon completion of the transaction (in addition to retaining their shares in Pervasip). Likewise, holders of Pervasip’s Series K and other convertible securities will receive an aggregate of 85,000,000 shares of Artizen common stock, the vast majority of which will be subject to lock-up restrictions prohibiting sales prior to Artizen’s realization of material growth and other performance objectives. Artizen will have 100,000,000 common shares outstanding on a fully diluted basis upon completion of the transaction, with no convertible debt or other securities.
The Company is working with its auditors to complete the required financial audits, including one for Pervasip on a consolidated basis and another for Artizen on a consolidated basis (without Pervasip). Once complete, a Form 10 Registration Statement will be filed with the SEC for Artizen to initiate the spin-off process. Additional information regarding the status and timing of the transaction and the various required regulatory and other approvals will be provided as it becomes available. Pervasip has commenced evaluation of potential targets for acquisition upon completion of the Artizen spin-off.
Pervasip Corporation
Pervasip Corp., a developer of companies and technologies in high value emerging markets, owns Artizen Corporation and its subsidiary, Zen Asset Management LLC, a diversified asset management company founded to acquire, develop, and support companies and technologies in the cannabis industry. ZAM’s existing clients operate four licensed cannabis cultivation and one processing facility in Washington. Most of the biomass produced by these independent cultivators has been sold historically under the Artizen™ brand, including all-time top selling products in flower in Washington state. Additional information on Artizen-branded products is available online at www.artizencannabis.com. Pervasip additionally owns 5% of KRTL Biotech, Inc., a developer of biotechnologies with a focus on pharmaceutical applications of cannabinol and psilocybin. Additional information on KRTL is available online at www.krtlbiotech.com.
$GBHL
$PVSP Keep this one on your radar.
$GBHL
$PVSP Pervasip common shareholders will receive 15% or 15,000,000 shares in Artizen Corp at the Record Date
The Record Date is the day end of day before the date at which the new Artizen Ticker goes life
Pervasip are not shortchanged. On an as if fully diluted basis, all Pervasip common shares amount to approximately 13.8% of all outstanding shares; we set 15% aside for all common shareholders in Artizen Corp
$PVSP SEATTLE, Jan. 17, 2023 (GLOBE NEWSWIRE) -- Pervasip Corp. (OTCPK: PVSP) (“Pervasip” and the “Company”) today announced that it will spin-off 100% of its wholly-owned subsidiary, Artizen Corporation (“Artizen”), as a separate public company, with an anticipated record date between July 1, 2023, and September 30, 2023. As a result of the spin-off, all Pervasip shareholders of record as of the designated record date shall receive shares in the newly public Artizen in proportion to their ownership in Pervasip. Artizen conducts 100% of Pervasip’s cannabis business segment operations through its Zen Asset Management LLC subsidiary ("Zen”).
https://www.nasdaq.com/press-release/pervasip-announces-artizen-spin-off-2023-01-17
$GBHL
$PVSP Overview https://www.otcmarkets.com/stock/PVSP/overview
$PVSP The company, Pervasip, Announces Artizen Spin Off https://finance.yahoo.com/news/pervasip-announces-artizen-spin-off-141500515.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance $GBHL
$PVSP the company has about 5,000,000,000 shares of common stock issued and outstanding, corresponding to about 15% of Pervasip’s fully diluted issued and outstanding common stock. Pervasip additionally has 850,000 shares of Series K convertible preferred stock issued and outstanding, corresponding to 85% of Pervasip’s fully diluted issued and outstanding common stock, as well as various other convertible securities.
Holders of Pervasip’s issued and outstanding common shares as of the record date will receive an aggregate of 15,000,000 shares of Artizen common stock, corresponding to 15% of Artizen’s fully diluted issued and outstanding common shares upon completion of the transaction (in addition to retaining their shares in Pervasip). Likewise, holders of Pervasip’s Series K and other convertible securities will receive an aggregate of 85,000,000 shares of Artizen common stock, the vast majority of which will be subject to lock-up restrictions prohibiting sales prior to Artizen’s realization of material growth and other performance objectives. Artizen will have 100,000,000 common shares outstanding on a fully diluted basis upon completion of the transaction, with no convertible debt or other securities.
The Company is working with its auditors to complete the required financial audits, including one for Pervasip on a consolidated basis and another for Artizen on a consolidated basis (without Pervasip). Once complete, a Form 10 Registration Statement will be filed with the SEC for Artizen to initiate the spin-off process. Additional information regarding the status and timing of the transaction and the various required regulatory and other approvals will be provided as it becomes available. Pervasip has commenced evaluation of potential targets for acquisition upon completion of the Artizen spin-off.
Pervasip Corporation
Pervasip Corp., a developer of companies and technologies in high value emerging markets, owns Artizen Corporation and its subsidiary, Zen Asset Management LLC, a diversified asset management company founded to acquire, develop, and support companies and technologies in the cannabis industry. ZAM’s existing clients operate four licensed cannabis cultivation and one processing facility in Washington. Most of the biomass produced by these independent cultivators has been sold historically under the Artizen™ brand, including all-time top selling products in flower in Washington state. Additional information on Artizen-branded products is available online at www.artizencannabis.com. Pervasip additionally owns 5% of KRTL Biotech, Inc., a developer of biotechnologies with a focus on pharmaceutical applications of cannabinol and psilocybin. Additional information on KRTL is available online at www.krtlbiotech.com. $GBHL
$PVSP today announced that it will spin-off 100% of its wholly-owned subsidiary, Artizen Corporation (“Artizen”), as a separate public company, with an anticipated record date between July 1, 2023, and September 30, 2023. As a result of the spin-off, all Pervasip shareholders of record as of the designated record date shall receive shares in the newly public Artizen in proportion to their ownership in Pervasip. Artizen conducts 100% of Pervasip’s cannabis business segment operations through its Zen Asset Management LLC subsidiary ("Zen”).
Positioning for Expansion
“Spinning out Artizen will allow us to properly capitalize the business to take advantage of multiple expansion opportunities to build on our existing foundation,” said German Burtscher, Chairman and Chief Executive Officer of Pervasip and Artizen.
Pervasip previously announced its identification of a series of exciting expansion opportunities, including potential acquisitions involving licensed wholesale and retail operations that would offer Artizen the potential to establish itself as a vertically integrated multi-state operator (“MSO”).
While the local Washington market presents compelling opportunities for the acquisition and roll-up of wholesale flower, concentrate, and other related production assets by Zen’s independent cultivators, the regulatory environment in Washington prevents vertical integration into retail assets. That limitation is a significant constraint on growth since Zen’s independent cultivators cannot acquire dispensary assets to internalize retail revenues that are typically about three times wholesale receipts. Stated differently, Artizen could be generating about three times its current revenues, or about $46 to $52 million, if Zen’s existing footprint was in another state. Thus, diversification has been and remains a key strategic focus – both within Washington by expanding Zen’s offerings and exploiting compliant acquisition opportunities, and in valuable emerging cannabis markets by replicating Artizen’s proven formula for success in new U.S. and Canadian geographies.
Burtscher added, “Timing is important. The valuations and fates of MSOs and other participants in the cannabis industry have vacillated in several waves since state legalization commenced. We believe that increased legalization is inevitable in time, both at the federal level and in the form of improved regulatory environments in Washington. Preparing ourselves for that day is an important aspect of our long-term plans, including by expanding our brands and building on our footprint in valuable new geographies, and accessing long term equity capital to do so with shareholder friendly structures. Critically, the Artizen capital structure after the spin-out is completed has been specifically designed to allow us to raise equity financing on terms that vastly exceed anything that we can accomplish with Pervasip’s current capital structure.”
Improved Capital Structure
Pervasip currently has about 5,000,000,000 shares of common stock issued and outstanding, corresponding to about 15% of Pervasip’s fully diluted issued and outstanding common stock. Pervasip additionally has 850,000 shares of Series K convertible preferred stock issued and outstanding, corresponding to 85% of Pervasip’s fully diluted issued and outstanding common stock, as well as various other convertible securities.
Holders of Pervasip’s issued and outstanding common shares as of the record date will receive an aggregate of 15,000,000 shares of Artizen common stock, corresponding to 15% of Artizen’s fully diluted issued and outstanding common shares upon completion of the transaction (in addition to retaining their shares in Pervasip). Likewise, holders of Pervasip’s Series K and other convertible securities will receive an aggregate of 85,000,000 shares of Artizen common stock, the vast majority of which will be subject to lock-up restrictions prohibiting sales prior to Artizen’s realization of material growth and other performance objectives. Artizen will have 100,000,000 common shares outstanding on a fully diluted basis upon completion of the transaction, with no convertible debt or other securities.
The Company is working with its auditors to complete the required financial audits, including one for Pervasip on a consolidated basis and another for Artizen on a consolidated basis (without Pervasip). Once complete, a Form 10 Registration Statement will be filed with the SEC for Artizen to initiate the spin-off process. Additional information regarding the status and timing of the transaction and the various required regulatory and other approvals will be provided as it becomes available. Pervasip has commenced evaluation of potential targets for acquisition upon completion of the Artizen spin-off.
Pervasip Corporation
Pervasip Corp., a developer of companies and technologies in high value emerging markets, owns Artizen Corporation and its subsidiary, Zen Asset Management LLC, a diversified asset management company founded to acquire, develop, and support companies and technologies in the cannabis industry. ZAM’s existing clients operate four licensed cannabis cultivation and one processing facility in Washington. Most of the biomass produced by these independent cultivators has been sold historically under the Artizen™ brand, including all-time top selling products in flower in Washington state. Additional information on Artizen-branded products is available online at www.artizencannabis.com. Pervasip additionally owns 5% of KRTL Biotech, Inc., a developer of biotechnologies with a focus on pharmaceutical applications of cannabinol and psilocybin. Additional information on KRTL is available online at www.krtlbiotech.com. $GBHL
|
Followers
|
157
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
10838
|
|
Created
|
10/20/10
|
Type
|
Free
|
| Moderator StocktraderDK | |||
| Assistants budfoxfun | |||

| Date : | Symbol : | PPS Called : | PPS Reached : | % Gains From Call : | Board Post # : |
| 11/13 | SRNE | $6,45 | |||
| 12/2 | IDEX | $2.01 | |||
| 11/12 | FSR | $14.1 | |||
| 01/27 | PDSB | $2.65 |
History Calls :
| Date : | Symbol : | PPS Called : | PPS Reached : | % Gains From Call : | Board Post # : |
| THRA | .0013 | .0035 | 169% | 958 | |
| NWGC | .0855 | .12 | 40% | 943 | |
| SHAR | .013 | .019 | 46% | 1000 | |
| SPQS | .0009 | .0014 | 55% | 1140 | |
| NNAN | .0026 | .0038 | 46% | 1286 | |
| ACYD | .021 | .0444 | 111% | 1329 | |
| BUCS | .053 | .087 | 64% | 1375 | |
| UYMG | .0077 | .01 | 29% | 2150 | |
| BRGO | .03 | .145 | 383% | 1775 | |
| EVXA | .0002 | .0005 | 150% | 2039 | |
| PAOS | .03 | .19 | 533% | 3284 | |
| SNET | .035 | .059 | 68% | 3394 | |
| UPZS | .105 | .29 | 176% | 4112 | |
| MYRY | .0145 | .035 | 5178 | ||
|
Posts Today
|
0
|
|
Posts (Total)
|
10838
|
|
Posters
|
|
|
Moderator
|
|
|
Assistants
|
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
