Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
It’s really easy to comment retrospectively, especially when looking at a chart.
It would be more appreciated if you could provide us with some insight on how you see the future value of the company over the next few years.
Who knows, there may even be a period where the share price is likely to turn around and you could capitalise on the growth? Given the share price weakness, as you’ve pointed out, could there be value now?
You came here just to say that? Btw, I'm back with a new small position of RCEL just to keep it up on my screen. Won't make me rich or broke.
@ 18:37, Perry mentions that they anticipate FDA submission of the PMA in the second half of this year for vitiligo.
@ 20:29, Perry mentions that they anticipate FDA submission of the PMA in the second half of this year for soft tissue reconstruction with approval in the second half of next year.
It's posted on the company website under Investors/Events/More Events. Highly recommend listening, answers many of the questions people are asking. I'd at a minimum listen from minute 32 on.
Is RCEL the worst stock of the year? Was just looking at its performance over various time frames.
2022 YTD Down -37%
Performance 12 months Down -63%
This Zoom webinar will start @ 7pm central time, approximately 90 minutes from now.
RECELL® System Vitiligo Clinical Trial Protocol to be Presented at the Global Vitiligo Foundation Annual Scientific Symposium
PDF Version
VALENCIA, Calif. and MELBOURNE, Australia, March 23, 2022 (GLOBE NEWSWIRE) -- AVITA Medical, Inc. (NASDAQ: RCEL, ASX: AVH), a regenerative medicine company that is developing and commercializing a technology platform that enables point-of-care autologous skin restoration for multiple unmet needs, announced today that an abstract highlighting the clinical trial protocol utilizing the RECELL® Autologous Cell Harvesting Device (RECELL® System) to treat vitiligo will be presented at the Global Vitiligo Foundation Annual Scientific Symposium. The conference is being held in Boston and will highlight research and knowledge of both up and coming vitiligo researchers and those who have and continue to build the foundational knowledge about vitiligo.
“We are pleased to share our clinical study protocol, which aims to establish safety and effectiveness for repigmentation of stable vitiligo lesions utilizing the RECELL System, with leaders of the vitiligo community,” said Dr. Mike Perry, Chief Executive Officer of AVITA Medical. “With enrollment of our vitiligo pivotal trial complete, we look forward to sharing results of our blinded, randomized study in the near future and working with the FDA to bring our novel treatment option to patients.”
RECELL System Abstract
March 24 at 9:38am, Abstract #4: RECELL Autologous Cell Harvesting Device: A Review of the Science of Autologous Skin Cell Suspension and Clinical Protocol Aimed in Establishing Safety and Effectiveness for Repigmentation of Stable Vitiligo Lesions. Authors: K. Bush, A. Hopkin, E. Kirshner, A. Quick
Vitiligo is a disease that attacks pigment-producing cells, called melanocytes, resulting in their destruction or malfunction. The result is a loss of pigmentation in patches of skin. Vitiligo affects up to 2% of the population worldwide,i including an estimated 3-6.5 million Americans.ii Vitiligo has a comparable market size and psychosocial impact to other major dermatology diseases including psoriasis (thick, scaly skin) and atopic dermatitis (red, cracked skin).iiiivv Like these diseases, those living with vitiligo may suffer from poor body image along with low self-esteem, leading to an impaired quality of life.
ABOUT AVITA MEDICAL, INC.
AVITA Medical is a regenerative medicine company with a technology platform positioned to address unmet medical needs in burns, chronic wounds, and aesthetics indications. AVITA Medical’s patented and proprietary collection and application technology provides innovative treatment solutions derived from the regenerative properties of a patient’s own skin. The medical devices work by preparing a RES® REGENERATIVE EPIDERMAL SUSPENSION, an autologous suspension comprised of the patient’s skin cells necessary to regenerate natural healthy epidermis. This autologous suspension is then sprayed onto the areas of the patient requiring treatment.
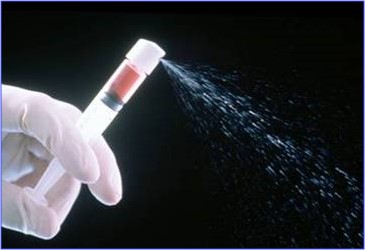
AVITA Medical’s first U.S. product, the RECELL® System, was approved by the U.S. Food and Drug Administration (FDA) in September 2018 and a new ease-of-use design was approved in 2022. The RECELL System is indicated for use in the treatment of acute thermal burns. The RECELL System is used to prepare Spray-On Skin™ Cells using a small amount of a patient’s own skin, providing a new way to treat severe burns, while significantly reducing the amount of donor skin required. The RECELL System is designed to be used at the point of care alone or in combination with autografts depending on the depth of the burn injury. Compelling data from randomized, controlled clinical trials conducted at major U.S. burn centers and real-world use in more than 10,000 patients globally reinforce that the RECELL System is a significant advancement over the current standard of care for burn patients and offers benefits in clinical outcomes and cost savings. Healthcare professionals should read the INSTRUCTIONS FOR USE - RECELL® Autologous Cell Harvesting Device (https://recellsystem.com/) for a full description of indications for use and important safety information including contraindications, warnings, and precautions.
In international markets, our products are marketed under the RECELL System brand to promote skin healing in a wide range of applications including burns, chronic wounds, and aesthetics. The RECELL System is TGA-registered in Australia and received CE-mark approval in Europe. To learn more, visit www.avitamedical.com.
AVITA invites you to attend its investor presentation.
Time: Mar 24, 2022 11:00 AM
https://zoom.us/webinar/register/WN_LgAAB_tyQGS6vyEPG1yAgw
Funds will support completion of the ongoing pivotal clinical trial
VALENCIA, Calif. and MELBOURNE, Australia, March 21, 2022 (GLOBE NEWSWIRE) -- AVITA Medical, Inc. (NASDAQ: RCEL, ASX: AVH), a regenerative medicine company that is developing and commercializing a technology platform that enables point-of-care autologous skin restoration for multiple unmet needs, announced today that the Biomedical Advanced Research and Development Authority (BARDA) has modified its existing contract with the Company to support AVITA Medical’s clinical trial in soft tissue reconstruction. BARDA is a part of the Office of the Assistant Secretary for Preparedness and Response (ASPR) at the U.S. Department of Health and Human Services (HHS).
“We are extremely pleased that BARDA is supporting advanced treatment options for soft tissue reconstruction,” said Dr. Mike Perry, Chief Executive Officer of AVITA Medical. “The RECELL System has already proven itself as a safe and effective tool for those with burns, and we are committed to expanding its use to include all acute wounds. We are pleased BARDA recognizes the potential it holds for a broader group of patients experiencing trauma. BARDA has been an outstanding partner, and we are excited to continue our work to expand the indication for the RECELL System with their support.”
Soft tissue reconstruction is of particular concern to BARDA and AVITA Medical, as skin grafting, the current standard of care for soft tissue reconstruction, requires the harvesting of donor skin which can result in an additional wound to the patient. Significant pain, delayed healing, risk of infection, the need for multiple procedures, discoloration and scarring are associated with donor site wounds. While skin grafting is commonly associated with burn treatment, in 2017, approximately 80% of acute wounds that required skin grafting were non-burn related injuries accounting for more than 200,000 procedures in the U.S.i
AVITA Medical is currently completing a pivotal trial for the use of the RECELL System for soft tissue reconstruction. Currently, the RECELL System is indicated in the U.S. for treatment of acute thermal burns. The clinical trial will compare the clinical performance of conventional autografting to that of widely meshed autografting with the RECELL System on acute non-burn full-thickness skin defects, with the goal of demonstrating that less donor skin is needed without compromising healing outcomes. Topline data from the trial will be shared later this year.
AVITA Medical has had a long-term positive relationship with BARDA since September 2015 and was of fundamental importance to the Company being able to achieve premarket approval for the RECELL System in late 2018.
This project has been funded in whole or in part with Federal funds from the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority, under Contract No. HHSO100201500028C
ABOUT AVITA MEDICAL, INC.
AVITA Medical is a regenerative medicine company with a technology platform positioned to address unmet medical needs in burns, chronic wounds, and aesthetics indications. AVITA Medical’s patented and proprietary collection and application technology provides innovative treatment solutions derived from the regenerative properties of a patient’s own skin. The medical devices work by preparing a RES® REGENERATIVE EPIDERMAL SUSPENSION, an autologous suspension comprised of the patient’s skin cells necessary to regenerate natural healthy epidermis. This autologous suspension is then sprayed onto the areas of the patient requiring treatment.
AVITA Medical’s first U.S. product, the RECELL® System, was approved by the U.S. Food and Drug Administration (FDA) in September 2018 and a new ease-of-use design was approved in 2022. The RECELL System is indicated for use in the treatment of acute thermal burns. The RECELL System is used to prepare Spray-On Skin™ Cells using a small amount of a patient’s own skin, providing a new way to treat severe burns, while significantly reducing the amount of donor skin required. The RECELL System is designed to be used at the point of care alone or in combination with autografts depending on the depth of the burn injury. Compelling data from randomized, controlled clinical trials conducted at major U.S. burn centers and real-world use in more than 10,000 patients globally reinforce that the RECELL System is a significant advancement over the current standard of care for burn patients and offers benefits in clinical outcomes and cost savings. Healthcare professionals should read the INSTRUCTIONS FOR USE - RECELL® Autologous Cell Harvesting Device (https://recellsystem.com/) for a full description of indications for use and important safety information including contraindications, warnings, and precautions.
In international markets, our products are marketed under the RECELL System brand to promote skin healing in a wide range of applications including burns, chronic wounds, and aesthetics. The RECELL System is TGA-registered in Australia and received CE-mark approval in Europe. To learn more, visit www.avitamedical.com.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This press release includes forward-looking statements. These forward-looking statements generally can be identified by the use of words such as “anticipate,” “expect,” “intend,” “could,” “may,” “will,” “believe,” “estimate,” “look forward,” “forecast,” “goal,” “target,” “project,” “continue,” “outlook,” “guidance,” “future,” other words of similar meaning and the use of future dates. Forward-looking statements in this press release include, but are not limited to, statements concerning, among other things, our ongoing clinical trials and product development activities, regulatory approval of our products, the potential for future growth in our business, and our ability to achieve our key strategic, operational and financial goal. Forward-looking statements by their nature address matters that are, to different degrees, uncertain. Each forward-looking statement contained in this press release is subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statement. Applicable risks and uncertainties include, among others, the timing of regulatory approvals of our products; physician acceptance, endorsement, and use of our products; failure to achieve the anticipated benefits from approval of our products; the effect of regulatory actions; product liability claims; risks associated with international operations and expansion; and other business effects, including the effects of industry, economic or political conditions outside of the company’s control. Investors should not place considerable reliance on the forward-looking statements contained in this press release. Investors are encouraged to read our publicly available filings for a discussion of these and other risks and uncertainties. The forward-looking statements in this press release speak only as of the date of this release, and we undertake no obligation to update or revise any of these statements.
This press release was authorized by the review committee of AVITA Medical, Inc.
That, of course, is not true. This stock is rubbish. I looked at RCEL, the ridiculous salaries paid, and immediately decided to pass. And the sooner one sells the better. It's not like it's generating fat profits and paying dividends.
I agree with you ... with the news of the past month being all positive there is no other explanation except MM's are short on shares. It never occurs to them that working to get the price moving upward would generate more opportunities to acquire shares ... ;)
As I said before no one has brought down at these levels so this must be manipulation by mms not selling
But they aren't profitable yet so they are using the cash. Plus we are in a bear market.
There are some biotech's trading at under cash with a 3-4 year amount of cash.
Over 50% of the market cap is cash and cash equivalents. Go figure.
I never though I'd see my wife's long position from September 2018 turn red ($7.86 cost basis). Sad, but still hopeful. GLTA
our day will come ... ;)
Can’t see why people selling down here no point no one brought this low
this low does not make sense to me either……If war breaks out….you would think there would be a greater demand….right?……why would anyone sell this stock at this low price knowing what we know about it?
One forward Two back. We are fighting quite strong resistance. Covid, the war, inflation, volatile markets. Avita needs an industry shaker like Japan's vitiligo trial completion and acceptance.
Any idea why the the new lows
Well AVH has followed RCEL back down to the equivalent of $8.65 so they’re back on par.
Cowen Conference Presentation:
https://hotcopper.com.au/documentembed?id=uOMxKKzFkiWRTLKhOROKAxjvSTYP6we%2FzxaZoeFxke92GA%3D%3D
Agree, couldn't have said it better myself.
Melody will have a more accurate view.
My guess is although the recent news will bode well for the company in the future, I think it comes down to revenue. The market wants to see higher revenue growth than the 20% YoY guidance.. 20% QoQ would turn the share price around.
In the quarterly call, the CFO mentioned a potential future capital raise in 2023-24 which probably doesn’t help either, even if it’s to fund the future E.B and Rejuvenation multi billion dollar markets.
In saying that, I think the company will follow through with their corporate strategies in the medium to long term which will sort out the share price.
Hi guys
2 good bits of news any reason for the huge drop in share price
Regards Steve
I thought it’s worth noting that there’s currently about a 17% share price difference between AVH (ASX) and RCEL (NAS).
AVH finished at A$3.21 which when converted is equivalent to US$11.74
Calculations: A$3.21 x 5 (5 AVH = 1 RCEL) x 0.7317 ($A to $US exchange rate) = US$11.74.
Highlights of the new agreement ...
AVITA Medical Inc (NASDAQ:RCEL) has been awarded a group purchasing agreement with Premier Inc (NASDAQ:PINC)
The new agreement allows Premier members, at their discretion, to take advantage of special pricing and terms pre-negotiated by Premier for the RECELL System for the treatment of adult and pediatric patients with severe burns.
The RECELL System is a device that enables healthcare professionals to produce a suspension of Spray-On Skin Cells using a small sample of the patient's skin to treat acute thermal burns.
This new agreement enables AVITA Medical access to Premier's member hospitals and regional representatives to help facilitate the adoption of the RECELL System into their facilities.
Premier is a leading healthcare improvement company, uniting an alliance of approximately 4,400 U.S. hospitals and 225,000 other providers.
Last month, the FDA approved the premarket approval application supplement for RECELL Autologous Cell Harvesting Device, an enhanced RECELL System that provides clinicians with a more efficient user experience and simplified workflow.
The launch of the new RECELL System in the United States will begin in Q2 2022.
Price Action: RCEL shares are up 5.22% at $10.48 on the last check Thursday.
This rating was given 2 days before the announcement this afternoon, but after the Japan distribution announcement:
BTIG Thinks Avita Medical’s Stock is Going to Recover
Brian Anderson
Feb 28, 2022, 09:25 PM
BTIG analyst Ryan Zimmerman reiterated a Buy rating on Avita Medical (RCEL – Research Report) yesterday and set a price target of $21.00. The company’s shares closed last Monday at $9.20, close to its 52-week low of $8.15.
According to TipRanks.com, Zimmerman is a 5-star analyst with an average return of 16.3% and a 50.0% success rate. Zimmerman covers the Healthcare sector, focusing on stocks such as Tactile Systems Technology, Organogenesis Holdings, and Zimmer Biomet Holdings.
The word on The Street in general, suggests a Moderate Buy analyst consensus rating for Avita Medical with a $21.00 average price target.
See today’s best-performing stocks on TipRanks >>
Avita Medical’s market cap is currently $225.1M and has a P/E ratio of -9.47.
Based on the recent corporate insider activity of 18 insiders, corporate insider sentiment is negative on the stock. This means that over the past quarter there has been an increase of insiders selling their shares of RCEL in relation to earlier this year.
TipRanks has tracked 36,000 company insiders and found that a few of them are better than others when it comes to timing their transactions. See which 3 stocks are most likely to make moves following their insider activities.
Avita Medical Ltd. engages in the development and distribution of regenerative and respiratory medicine and treatments. It operates through the following segments: Asia Pacific, Europe, and Americas. The Asia Pacific, Europe, and Americas segments involve in the sale of RECELL Devices. The company was founded on December 21, 1992 and is headquartered in Royston, the United Kingdom.
YES Sir and nice to see Avita with back to back agreements ... ;)
It's up 4.82% in after hours trading from the news. One more percent and it will erase today's loss.
This should really help us get back our positive trend ... ;)
Avita Medical, Inc. Signs Agreement with Premier, Inc. to Supply the RECELL® System to Hospitals and Health Systems Across the U.S.
AVITA Medical, Inc. (AVHHL) (NASDAQ: RCEL, ASX: AVH), a regenerative medicine company that is developing and commercializing a technology platform that enables point-of-care autologous skin restoration for multiple unmet needs, announced today that they have been awarded a group purchasing agreement with Premier, Inc. The new agreement allows Premier members, at their discretion, to take advantage of special pricing and terms pre-negotiated by Premier for the RECELL® System for the treatment of adult and pediatric patients that require treatment for severe burns. The RECELL® System is a device that enables healthcare professionals to produce a suspension of Spray-On Skin™ Cells using a small sample of the patient’s own skin for the treatment of acute thermal burns.
“When someone experiences a severe burn injury, time matters, and that is why this agreement with Premier is so important – it will help ensure that patients in need of treatment are able to efficiently and effectively access the RECELL System without delay,” said Dr. Mike Perry, AVITA Medical’s Chief Executive Officer. “As a leading GPO, Premier is a trusted partner among their alliance of members and AVITA Medical is committed to building on that trust by providing quality products to help ensure the best patient care possible. This agreement represents a major milestone for AVITA Medical as it puts our innovative RECELL System into the hands of those caring for burn patients across the U.S.”
This new agreement enables AVITA Medical access to Premier’s member hospitals and regional representatives to help facilitate the adoption of the RECELL System into their facilities.
Premier is a leading healthcare improvement company, uniting an alliance of approximately 4,400 U.S. hospitals and 225,000 other providers to transform healthcare. With integrated data and analytics, collaboratives, supply chain solutions, consulting and other services, Premier enables better care and outcomes at a lower cost.
Avita Medical, Inc. Signs Agreement with Premier, Inc. to Supply the RECELL® System to Hospitals and Health Systems Across the U.S.
AVITA Medical, Inc. (AVHHL) (NASDAQ: RCEL, ASX: AVH), a regenerative medicine company that is developing and commercializing a technology platform that enables point-of-care autologous skin restoration for multiple unmet needs, announced today that they have been awarded a group purchasing agreement with Premier, Inc. The new agreement allows Premier members, at their discretion, to take advantage of special pricing and terms pre-negotiated by Premier for the RECELL® System for the treatment of adult and pediatric patients that require treatment for severe burns. The RECELL® System is a device that enables healthcare professionals to produce a suspension of Spray-On Skin™ Cells using a small sample of the patient’s own skin for the treatment of acute thermal burns.
“When someone experiences a severe burn injury, time matters, and that is why this agreement with Premier is so important – it will help ensure that patients in need of treatment are able to efficiently and effectively access the RECELL System without delay,” said Dr. Mike Perry, AVITA Medical’s Chief Executive Officer. “As a leading GPO, Premier is a trusted partner among their alliance of members and AVITA Medical is committed to building on that trust by providing quality products to help ensure the best patient care possible. This agreement represents a major milestone for AVITA Medical as it puts our innovative RECELL System into the hands of those caring for burn patients across the U.S.”
This new agreement enables AVITA Medical access to Premier’s member hospitals and regional representatives to help facilitate the adoption of the RECELL System into their facilities.
Premier is a leading healthcare improvement company, uniting an alliance of approximately 4,400 U.S. hospitals and 225,000 other providers to transform healthcare. With integrated data and analytics, collaboratives, supply chain solutions, consulting and other services, Premier enables better care and outcomes at a lower cost.
Avita is still owned by alot of investors in Australia. So, if it was down in Australia (last night our time) then there is a good chance will will memic them. Until the stock is more heavily traded in the U.S. it will probably remain the same ... jmho
market is up today…..RCEL is down?
Our pre-market Bid is $ 9.90 and Ask is $ 11.43 ... looks good to me ...;)
The Aussie stock closed up 25.68% tonight. I'm really curious what the stock will do tomorrow at the morning bell.
That is great news ... hopefully we follow suit ... ;)
The Aussie stock is up 21% so far tonight.
Me too ... hope they can begin to recover from their down trend. Wonderful product for those who need it the most.... ;)
Yup...up 6.89% in after hours trading. Curious to see what will happen at the morning bell tomorrow.
|
Followers
|
102
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
7485
|
|
Created
|
06/20/12
|
Type
|
Free
|
| Moderators | |||


| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
