Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
If it is they injected the same volume of liquid with twice the amount of toxin it means the toxin would diffuse at the same rate and to the same distance away as before.
Moreover, doubling the amount of active toxin administered to a fixed region of the face would be expected to produce a higher rate of ptosis, which EOLS’ dataset did not show—a red flag that something is amiss.
it’s pretty obvious to me that double-dose Jeuveau is a ploy that EOLS engaged not to further its own business, but rather to create confusion in the marketplace about Daxxify and thereby impede Daxxify’s commercial uptake. On this score alone, EOLS may have been successful in accomplishing what it hoped to accomplish, but the impeding of Daxxify’s uptake will be transient. A year from now, nobody will be talking about double-dose Jeuveau.
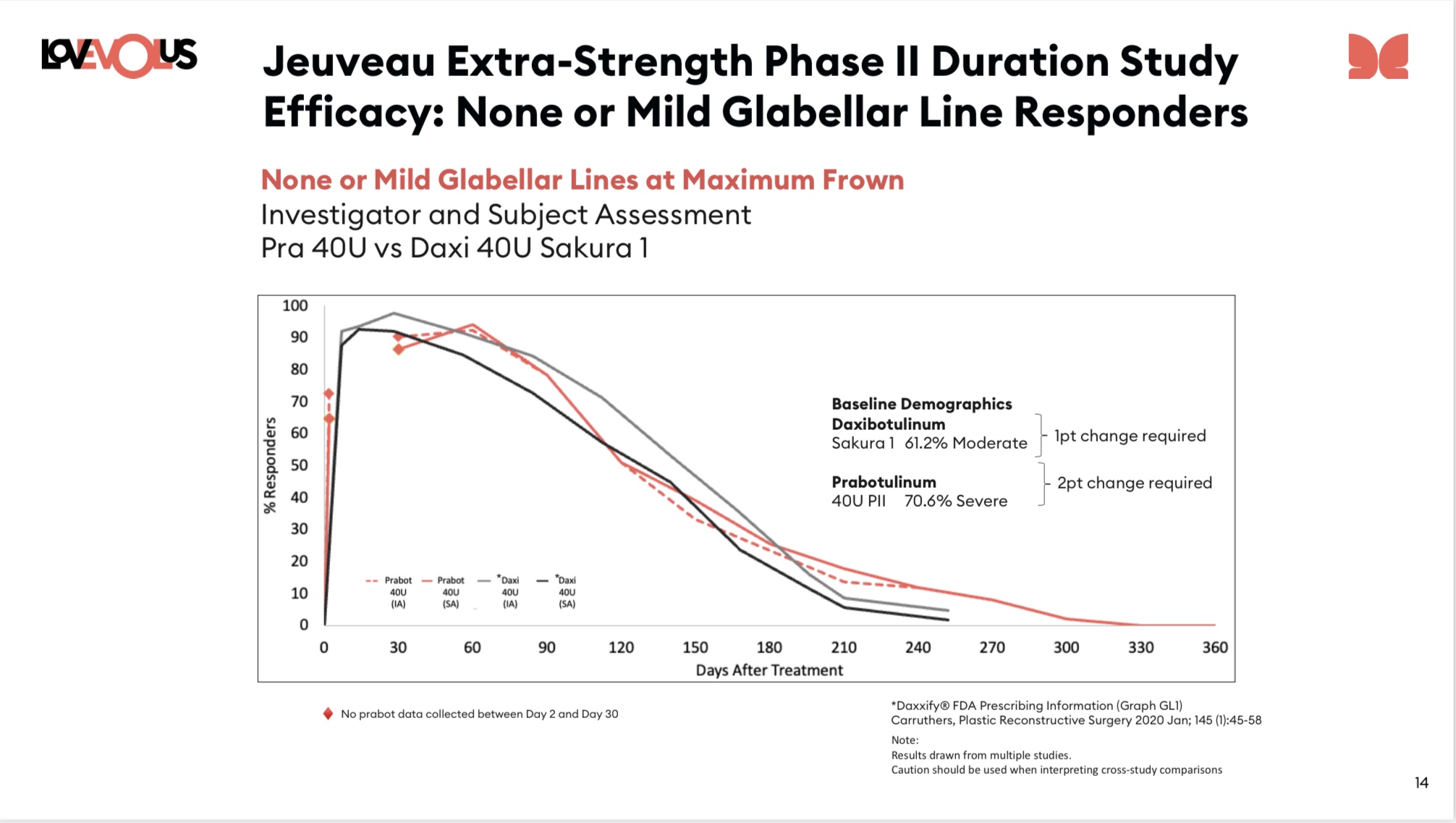
EOLS - Here are the pictures from the paper. I've now done more than a 'quick check' for *one* of the Jeuveau 40 unit curves (the Investigator curve - but not the Patient curve).
For the investigator curves given below (left curve) it is, as best it's possible for me to measure, identical to the 23Q3 version of the same (investigator) curve out through 120 days (which is also the median - about 15 days later than 20 unit dose of Jeuveau, and about 30 days later than botox dose and 23 days earlier than Sakura 1). But from there on the formally published paper is meaningfully lower than the 23Q3 Jeuveau 40 unit dose curve, although still above the Sakura 1 dose from day 190 on.
Essentially: Sakura 1 has a better median by about 23 days (but beware the caveats below), but the last quarter of the population seems to be about the same for Daxxy and Jeuveau 40 unit dose
And the caveats:
a) The comparison to Sakura is a cross trial comparison... inherently noisy. See next point.
b) The Jeuveau 40 unit population is a meaningfully tougher population in which to get no-or-mild-lines than either of the Sakura populations (Jeuveau is 70% Severe, Sakura closer to only 40% Severe).
And some corrections for which I ran out of edit time:
A) A link to the EOLS paper that, hopefully, won’t time out.
B) I mistakenly said Figures 8 and 9 (the EOLS figures in the above link that are comparable to Sakura figures) were the last figures. They aren’t. There are three more figures in the EOLS paper (10, 11 and 12.. obviously). Another ‘my bad’, although not really pertinent to any argument. This is just an administrative clean up by me. C’est la vie.
EOLS and RVNC
In other words, EOLS declined to publish what they said they would, which is consistent with what I posited in #msg-173201637.
Right, EOLS is using an different and easier metric to fool uninformed people. Thanks for pointing that out.
My guess is the entire double dose strategy was not to make money on a longer duration formulation per se, but rather for competitive reasons to upend any perception that daxi was special or unique
Price them both the same and office makes $34 more profit with Daxxify.
Hmm… would they need P3 study and BLA for this?
EOLS Extra Strength
As for Dew’s assertion that they will never publish… I’d wager they do publish (paper or poster), but there is no urgency to do so until they feel Daxxy is starting to impact their growth.
Therapeutics might be the only way out of this mess.
Why would either a patient or injector make a video like that? Makes absolute zero sense. You want an injector to say... I'm an idiot and didn't know what I was doing, but now I do? Or you want a patient to say I went to a really bad injector but then found a new injector who knows what they are doing?
Instagram - I’ve yet to see a *recent* Instagram of either a patient or injector who made reference to fact that initially they had problems, but decided to give it another try with a more experienced injector and it worked great.
Revance is going to need a lot more of this (above) kind of patient or injector.
Is there any way for a patient to determine whether or not her injector was untrained, improperly trained, or inadequately trained?
In my opinion, it’s more likely that she will blame the result on Daxxify. … loudly and vociferously to many others.
Ian is clearly talking non-sense (on the topic of whether Daxxy lasts longer)
We get it that you want Foley fired
ALPN - another company where someone posted on this board
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=99588181
I don’t know what concentration is reached at the cornea and 26 micrograms of active ingredient sounds small but it’s roughly the size of an eyedrop. (I know this as I instill my glaucoma meds with a micropipette set at 12 mcg). And a bimatoprost eyedrop is .03% active ingredient, so 26 mcg is about 900 x the active ingredient in one drop. Of course, it’s released over about 180 days.
The implant was designed to release drug for 3–4 months.
Drug release from the implant is complete within 90 days
in vitro, and an in vivo study using beagle dogs administered
a 15 µg implant similarly showed complete drug
release and intraocular tissue drug levels below the limit
of detection by 4.2 months after implant administration
[23]. A drug-distribution study using beagle dogs further
showed that drug concentrations achieved in the
iris-ciliary body (a target tissue for IOP lowering) were
4400-fold higher after intracameral administration of
a 15 µg bimatoprost implant than after 7 days of daily
application of bimatoprost 0.03% eye drops [25]. In contrast,
drug distribution to the bulbar conjunctiva, eyelid
margins, and periorbital fat (tissues associated with
topical PGA-related side effects) was below detectable
levels or limited after bimatoprost implant administration
compared with topical dosing [25]. T
On the other hand, Durysta doesn’t seems to be fully absorbed, see this discussion, so that might explain why repeated dosing is problematic:
OCUL Glaucoma
FWIW - both glaucoma inserts on the market, the second having just been approved, have a label restriction disallowing use more than once in an eye. For at least Durysta this was not the original plan; it just showed up in the later/bigger trials that repeated use showed problems. My point is that this was not obvious in the earlier/smaller trials that are more like this OCUL one, and I haven’t yet seen strong data on what small trial, single use proxies accurately predict that reuse risk in the later ph3.
Small trial, but the slight improvement in the tx arm vs decline in the control arm translated to a p-value of.007.
I don't see why they can't adjust the price upward as long as Daxxify offers real benefits to injectors and consumer. (aesthetics side).
Why can’t they raise prices as therapeutics comes online to levels that are still acceptable to payers (especially if duration Pam’s out saving md visits etc)
The key questions we should ask are whether the initial blew up is a 4 billions plus mistake?
Not easy to get those burned accounts back. Last Q conf call talked about that.
our growth will come from a combination of new account adds, along with ongoing reorders. And as we noted in Q4, more than 2/3 of our revenue came from reordering accounts, which I think points to the stickiness of the product in those accounts where we’re able to secure. So we feel very good about where we are"
the CEO sounds like a straight shooter.
That though should get sorted out through real world experience over time
depends on how fast Daxxify gains market shares, doesn't it?
Don't companies raise prices at some point? I mean Allergan/ABBV just did that earlier in the year.
The best leadership can make mistakes and take time to right the ship, but the last several years have been too many sucker punches to not recognize a pattern and want out.
Speaking of Martin Shkreli he had a cameo on this board right when his news started swirling IIRC
The outgoing CEO of AlmataBio (the private company AVTX is acquiring) is Patrick Crutcher, who used to be a co-moderator of this message board.
IBIO—A second company issued a PR today mentioning both AI and obesity drugs—and it was good for a +192% move:
There is data that shingrix (and before that zostavax) is associated with lower rates of dementia.
RVNC
In 4th q 22 sales were $34.7m and 2023 sales were $30.3, $31.7, $32.1 and $34.5 for the 4 quarters. Total sales year over year were nice but I'm not sure why it looks they hit a wall.
So what do you think RVNC has been doing in the last couple quarters? Aren't they not do that?
I adhere to a diet which baffles me why it is so hard for others to follow.
Repairing an injector relationship takes that injector losing 3 or 4 regular patients to another practice who is offering Daxi (2 Daxi units to 1 Botox) at the SAME price as Botox or any other toxin and their patients are getting double the duration of Botox or Jeaveau .
RVNC is not going to spend money on a migraine trial anytime soon (if ever).
